Аннотация
In assessing the nutritional value of a food product, its fatty acid composition is important. Meat products contain essential fatty acids that positively influence human health, and saturated fatty acids, the high level of which does not allow them to be attributed to healthy nutrition. The article describes the use of raw meat from various species of animals and poultry according to their fatty acid composition; presents the possibility of changing the composition of fatty acids to produce healthy food. The presence of trans-isomer fatty acids (TIFA) in food products is the risk factor for human health. Meat raw materials contain trans fatty acids of natural and industrial origin. The results confirming trans fatty acids formation at the stage of animal growth (biohydrogenation and as the result of the use of growth promoters) and during meat processing (heat treatment at high temperatures, storage, use of sodium nitrite) are presented. At present, meat products are not considered to be a source of health-threatening TIFA, but the results presented in the review confirm the need for further research to identify factors that affect the fatty acids isomerization process in meat and semi- meat products and the normalization of their content in finished productsКлючевые слова
Meat raw materials, fatty acids, trans fatty acids, safety, trans-isomerization, biological efficacyВВЕДЕНИЕ
Fat is one of the main components of meat, upon which the nutritional value and functional properties of the finished product largely depend. Food product, depending on the characteristics of the fat contained in it (the level of content in the product, the qualitative and quantitative composition), can be attributed to the consumer product of functional, therapeutic and preventive action, or adversely affecting health. Along with the high level of saturated fatty acids, meat and meat products contain TIFA [1, 2]. Due to the negative influence of some TIFA on health [3, 4], in many countries around the world the issue of limiting their content in food products has been resolved at the legislative level. Various conditions have been determined that affect the formation of TIFA in meat products [5, 6, 7, 8, 9], depending on which the TIFA are divided into natural and industrial. Therefore, the problem of the normalization of natural TIFA, which are synthesized in the animal's organism and have a beneficial effect on human health remains important.
In order to recommend the use of meat raw materials for functional and dietary nutrition, the biological effectiveness of meat lipids of various species of productive animals and poultry was assessed, the indices of the content of TIFA in various types of meat were shown, and technological factors affecting their formation were revealed.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
The quality of the fat tissue of a productive animal largely depends on the species of the animal, the breed, and the diet. The process of digestion and formation of tissues in ruminant and monogastric animals take place in different ways, as a result of which the composition of the fatty acids in the meat of these animals is not the same. In animals with a single-chamber stomach, fatty acids from the diet undergo slight transformation during digestion and absorption, therefore, the fatty acid composition of the tissues of animals with a single-chamber stomach can be regulated by the fat-acid composition of the diet. It is very difficult to predict the composition of fatty acids in ruminants. Attempts to modify the fatty acid profile require simultaneous study of both the diet of productive animals, especially ruminants, and the mechanisms of bioconversion in the stomach (rumen) of animals [10, 5].
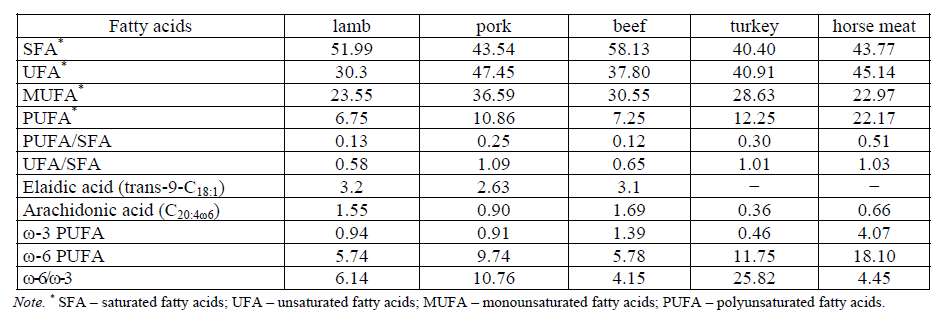
Specialists of V.M. Gorbatov's All-Russian Meat Research Institute (VNIIMP) summarized and systematized the experimental data on the composition of fatty acids in various types of raw materials obtained from farm animals and birds [11]. For the analysis we used m. longissimus dorsi muscle tissue samples, selected from adults of sheep, pigs, cows, turkeys and horses. The obtained results showed the presence of significant qualitative and quantitative differences in the lipid composition of muscle tissue of various animal and poultry species: cattle, pigs, sheep, horses and turkeys (Table 1).
Saturated fatty acids (SFA) in animal raw materials are mainly represented by myristic, palmitic and stearic acids [11, 2, 12, 13, 6, 14, 15]. The lipid fraction of beef is characterized by the maximum level of SFA (58.13%), the lower content of SFA is in the lipid fraction of lamb (51.99%). Lipids of muscle tissue of pork and horse meat have approximately the same amount of these fatty acids (43.54 and 43.77%, respectively). The lowest content of SFA among the studied meat samples was obtained in the lipid fraction of turkey meat.
Monounsaturated fatty acids (MUFA) are basically palmitoleic, oleic fatty acids and its trans-isomer – elaidic fatty acid [11, 2, 12, 13, 6, 14, 15]. The lipid fraction of pork contains the highest level of MUFA (36.59%), then in descending order – the lipid fractions of beef < turkey < lamb < horse meat (Table 1).
Polyunsaturated fatty acids (PUFA) are mainly: linoleic, linolenic and arachidonic fatty acids [11, 2, 12, 13, 6, 14, 15]. The highest amount of PUFA is contained in the lipid fraction of horse meat (22.17%), almost three times less – in the lipid fraction of beef and lamb (7.25 and 6.75%, respectively) and about two times less – in the lipid fraction of pork (11.49%). In contrast to plant fats, animal fats are characterized by a high content of arachidonic acid (0.36–1.69%).
The balance of the fatty acid composition was evaluated by the ratio of PUFA/SFA, the optimum range of which should be from 0.2 to 0.4 and UFA/SFA – 2.3 : 1 [16]. The fatty acid composition of the lipids of the muscle tissue of pork, horse meat and turkey is more balanced than that of lamb and beef (see Table 1).
The optimal ratio of PUFA of the ω-6 and ω-3 family plays an important role in healthy nutrition. The diet is considered healthy if the ratio of ω-6 to ω-3 PUFA is 10 : 1, is considered to be a therapeutic nutrition with ratios ranging from 3 : 1 to 5 : 1. According to the data of Table 1, horse meat and beef meet the requirements for raw materials for preventive and therapeutic nutrition, all other types – for raw materials for general purpose products. Lipid fraction of horse meat is characterized by a high content of ω-3 PUFA. Belaunzaran et al. associate the accumulation of ω-3 PUFA in horse meat with a unique digestive system of horses [15].
The high content of oleic acid trans-isomer-elaidic acid (trans-9-С18:1) [11] in the intramuscular fat of lamb (3.2%), beef (3.1%) and pork (2.63%) deserves attention. In deer fat, this indicator was 2.2%, in chicken – 1.4%, in maral fat – 0.9%, in wild boar fat – 0.4%, in seal fat – 0.3% [2]. In the lipid fraction of turkey meat and horse meat, this isomer was not detected [11]. Some researchers report a low content of TIFA in horse meat, the level of which depends on the diet [15]. In the meat of maral, elaidic acid was detected in two muscles of the three examined (quadriceps muscle – 0%, hip muscle – 1.68%, longissimus muscle – 1.04%). The content of TIFA in meat of kangaroo, african ostrich, mallard, pheasant, sea animals (Lahtak, walrus, seal, gray whale) was not determined [17].
The review, presented by Brazilian scientists, reports that the content of natural TIFA in food products (milk, lamb and beef) varies from 3 to 8% of the total amount of fatty acids [18]. According to Stender S. et al (2008), trans fatty acids of natural origin can account for up to 6% of fat in meat and dairy products obtained from ruminants (cattle, sheep, goats, buffaloes, deers, elks, giraffes and camels) [19].
In this paper, the authors adhere to the following definition of TIFA – a type of unsaturated fats being in the trans configuration, that is, having hydrocarbon substituents on opposite sides of the carbon-carbon double bond.
The studies of recent years in the field of TIFA in food products are focused on determining the conditions for their occurrence and their impact on human health.
Natural and industrial sources of TIFA have different effects on the human body. Natural transisomers are not a risk factor for cardiovascular diseases, because, unlike industrial ones, they do not contribute to the reduction of high-density lipoproteins and increase in low-density lipoproteins [20, 21] and are not associated with coronary heart disease [22].
Conjugated linoleic acid (CLA) has anticarcinogenic and anti-allergic properties [23]. CLA is a mixture of fatty acid isomers of the trans and cis configurations, and, therefore, it is difficult to establish the mechanism of action of each of these isomers on the immune function of humans and it is still unexplored. Among them, some trans-isomer fatty acids have a positive effect on human health, others do not have biological activity, and some, like industrial trans fats, can be harmful to health [24].
In the CLA study, a mixture of cis-9-trans-11-CLA isomers (predominant in milk and beef) and trans-10-cis-12-CLA was used. In experiments on laboratory animals, evidence has been obtained that two isomers of CLA (C18:2 cis-9, trans-11 and C18:2 trans-10, cis-12) inhibit the carcinogenesis of the mammary gland and large intestine in animals. The mechanisms consist in the anti-angiogenic activity, induction of apoptosis in cancer cells and changes in fat metabolism [25]. In experiments on rats and chickens, CLA's ability to activate the immune system has been proven (no convincing human subject research has been conducted). The results of some studies of the effect of different CLA isomers on the immune function led to the assumption that the isomeric trans-10-cis-12-C18:2 has the immunological activity, rather than cis-9-trans-11-C18:2 [24]. In young rats, CLA can act as a growth factor and promote the release of energy from fat stores. Human subject research has shown that a diet rich in natural CLA leads to weight loss [5].
Rumenic acid (cis-9-trans-11-C18:2) has an anticancer effect. The positive effect of its action on different types of cancer has been obtained: breast, skin, and gastrointestinal tract cancer. On the contrary, linoleic acid (C18:2) exerts a stimulating effect on cancer cells. Scientists argue that the high level of consumption of CLA, and rumenic acid in its composition, provides antitumor protection due to the fact that this substance replaces linoleic acid in some lipoproteins, which in large quantities increases the risk of cancer in rodents. In experiments on animals, it has been shown that the addition of rumenic acid to feed reduces the formation of plaques in the aorta and reduces existing ones, significantly lowers the level of low-density lipoprotein cholesterol, inhibits the development of atherosclerosis, reduces inflammation [26, 27].
The precursor of rumenic acid - vaccenic acid (trans-11-C18:1) also has a positive effect in oncology. Running an experiment one should take into account the fact that vaccenic acid is easily converted into rumenic, and its positive effects can be justified [28]. Final conclusions about the anti-cancer properties of these natural TIFA can be made after studying their influence on cancer risk markers in humans.
The results of studies of the effect of natural TIFA on health do not give an unambiguous answer to the question of the mechanisms of their action in comparison with industrial ones, but negative effects on the functions of the organism have not been established [27, 29], possibly because of their small content in products [20].
Unlike natural TIFA, the consumption of excessive amounts of non-natural, industrial TIFA with food leads to a risk of developing coronary heart disease, and more than any other source of nutrients, abdominal obesity, diabetes, Alzheimer's disease, breast cancer, reproductive harm, endometriosis and cholelithiasis [30, 31].
Consumption of TIFA during pregnancy and lactation period can lead to deviations from normal fetal and postnatal development, which can cause the development of metabolic diseases [32]. People who have low social status, who buy cheap food with a higher content of industrial trans fats fall into the risk group [33, 34, 35].
Based on the clinical trials conducted, WHO experts concluded that consumption of trans fatty acids increases the risk of cardiovascular disease and coronary heart disease due to adverse effects associated with increased levels of low-density lipoproteins, decrease in high-density lipoprotein levels, provocation of inflammation and endothelial dysfunction, and also influence on coagulability of blood, decrease in sensitivity of cells to insulin, replacement of essential fatty acids in cell membranes, which leads to dysfunction of processes associated with prostanoids and key functions performed by membranes [29].
There are several ways of forming TIFA in meat products: natural, as a result of synthesis in the rumen of the animal, and in the process of industrial processing of raw materials (industrial transisomers of fatty acids).
The natural way of forming trans-forms of fatty acids in meat raw materials is the biohydrogenation of unsaturated fatty acids under the influence of the microflora of the rumen. Mapiye C. et al. (2012) presented material on the mechanisms of synthesis of trans-isomers and ways to increase their content in beef [5]. In the rumen, the incoming feed lipids are hydrolyzed by plant or microbial lipases, resulting in the formation of free fatty acids. Free fatty acids are toxic to microorganisms, they are detoxified through biohydrogenation (combined isomerization of double bonds and hydrogenation). Rumen microorganisms (mainly bacteria), first of all, biohydrogenate the essential fatty acids (linoleic and linolenic fatty acids) to stearic acid (С18:0). As a result of the experiments, it was found that behenic acid (С22:0) is the main end product of the biohydrogenation of docosahexaenoic acid (С22:6, DHA). Different conditions in the rumen can lead to a series of biohydrogenation intermediate substances with one or two double bonds with different cis and trans configurations.
At present, meat of animals grown using intensive technologies in industrial-scale volumes contains a lower level of natural cis and trans isomers, in contrast to the meat of a pasture grown animal. The feed system can regulate the level of fatty acids in meat. The proportion of linoleic acid isomers in meat raw materials can be increased using pasture management (0.59% for pasture management and 0.28% for stall barn housing). Moreover, the feed system at the final stage of store feeding affects the fatty acid composition of the meat and the level of isomer content of fatty acids more than at the initial stage [6].
To increase the CLA level in meat, linoleic acid-rich vegetable oils, sunflower seeds or safflower are added to the cattle diet in combination with a high concentrate feed (> 75% of the grain in dry matter). Combination of vegetable oils with grain leads to an increase in vaccenic and rumen acids in beef. When feeding rations with high grain content, trans-10-С18:1 (trans-isomer of octadecenic acid) is produced due to an increase in microbial metabolism in the rumen through an alternative pathway of biohydrogenation [5]. The introduction of flaxseed into the diet of small bulls leads to a significant increase in the content of CLA (cis-9, trans-11) [36].
The level of content, the type of concentrate and the degree of its processing affect the mechanism of biohydrogenation. Small grains, such as wheat and barley with a rapidly fermentable starch, stimulate the production of trans-10-C18:1 fatty acids. Corn starch is more resistant to fermentation and can stimulate a greater accumulation of vaccenic and rumenic acids compared to starch from other cereals [5].
In the experiment of Uruguayan researchers on the influence of a different ratio of hay and concentrates (corn and soy) in the diet on the quality characteristics of meat and the fatty acid composition of intramuscular fat lambs of the Corydale breed, these data were not confirmed [37]. There was no increase in the transisomer content of oleic and linoleic fatty acids from a change in the proportion of cereal concentrates in the diet of lambs.
Regulation of the content of vaccenic acid (trans-11-C18:1) is impossible by changing the profile of fatty acids (SFA, MUFA, PUFA). This was the conclusion of specialists of the Leibniz Institute of Animal Biology [38] in the store feeding experiment of bulls of the Holstein breed with a high proportion of linoleic and alpha-linolenic fatty acids in the diet. Regardless of the level of the MUFA and PUFA in the lipid composition of feed, the content of vaccenic acid was constant. Sausage and pate, produced from meat from grown animals, contained the same number of CLA isomers (cis-9, trans-11 and trans-10, cis-12) that meat raw materials. This indicates that the heat treatment, at which sausages and pates were produced, maintains the level of TIFA in finished products.
Work is under way to increase the content of trans-linoleic acids in livestock products, both through the use of vegetable fats, and through synthetic conjugates of linoleic acid. V.A. Matveyev, V.P. Galochkina et al. (2010) patented a method for increasing the intensity of growth of Kholmogorsky bull calves raised for meat production, using a biologically active substance (BAS) based on conjugated linoleic acid (CLA, C18:2 cis-9, trans-11 isomer). BAS, added to the main diet, changed the biochemical processes in the body of the bull calves (due to the higher activity of the work of the Krebs cycle in the bulls' tissues compared to glycolysis) grown for meat production, and led to an increase in the intensity of their growth [39].
Growth promoters, widely used in modern technologies of growing animals, lead to an increase in both the proportion of trans-forms of fatty acids, and the emergence of mixed cis-, trans-forms [7]. As a result of the study of the effect of the use of the hormonal estrogen regulator diethylstilbestrol introduced into the pig's diet on the isomerization of fatty acids, it was found that animal lipids obtained by the action of forbidden to use diethylstilbestrol contained an increased content of TIFA and an appreciable amount of mixed cis and trans isomers. The authors conclude that a high content of TIFA in meat raw materials may indicate the use of metabolic growth regulators.
The processes of fat oxidation during storage lead to the formation of fatty acid isomers. At the initial oxidation period, the hydrogen atom is stripped from the fatty acid molecule to form a fatty acid radical, which is stabilized by delocalization via double bonds. This leads to a shift in the double bond, in the case of PUFA, by forming conjugated double bonds. Such a shift forms double bonds in both cis and trans configurations, with the predominance of the trans configuration due to its greater stability [40]. The process of storing meat raw materials contributes to an increase in the proportion of TIFA, as well as to the emergence of mixed isomers that contain both cis and trans forms [7]. Significant contribution to the process of cis-trans-isomerization is made by enzymes, and the rate of formation of trans-forms under the action of pancreatin (olienzyme preparation with broad substrate specificity) was higher in comparison with lipase of microbial origin (Candida rugosa lipase). Under natural conditions of storage of raw materials (at room temperature), the rate of accumulation of trans-forms is much higher than at storage at a temperature of + 4°C, and at temperatures below zero the process practically does not develop [7].
A number of studies have shown that sodium nitrite catalyzes cis-trans-isomerization of fatty acids [8]. In order to verify this fact, scientists from Japan evaluated the content of TIFA in ready-made pork cutlets, prepared with the use of a mixture of KNO3 and NaNO2 in a ratio of 5 : 1. During the production of pork cutlets, a mixture of KNO3 and NaNO2 in the amount of 0; 100; 1000; 10000 mg/kg of meat was introduced in the salting process. The total content of trans-isomers of oleic and linoleic acids in the finished product was (mg/100 g of product) 0.538; 2.65; 3.38 and 27.6 respectively. As a result of the experiment, there was no evidence that the heat treatment under normal salting and cooking conditions in the presence of sodium nitrite leads to an increase in the proportion of TIFA in finished products. Meanwhile, it was suggested that oleic and linoleic acids in cis-trans meat are isomerized by a large amount of nitrite in the presence of nitric acid. Therefore, the addition of excess sodium nitrite during meat processing should be avoided [41].
Another way of forming TIFA is heat treatment at high temperatures, at which the double cis-bond is subjected to the isomerization process in the trans position. Henon (1999) believes that for the isomerization of alpha-linolenic acid in the trans form, heating to 220–230°C, and for linoleic – heating to 240°C is required. Four hours of heat treatment at 270°C led to isomerization with the formation of trans fatty acids of 80% alpha-linolenic acid and only 13% linoleic acid [42]. High temperature regimes are used in the production of certain types of meat products (fried, baked) and can lead to isomerization of fatty acids.
The processes of conservation and storing the finished product affect the change in fatty acid composition, including the level of TIFA. The fatty acid composition of the canned food "Stew pork meat" was studied, which was produced in two ways: under a strict sterilization regime with the achieved sterilizing effect of 18 conventional minutes and under a moderate sterilization regime with the achieved sterilizing effect of 12 conditional minutes [9]. It has been established that the strict regimes of thermal processing of meat raw materials and the duration of storage of the finished canned product lead to the formation of trans isomers in the fatty acids of the product: elaidinic (C18:1, 9-trans) and brassidine (C22:1 -trans).
The regimes of sterilization, strict and moderate, affect the content of brassidic acid in different ways: strict sterilization leads to an increase in the mass fraction of acid in 1.6 times, moderate sterilization reduces the content of this acid by 3.5 times. As a result of long-term storage (7.5 months), brassidic acid also behaves differently, depending on the sterilization regimes. In canned goods made under the strict sterilization regime, its proportion is reduced by 42.9%, and in canned goods, made under a moderate regime, it increases sharply (by 260% from the initial value). Its maximum proportion was 0.18% of the total number of fatty acids and did not exceed the norms established for oils for the content of transisomers in food products [9].
The effect of different types of processing and storage conditions on the content of elaidin fatty acid is different from that of brassidine. After 1.5 months of canned food storage, the proportion of elaidic acid in the lipids of a product manufactured under the strict sterilization regime increases dramatically, 5 times with respect to the mass fraction of this acid in the product produced under moderate regime. Its content in the moderate product after 1.5 months of storage is reduced and by the end of 7.5 months of storage elaidic acid is not identified [9].
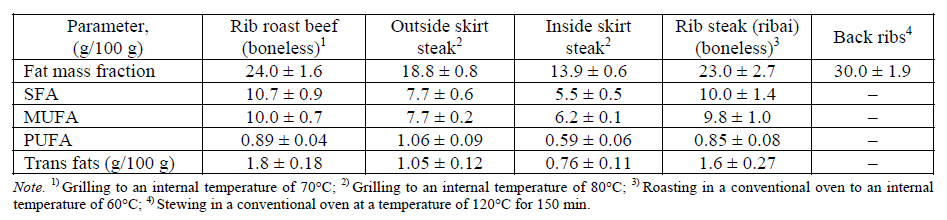
In the USA, in order to improve the consumer quality of meat raw materials, the methods of growing animals, the procedures for carcass cutting and cooking methods are constantly being improved. J. N. Martin et al. (2012) presented the results of a study of the nutritional value of beef cuts to update the reference materials of the US National Database (SR24) on nutrients, indicating the content of TIFA [43]. As a result of the evaluation of the data obtained, 29 beef cuts were considered to be lean because they contained less than 10 g of fat, 95 mg of cholesterol and 4.5 g of saturated fat (per 100 g of product) or had a fat content of no more than 10% (Table 2). As can be seen from Table 2, the content of TIFA depends on the way the meat product is prepared and on the total amount of fat on the cut.
As a result of the evaluation of clinical research data on the effects of TIFA on health, WHO experts stated that the consumption of such fats should be reduced to a minimum level (< 1% of the total energy consumed, i.e. no more than 2 g of trans fats a day with average consumption of 2000 kilocalories per day). Consumption of more than 5 g TIFA per day is a risk factor for health [19].
Denmark was the first European country to take measures that banned all products, including catering products, in which the content of TIFA exceeds 2% of total fat. In the United States, Canada, Finland, Norway, Austria, Hungary, Sweden and other countries, legislative restrictions on the content of TIFA in products have been introduced. The labels of packaged foods must indicate the amount of trans fats. At the same time, when determining the maximum permissible value of trans fat in food products, Denmark did not include natural TIFA (vaccenic, rumenic and other fatty acids) in this list on the labels [18]. Experts recommend consuming about 3 grams of conjugated linolenic acid per day from animal fats or foods enriched with this acid to maintain optimal health [44].
Foreign experience shows that the most effective measure to reduce the consumption of industrial TIFA by the population is the introduction of legislative restrictions on their content in food. In Russia, the content of TIFA is regulated only in dairy products. It is planned to reduce the content of trans-isomers of fatty acids in all food products to 2% by 2018. At the moment, the Russian consumer cannot evaluate their content in food products due to lack of this information on the labels. Unlike the EU, in Russia there is no harmonization of regulatory documentation regulating the content of trans fats in meat products. Meat products are not considered as a source of trans fats, however, under the influence of risk factors (intravital or technogenic origin), initiation of processes of transisomerization of lipids to potentially dangerous levels for humans is possible.
ВЫВОДЫ
The content of saturated fatty acids in meat raw materials is not a limiting factor for reducing meat consumption. Horse meat and beef are recommended for dietary and preventive nutrition for their biological effectiveness, and a balanced fatty acid composition of pork, horse and turkey lipids meets the requirements for functional foods. In meat raw materials TIFA are found, which become a matter of concern for an increasing number of consumers in the world and subject to close scrutiny by scientists. Meat raw materials of ruminants (cattle, sheep, goats, buffalo, deer, elks, giraffes and camels, etc.) have a high TIFA level. Transisomerization of fatty acids can occur as a result of natural synthesis in the rumen and can be regulated by diets. The use of growth promoters, high-temperature processing of meat raw materials, prolonged storage of a product and high sodium nitrite content during meat processing can contribute to TIFA increase in raw meat. Positive effects on animal health from the consumption of natural TIFA are shown, however, the mechanisms of the influence of TIFA on the functions of the organism are not fully explored. Consumption of foods with a high trans fats content shows adverse effects on human health, as a result of technological processing of raw materials. The necessity to normalize the content of industrial trans fats and to develop measures to reduce their level in food production has been discussed.
СПИСОК ЛИТЕРАТУРЫ
- Kulakova S.N., Viktorova E.V., and Levachev M.M. Trans-izomery zhirnykh kislot v pishchevykh produktakh [Trans-isomers of fatty acids in food]. Oils and Fats, 2008, no. 3, pp. 11-14. (In Russian).
- Ivankin A.N. Fats in the composition of modern meat products. Meat Industry, 2007, no. 6, pp. 8-15. (In Russian).
- Zaytseva L.V. and Nechaev A.P. Biochemical aspects of consumption of trans-isomer fatty acids. Nutrition, 2012, vol. 2, no. 4, pp. 17-23. (In Russian).
- Mozaffarian D. and Stampfer M.J. Removing industrial trans fat from foods. British Medical Journal, 2010, vol. 340, no. 7756, p. 1826. DOI: 10.1136/bmj.c1826.
- Mapiye C., Aldai N., Turner T.D., et al. The labile lipid fraction of meat: from perceived disease and waste to health and opportunity. Meat Science, 2012, vol. 92, no. 3, pp. 210-220. DOI: 10.1016/j.meatsci.2012.0316.
- Brito G., San Julian R., Luzardo S., et al. Effect of different nutritional strategies on carcass traits, meat quality and fatty acid composition of Uruguayan steers. 56th International Congress of Meat Science and Technology Proceedings, Korea, 2010, C0053.
- Ivankin A.N., Kulikovskiy A.V., Vostrikova N.L., and Chernukha I.M. Cis and trans conformational changes of bacterial fatty acids in comparison with analogs of animal and vegetable origin. Applied Biochemistry and Microbiology, 2014, vol. 50, no. 6, pp. 604-611(In Russian).
- Jiang H.L., Kruger N., Lahiri D.R., and Balazy M. Nitrogen dioxide induces cis-trans-isomerization of arachidonic acid within cellular phospholipids. Journal of Biological Chemistry, 1999, vol. 274, no. 23, pp. 16235-16241. DOI: 10.1074/jbc.274.23.16235.
- Lisitsyn A.B., Krylova V.B., and Gustova T.V. Degradatsiya lipidov v myasnykh konservakh v protsesse khraneniya [Degradation of lipids in canned meat during storage]. Materialy vserossiyskoy nauchno-prakticheskoy konferentsii «Nauchno-innovatsionnye aspekty pri sozdanii produktov zdorovogo pitaniya» [Proc. of the All- Russian scientific-practical conference “Research and innovation aspects in creating a healthy food”], Uglich, 2012, pp. 143-145.
- De Smet S., Van Paemel M., and Dierick N. Altering the content of essential nutrients in meats. 56th International Congress of Meat Science and Technology, Korea, 2010, pp. 25-31.
- Lisitsyn A.B., Chernukha I.M., and Ivankin A.N. Comparative study of fatty acid composition of meat material from various animal species. Scientific Journal of Animal Science, 2013, vol. 2, no. 5, pp. 124-131.
- Hanczakowski P., Szymczyk B., and Hanczakowska E. Fatty acid profile and cholesterol content of meat from pigs fed different. Annals of Animal Science, 2009, vol. 9, no. 2, pp. 157-165.
- Jungsuck C., Sangwook K., and Sooyong K. Pork quality is correlated with muscle fatty acid composition and plasma metabolites. 55th International Congress of Meat Science and Technology Proceedings, Denmark, 2009, PS1.06.
- Alfaia M., Ribeiro S., Quaresma G., et al. Biologically active fatty acids in bovine meats from intensive feeding systems and carnalentejana-PDO, a Portuguese meat with protected denomination of origin. 49th International Congress of Meat Science and Technology, Brazil, 2003, pp. 71-72.
- Belaunzaran X., Bessa R.J., Lavín P., et al. Horse-meat for human consumption - Current research and future opportunities. Meat Science, 2015, vol. 108, pp. 74-81. DOI: 10.1016/j.meatsci.2015.05.006.
- Lisitsyn A.B., Chernukha I.M., Kuznetsova T.G., Orlova O.N., and Mkrtichyan V.S. Khimicheskiy sostav myasa: Spravochnye tablitsy obshchego khimicheskogo, aminokislotnogo, zhirnokislotnogo, vitaminnogo, makro- imikroelementnogo sostavov i pishchevoy (energeticheskoy i biologicheskoy) tsennosti myasa [Chemical composition of meat: reference tables of general chemical, amino acid, fatty acid, vitamin, macro- and microelement compositions and food (energy and biological) meat values]. Moscow: The V.M. Gorbatov All-Russian Meat Research Institute Publ., 2011. 104 p.
- Martin C.A., Milinsk M.C., Visentainer J.V., Matsushita M., and De-Souza N.E. Trans fatty acid-forming processes in foods: A review. Anais da Academia Brasileira de Ciências, 2007, vol. 79, no. 2, pp. 343-350.
- Stender S., Astrup A., and Dyerberg J. Ruminant and industrially produced trans fatty acids: health aspects. Food and Nutrition Research, 2008, vol. 52, no. 1, pp. 1621. DOI: 10.3402/fnr.v52i0.1651.
- Kuhnt K., Degen C., and Jahreis G. Evaluation of the Impact of Ruminant trans fatty acids on human health: important aspects to consider. Critical Reviews in Food Science and Nutrition, 2016, vol. 56, no. 12, 1964-1980. DOI: 10.1080/10408398.2013.808605.
- Gayet-Boyer C., Tenenhaus-Aziza F., Prunet C, Marmonier C, Malpuech-Brugere C., Lamarche B., and Chardigny J-M. Is there a linear relationship between the dose of ruminant trans-fatty acids and cardiovascular risk markers in healthy subjects: results from a systematic review and meta-regression of randomised clinical trials? British Journal of Nutrition, 2014, pp. 1914-1922 DOI: 10.1017/S0007114514002578.
- De Souza R.J., Mente A., Maroleanu A., et al. Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: Systematic review and meta-analysis of observational studies. BMJ (Online), 2015, vol. 351, Article number h3978. DOI: 10.1136/bmj.h3978.
- Fernandez-Gines J.M., Fernández López J., Sayas-Barbera E., and Perez-Alvarez J.A. Meat products as functional foods: A review. Journal of Food Science, 2005, vol. 70, no. 2, pp. 37-43. DOI: 10.1111/j.1365-2621.2005.tb07110.
- Remacle C. and Reusens B. (ed.) Functional foods, ageing and degenerative disease. Woodhead Publishing Ltd., 2004. 771 p.
- Tsuda H., Ohshima Y., Nomoto H., et al. Cancer prevention by natural compounds. Drug Metabolism and Pharmacokinetics, 2004, vol. 19, no. 4, pp. 245-263. DOI: 10.2133/dmpk.19.245.
- Cesano A., Visonneau S., Scimeca J.A., Kritchevsky D., and Santoli D. Opposite effects of linoleic acid and conjugated linoleic acid on human prostatic cancer in SCID mice. Anticancer Research, 1998, vol. 18, no. 3A, pp. 1429-1434.
- Sarah K., Gebauer S. K., Chardigny J.-M., et al. Effects of Ruminant trans Fatty Acids on Cardiovascular Disease and Cancer: A Comprehensive Review of Epidemiological, Clinical, and Mechanistic Studies. Advаnces in Nutrition, 2011, vol. 2, pp. 332-354. DOI: 10.3945/an.111.000521.
- Kuhnt K., Kraft J., Moeckel P., and Jahreis G. Trans-11-18:1 is effectively Delta9-desaturated compared with trans- 12-18:1 in humans. British Journal of Nutrition, 2006, vol. 95, pp. 752-761.
- Uauy R., Aro A., Clarke R., et al. Who scientific update on trans fatty acids: Summary and conclusions. European Journal of Clinical Nutrition, 2009, vol. 63, pp. 68-75. DOI: 10.1038/ejcn.2009.15.
- Mozaffarian D, Katan M.B., Ascherio A., et al. Trans fatty acids and cardiovascular disease. New England Journal of Medicine, 2006, vol. 354, no. 15, pp. 1601-1613. DOI: 10.1056/NEJMra054035.
- Teegala S.M., Willett W.C., and Mozaffarian D. Consumption and health effects of trans fatty acids: a review. Journal of Association of Official Analytical Chemists International, 2009, vol. 92, no. 5, pp. 1250-1257.
- Mennitti L.V., Oliveira J.L., Morais C.A., et al. Type of fatty acids in maternal diets during pregnancy and/or lactation and metabolic consequences of the offspring. Journal Nutritional Biochemistry, 2015, vol. 26, no. 2, pp. 99-111. DOI: 10.1016/j.jnutbio.2014.10.001.
- Stender S., Astrup A., and Dyerberg J. A trans European Union difference in the decline in trans fatty acids in popular foods: a market basket investigation. British Medical Journal Open, 2012, vol. 2, no. 5, e000859. DOI: 10.1136/bmjopen-2012-000859.
- Šatalić Z., Barić I.C., Keser I., and Marić B. Evaluation of diet quality with the mediterranean dietary quality index in university students. International Journal of Food Sciences and Nutrition, 2004, vol. 55, no. 8, pp. 589-597. DOI: 10.1080/09637480500086141.
- Reremoana F., Theodore R.F., et al. Dietary patterns and intelligence in early and middle childhood. Intelligence, 2009, vol. 37, no. 5, pp. 506-513.
- Gatellier P., Bauchart D., Durand, and Renerre M. Effect of linseed oil supplementation on total fatty acids of muscle and on color stability and lipid oxidation of bovine meat. 50th International Congress of Meat Science and Technology Proceedings, Finland, 2004, pp. 1155-1158.
- Luzardo S., Montossi F., Monteverde M., et al. Effect of different hay - concentrate ratios on carcass traits meat quality and fatty acid composition on corriedale heavy lambs. 56th International Congress of Meat Science and Technology Proceedings, Korea, 2010, D096.
- Herdmann A., Martin J., Nuernberg G., Dannenberger D., and Nuernberg K. Dietary n-6 and n-3 fatty acids alter the fatty acid composition of tissues and the fate of beneficial fatty acids during processing. 56th International Congress of Meat Science and Technology Proceedings, Korea, 2010, C012.
- Matveyev V.A., Galochkina V.P., Agafonov V.I., et al. Sposob povysheniya intensivnosti rosta u bychkov [Method of increasing intensity of growth of bull calves]. Patent RF, no. 2379945, 2010.
- Damodaran Sh., Parkin K.L., and Fennema O.R. Khimiya pishchevykh produktov. St. Petersburg: Professija Publ., 2012. 1040 p. (Russ. ed.: Damodaran S., Pakin K.L., Fennema O.R. Fennemaʼs Food Chemistry, 4th ed. CRC Press Taylor&Fracis Group, Boca Raton, London, New York, 2008.).
- Kawahara S., Shibata K., Matsuoka Y., and Muguruma M. Cis-trans isomerization of unsaturated fatty acids in pork lipids by nitrite. 57-th International Congress of Meat Science and Technology Proceedings, Belgium, 2011, p. 424.
- Hénon G., Kemény Zs., Recseg K., Zwobada F., and Kovari K. Deodorization of vegetable oils. Part I: Modeling the geometrical isomerization of polyunsaturated fatty acids. Journal of the American Oil Chemists' Society, 1999, vol. 76, no. 1, pp. 73-81.
- Martin J. N., Brooks J. C., Thompson L. D., et al. Updating the united states national nutrient database with nutrient data for eight cooked beef cuts. 58th International Congress of Meat Science and Technology Proceedings, Canada, 2012, А12.
- Kodentsova V.M., Kochetkova A.A., Risnik D.V., Sarkisyan V.A., and Bessonov V.V. The effect of microwaves on the fat component and preserve vitamins in foods. Problems of Nutrition, 2015, vol. 84, no. 5, pp. 16-30.