Аннотация
Introduction. The problem of food adulteration is highly relevant today. Food manufacturers are increasingly replacing expensive raw materials with cheaper poultry. We aimed to develop an effective method for identification and quantification of chicken meat and egg products in multicomponent meat systems using real-time PCR.Study objects and methods. We studied native animal tissue, namely that of chicken, pork, beef, turkey, quail, duck, horse meat, rabbit, sheep, and goat. Standard samples were taken from pure fresh chicken muscle tissue. We also used raw, boiled, and powdered chicken eggs. For a semiquantitative analysis of chicken mass in the sample, we compared the threshold cycle (Сt) of chicken DNA and the threshold cycles of calibration samples. To ensure the absence of PCR inhibition, we used an internal control sample which went through all the stages of analysis, starting with DNA extraction.
Results and discussion. We developed a methodology to qualitatively determine the content of chicken tissue in the product and distinguish between the presence of egg products and contamination on the production line. The method for chicken DNA identification showed 100% specificity. This genetic material was detected in the range of 0.1% to 0.01% of chicken meat in the sample. The efficiency of the duplex PCR system for chicken DNA detection was more than 95% (3.38 on the Green slope channel and 3.45 on the Yellow slope channel). The analytical sensitivity of the primers was 40 copies/reaction.
Conclusion. Our methodology is suitable for analyzing multicomponent food products, raw materials, feed, and feed additives. It can identify the content of chicken meat at a concentration of up to 1%, as well as distinguish egg impurities from contamination of various origin. PCR allows differentiation between chicken meat and egg products.
Ключевые слова
Multicomponent products, canned food, chicken meat, egg melange, PCR, adulteration, sausagesВВЕДЕНИЕ
The Russian Federation strategy to improve the quality of food products until 2030 prioritizes research in the field of quality management.
Today, the problem of food adulteration is of particular concern. Food manufacturers are increasingly replacing expensive raw materials, such as good quality beef, with cheaper poultry. According to the public report “Consumer Protection in the Russian Federation in 2017”, Rospotrebnadzor (Russian Federal Service for Surveillance on Consumer Rights Protection and Human Wellbeing) detected 3410 adulterated products out of 310 000 inspected food samples [1]. In 2018, the volumes of rejected meat, poultry, and their products doubled compared to 2017. In particular, Rospotrebnadzor rejected 519 batches of meat and meat products weighing 3509 kg (compared to 459 batches of 1685 kg in 2017) and 168 batches of poultry, eggs, and their products weighing 1951 kg (compared to 159 batches of 975 kg in 2017).
Species identification of meat and meat products is becoming more important due to increased international trade and labeling rules introduced in many countries. Morphological and anatomical characteristics are used to identify fresh and unprocessed meat. However, processed meat loses its characteristic morphological features, which creates favorable conditions for adulteration, namely for replacing one type of meat with another, less valuable type. Poultry – a cheaper raw material compared to pork, beef or other meats – is often used to adulterate products, both semi-finished and finished. Especially difficult is species identification of multicomponent products containing several types of meat, egg impurities, various food additives, enzyme preparations, as well as products subjected to rigorous mechanical or thermal processing, such as canned foods and pastes [2–7]. According to Rospotrebnadzor, most violations of the technical standards in 2018 were detected in canned meat and sausages [1].
At the moment, the Russian Federation has no method for quantifying the content of chicken and/or egg melange in food products and isolating possible contamination on the production line.
Scientific literature reports numerous methods for qualitative identification of meat species [8–11]. A group of scientists from Gorbatov’s Federal Scientific Center for Food Systems and the National Center for Fishing Products Safety attempted to identify egg melange at the 30th PCR cycle [12, 13]. However, there were no data on the quantitative identification of impurities [14, 15]. Therefore, we need to develop a quantitative method for identifying ingredients in the analyzed products to prevent producers from replacing a specified content of meat with cheaper raw materials and to distinguish between adulteration and inevitable contamination in production [16–20].
The highly sensitive PCR method can reveal even trace amounts of meat ingredients, which are essentially technical impurities. However, in order to distinguish a minor technical impurity from intentional adulteration, we need a methodology for a quantitative or semiquantitative evaluation of meat, for example, chicken, in food products [20–38].
Therefore, we aimed to develop an effective method for identification and quantification of chicken meat and egg products in multicomponent meat systems using the real-time PCR.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
Our objects of study included native animal tissue purchased in retail chain stores (chicken, pork, beef, turkey, quail, duck, horse meat, rabbit, sheep, and goat) or obtained at the Russian State Center for Animal Feed and Drug Standardization and Quality, Moscow (mink, cat, and dog). Pure fresh chicken muscle tissue was used as standard samples. The species identity of all the materials was confirmed by the Sanger DNA sequencing method based on the standard CytB gene [3]. In addition, we used raw, boiled, and powdered chicken eggs.
We used only certified equipment, materials, reagents, and utensils.
The tests were conducted using the following methods:
– taking laboratory samples from different product groups (State Standard 31904-2012);
– adsorption DNA extraction based on silicon dioxide (State Standard R 56140-2014);
– guanidine-chloroform-based DNA extraction (State Standard R ISO 21571-2014). This method can purify DNA from fatty and protein impurities, reduce the inhibition of the reaction, and eliminate the influence of food additives on the final result (it also works well with egg impurities);
– real-time polymerase chain reaction with hybridization-fluorescence detection (State Standard ISO 22119-2013);
– evaluation of metrological characteristics of measurement procedures (RIS 61-2010);
– certification of measurement procedures (State Standard R 8.563-2009).
When sampling and preparing test samples, we took measures to prevent the seeding of environmental objects in line with State Standard 8756.0-70 and State Standard 31719-2012. The samples were homogenized and 0.05 g weighed, placed in a 1.5 cm Eppendorf type disposable microcentrifuge tube, labeled, and used to isolate DNA.
Three sets of samples were prepared in duplicate. The first set was not subjected to heat treatment. The samples of the second set were mixed with 100 mm3 of water and heated at 99°С on a Termite solid-state thermostat (DNA-Technology, Russia) for 30 min. The third set was sampled in quadruplicate and autoclaved at 110°C and 0.5 atm. for 30 min and an hour, respectively. For the purity of the experiment, we used chicken muscle tissue (breast fillet and drumstick), parenchymal and hollow internal organs (kidney, heart, liver), skin and cartilage, as well as minced pork meat containing 1% and 10% chicken.
Since chicken eggs are widely used in the food industry, we had to determine their effect on the PCR results. For this, we analyzed raw, boiled and powdered eggs, as well as pancake flour. In addition, we investigated 20% egg in minced pork, 10% raw egg in water, and 10% egg in minced chicken. A model panel was made from the above samples.
To eliminate the likelihood of PCR inhibition, we used an internal control sample (ICS) which was added to each test sample starting from the DNA extraction stage.
DNA was extracted by the sorbent method recommended by State Standard R 52723-2007, using a standard set of DNA-Sorb-S reagents (Central Research Institute of Epidemiology, Russia). A number of experiments performed with the extracted DNA showed that a 100% chicken content (whether fillet, hollow and parenchymal internal organs or connective tissue) produced a threshold cycle (Ct) ≤ 15, whereas 10% and 1% chicken contents in minced meat produced Ct ≤ 18 and Ct ≤ 21, respectively. There is a correlation with the ICS detection. When egg is present, the values decrease to Ct ≥ 23 and the ICS also drops to Ct ≥ 28 due to inhibition (Ct ≥ 24 with no inhibitors). DNA is obviously less degraded in a pure product (raw and boiled egg) than in egg powder, but Ct is inversely related: Ct ≥ 27 and Ct ≥ 20 for the egg powder sample and the ICS, respectively; Ct ≥ 30 and Ct ≥ 28 for the raw and boiled egg sample and the ICS, respectively.
Thus, we can conclude that raw and boiled eggs contain PCR-inhibiting substances. The presence of 10% raw eggs in minced chicken leads to ICS Ct ≥ 27 versus ICS Ct ≤ 21 for 100% minced chicken. It is impossible to evaluate the results when the reaction is so strongly inhibited. Therefore, we chose a different DNA extraction method described by Minaev et al. [2]. For this, we used a SORB-GMO-B kit (Syntol, Russia) in accordance with the manufacturer’s recommendations. The PCR results are shown in Table 1. As we can see, the ICS threshold cycle values indicate insignificant inhibition of the reaction, confirming the right choice of the DNA isolation method.
We selected those primers and probes that fluoresce to the target DNA of chicken and the ICS in the Green and Yellow channels. The solutions of direct and reverse PCR primers and a probe at a known molar concentration were diluted to a working molar concentration of 6 μmol/dm3 and 3 μmol/dm3, respectively. For PCR, we used a dNTF solution (Syntol, Russia), a PCR buffer-Flu and TaqF DNA polymerase (Central Research Institute of Epidemiology, Russia).
The DNA extracted from each test sample was analyzed in at least two replicates. For amplification control reactions, we used recombinant plasmids based on the pAL-2 vector (solutions of plasmid DNA at a concentration of 0.01 mg/dm3) as positive reaction controls. They were a plasmid containing a chicken DNA fragment (pCh) and a plasmid of the internal control sample (pICS).
For real-time PCR, we used Rotor-Gene Q amplifiers (QIAGEN, Germany) and Rotor-Gene 6000 amplifiers (Corbett Research Pty Ltd., Australia). We programmed the device according to the operating instructions and optimized the PCR-RT conditions for the duplex format. The primer annealing temperature was 60°С, with a PCR total temperature profile of 40 cycles.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
The PCR results for the model meat systems before and after heat treatment (at various temperatures) are presented in Table 1. The Background Threshold was set at 15% and the Threshold was 0.05. We interpreted the results based on the presence (or absence) of the intersection between the fluorescence curve and a threshold line set at an appropriate level. The conditions for analysis were as follows: for a positive PCR control, the threshold cycle values of Ct < 26 were present in the Green and Yellow channels; for a negative extraction control and a negative PCR control, the threshold cycle values were absent in all the channels; the threshold cycle value for the ICS was not lower than Ct ≤ 24 for qualitative determination, since higher values indicate PCR inhibition.
As we can see in Table 1, all the raw samples containing meat or offal (including extremely low concentrations) were identified at no later than the 19th cycle; egg impurities, no earlier than the 25th cycle; and egg powder and pancake flour, at the 29–30th cycle. Interestingly, pure chicken meat, whether fillet or offal, was identified at no later than the 14th cycle, while connective tissue, no later than the 17th cycle. The chicken contents of 10% and 1% produced Ct ≤ 15 and Ct ≤ 19, respectively. These results allowed us to conclude that:
– Ct < 15 indicated over 10% chicken in the test sample;
– Ct < 19 indicated over 1% chicken or high concentrations of connective tissue in the test sample. This conclusion makes it impossible to quantify the chicken content at this stage of the study. However, it leaves a possibility of a semi-quantitative analysis, whose result can be expressed as “chicken content at least N%”.
The heat-treated samples containing meat or offal (including extremely low concentrations, up to 1%) were identified at no later than the 21st cycle and egg impurities, no earlier than the 21st cycle. A 10% chicken content in minced meat produced Ct ≤ 17, whereas 1% chicken showed Ct ≤ 21. From these results, we concluded that Ct < 21 indicated more than 1% chicken in the test sample.
The autoclaved samples containing chicken meat or offal were identified at no later than the 17th cycle, whereas the samples with extremely low concentrations of chicken meat (up to 1%) and egg impurities, no later than the 26th cycle. The chicken contents of 10% and 1% resulted in Ct ≤ 21 and Ct ≤ 25, respectively. Thus, the detection of Ct < 25 indicated over 1% chicken in the test sample.
Next, we proceeded to the development of a semiquantitative method for determining chicken meat in food products, since a quantitative method was not possible due to the equality of cycles for the 10% minced chicken samples and the connective tissue samples. As adulterating a product with less than 1% meat (1 g chicken meat per 1 kg of product) seems impractical, we decided that the methodology should allow us to determine the content of chicken in the product in relation to several threshold values of calibration samples, namely:
– “at least 1%” if Ct 10% < sample’s Ct ≤ Ct 1%;
– “at least 10%” if Ct 50% < sample’s Ct ≤ Ct 10%;
– “high content” if the sample’s Ct ≤ Ct 50%;
– “low DNA, possible egg presence” if the sample’s Ct > Ct 1%.
Further, we evaluated the following criteria: sensitivity and specificity of the primers, detection limits, and a range of values for calibration samples and internal control samples. Each experiment was performed by two different researchers, at different times, with reagents of different series, on different amplifiers of the same type. Each sample was tested in duplicate.
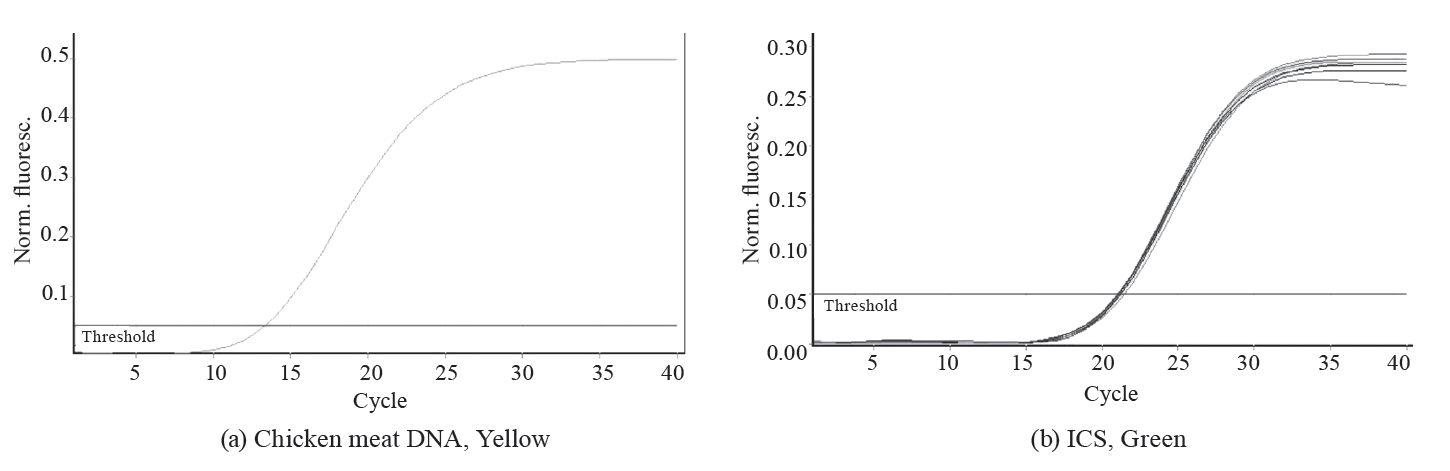
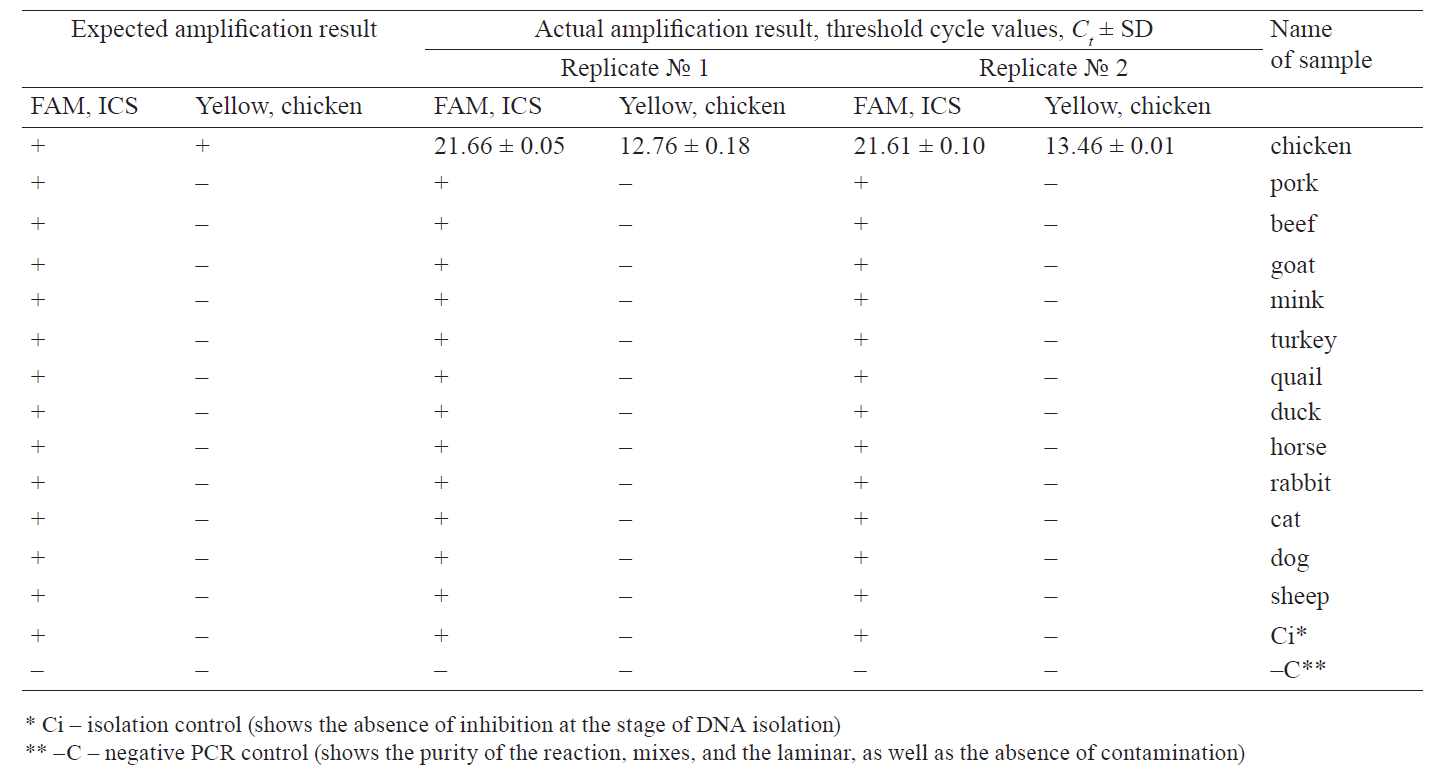
To assess the specificity of PCR, we created a panel of DNA samples isolated from chicken, pork, beef, turkey, quail, duck, horse, mink, rabbit, cat, dog, goat, and sheep. The results are shown in Fig. 1 and Table 2.
Within the proposed panel, the chicken DNA identification methodology showed 100% specificity: we observed the ICS amplification only on the Green channel and the target chicken DNA on the Yellow channel.
The assessment of the control panel for validation confirmed a 100% convergence of the results.
To determine the analytical sensitivity of the primers, we isolated DNA from a sample of 100% chicken meat and prepared a series of 10-fold dilutions. The maximum dilution was determined which allowed reproducible (in duplicate) detection of DNA.
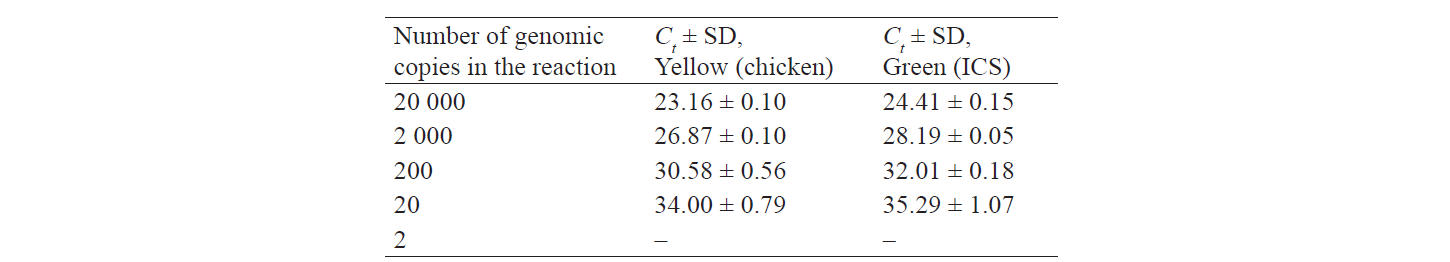
In addition, we used plasmid DNA solutions at a specified concentration containing a cloned chicken gene fragment and a ICS fragment. Two series of ten-fold dilutions were prepared in a TE buffer with various concentrations: series № 1 – pICS plasmid DNA solution; series № 2 – pCh plasmid DNA solution. The initial concentration of plasmid DNA in each series was 4 ng/μL, which corresponds to ~ 20 000 genomic copies in PCR (5 μL of a DNA solution for a 25 μL reaction). The results are presented in Table 3.
To determine the absolute limit of detection (LOD) at which the PCR method is able to detect and quantify chicken genetic material, we performed 10 PCRs, with 5, 10, 20, and 40 genomic copies of chicken DNA in each. Our PCR methodology detected chicken even in the strongest dilution, with only five genomic copies in the PCR.
To determine the limit of detection of chicken and egg products in multicomponent raw and heat-treated products, we used a number of model samples prepared in two replicates and containing 10, 1.0, 0.1, 0.01, and 0.001% chicken in minced pork (isolated DNA). The samples were preliminarily cooked at 99°С for 30 min. To determine the LOD of chicken and egg products in canned foods, the model samples were autoclaved at 110°C and 0.5 atm. The minimum chicken content in minced pork was determined, at which chicken DNA was reproducibly (in duplicate) detected. The results are shown in Table 4.
The limit of detection for chicken DNA ranged from 0.1 to 0.001% of the chicken content in the sample.
The methodology should allow us to assess the content of chicken and egg products in food products relative to several selected threshold values of calibration samples. To prepare calibration samples of various compositions for the semi-quantitative analysis of raw and cooked products, we mixed 100% minced chicken meat with 100% minced pork (1%, 10%, and 50% chicken) and heated at 99°C for 30 min.
We decided to evaluate both cooked and raw products in relation to the values of heat-treated calibrators, since fresh chicken meat was used to prepare model samples of raw products, which cannot be guaranteed by product manufacturers. Moreover, samples for analysis do not always get delivered to the laboratory directly, bypassing the stages of storage or freezing, which increases the likelihood of DNA degradation. The calibration samples for canned products were autoclaved at 110°C and 0.5 atm. The uniformity coefficient of the calibrators was 0.99 (99%).
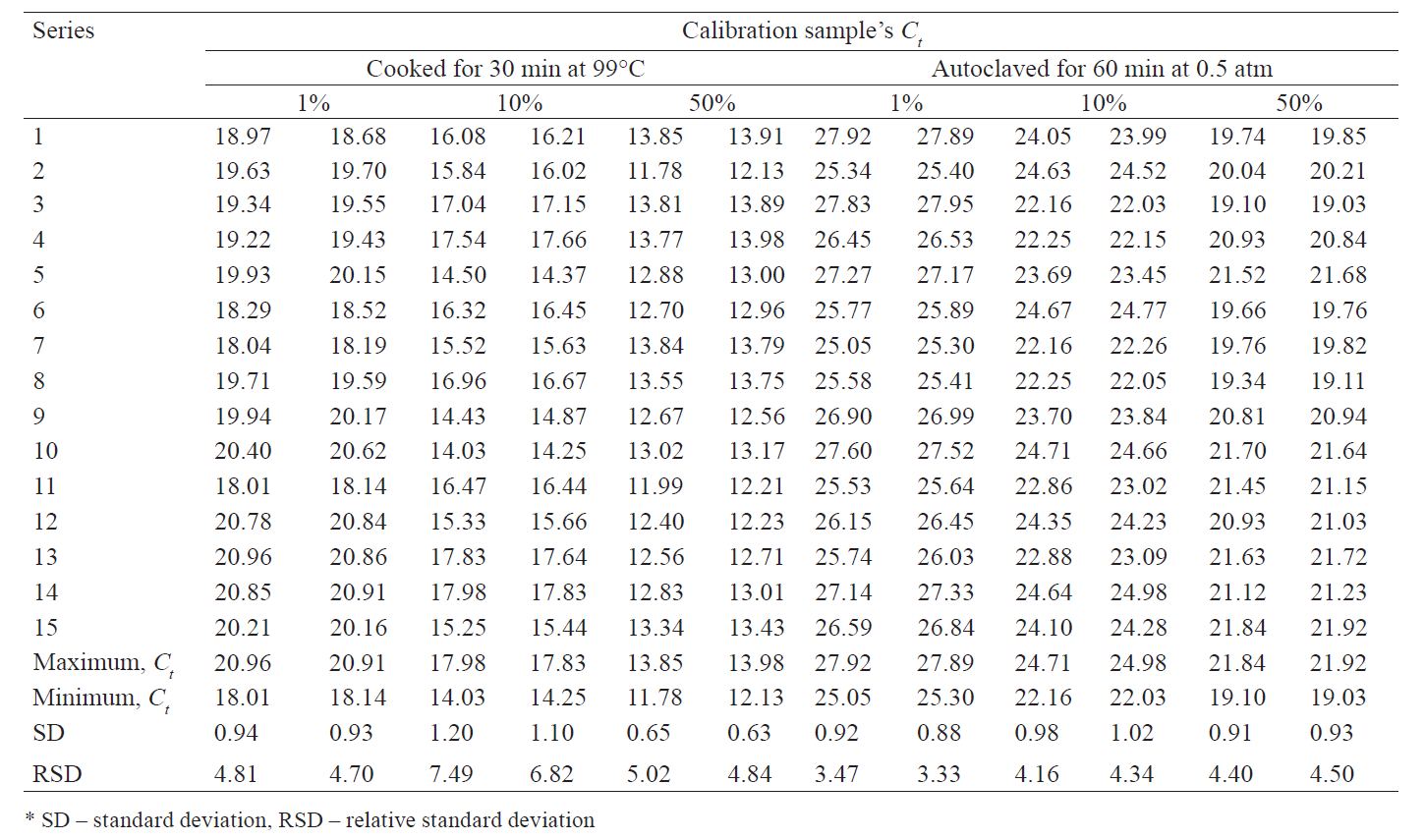
To confirm the constancy of the calibrators’ Ct ranges, we performed a series of tests. In particular, we studied 15 series of calibration samples prepared on different days, by different people, each in two replicates. For each series, we determined the minimum and maximum values of the threshold cycle on the Yellow-chicken channel, a standard deviation, and a relative standard deviation. The results are presented in Table 5.
As a result, we selected the following threshold cycle values on the “Yellow-Chicken DNA” channel for the calibrators of:
– raw products and those subjected to light heat
treatment: 18 ≤ Ct 1% < 21; 14 ≤ Ct 10% < 18; Ct 50% < 14;
– autoclaved products (canned food): 25 ≤ Ct 1% < 28; 22 ≤ Ct 10% < 25; Ct 50% < 22.
Also, a threshold cycle value of at least Ct ≤ 24 was chosen as acceptable on the “Green-ICS” channel for the calibrators (Ctics 1%, Ctics 10%, Ctics 50%) and the negative control sample.
ВЫВОДЫ
We developed a method (certified methodology) for a semi-quantitative assessment of chicken content in multicomponent food systems of varying degrees of heat and mechanical treatment: raw, heat-treated, canned, finely ground, and homogenized. Having tested various DNA extraction methods, we concluded that the guanidine-chloroform method reduces the content of PCR-inhibiting substances compared to the sorption method.
Our methodology was tested on model samples, as well as product samples from retail stores, to exclude the possibility of PCR inhibition by food additives, stabilizers, emulsifiers, etc. With PCR, we can distinguish between chicken meat and egg products in raw and cooked products (over 21 cycles), as well as canned foods (over 28 cycles). Our results suggest that this methodology is suitable for analyzing multicomponent food products, raw materials, feeds, and feed additives. In addition, it can identify the content of chicken meat at a concentration of up to 1%, as well as detect egg impurities and contamination of various origins.
Taking into account the current need for distinguishing adulteration from the inevitable contamination on the production line, as well as preventing adulteration of expensive raw materials with chicken meat by introducing egg products, we believe that our methodology could make a significant contribution to the production of high-quality foods.
КОНФЛИКТ ИНТЕРЕСОВ
The authors state that there is no conflict of interest.
СПИСОК ЛИТЕРАТУРЫ
- Zashchita prav potrebiteley v Rossiyskoy Federatsii v 2017 godu: Gosudarstvennyy doklad [Consumer Rights Protection in the Russian Federation in 2017: Public Report]. Moscow: Federal Service for Surveillance on Consumer Rights Protection and Human Wellbeing; 2017. 318 p.
- Lisitsyn AB, Chernukha IM, Lunina OI. Functional food products: innovative methods of counterfeit detection. Production Quality Control. 2018;(4):13–18. (In Russ.).
- Sattarova BN, Asqarov IR, Dzhurayev AM. Some questions classification of chicken meat. Universum: khimiya i biologiya [Universum: chemistry and biology]. 2018;53(11):36-38. (In Russ.).
- Galkin AV, Trepalina E. Opredelenie soderzhaniya myasa v farshe i kolbasnoy produktsii [Determination of meat content in minced meat and sausage products]. Myasnoy ryad [Meat row]. 2019;74(4):63. (In Russ.).
- Montowska M. Proteins and peptides in the authentication of meat and meat products. In: Sforza S, editor. Food Authentication using bioorganic molecules. Lancaster: DEStech Publications; 2013. pp. 75–102.
- Amaral JS, Santos G, Oliveira MBPP, Mafra I. Quantitative detection of pork meat by EvaGreen real-time PCR to assess the authenticity of processed meat products. Food Control. 2017;72:53-61. DOI: https://doi.org/10.1016/j.foodcont.2016.07.029.
- Shehata HR, Li JP, Chen S, Redda H, Cheng SM, Tabujara N, et al. Droplet digital polymerase chain reaction (ddPCR) assays integrated with an internal control for quantification of bovine, porcine, chicken and turkey species in food and feed. PLoS ONE. 2017;12(8). DOI: https://doi.org/10.1371/journal.pone.0182872.
- Lluehlin MB. Opredelenie nukleotidnoy posledovatelʹnosti DNK. Molekulyarnaya klinicheskaya diagnostika. Metody [Determination of the nucleotide sequence of DNA. Molecular clinical diagnosis. Methods]. Moscow: Mir; 1999. pp. 428–447. (In Russ.).
- Makkredi BDzh, Chimera DA. Obnaruzhenie i identifikatsiya patogennykh mikroorganizmov molekulyarnymi metodami. Molekulyarnaya klinicheskaya diagnostika. Metody [Detection and identification of pathogenic microorganisms by molecular methods. Molecular clinical diagnosis. Methods]. Moscow: Mir; 1999. pp. 496–506. (In Russ.).
- Bobo LD. PCR detection of chlamydia trachomatis. Diagnostic molecular microbiology. Principles and applications. Washington: ASM Press; 1993. pp. 235–241.
- Cockerill FR. Genetic methods for assessing antimicrobial resistance. Antimicrobial Agents and Chemotherapy. 1999;43(2):199–212. DOI: https://doi.org/10.1128/AAC.43.2.199.
- Crotchfelt KA, Welsh LE, Debonville D, Rosenstraus M, Quinn TC. Detection of Neisseria gonorrhoeae and Chlamydia trachomatis in genitourinary specimens from men and women by a coamplification PCR assay. Journal of Clinical Microbiology. 1997;35(6):1536–1540.
- Minaev MYu, Solodovnikova GI, Kurbakov KA, Fomina TA, Zaitseva EV. PCR analysis upon chicken DNA detection in meat products with egg m lange. Meat Industry. 2017;8:49–52. (In Russ.).
- Zabezhinskaya OM, Aslamazov EhG, Glukhov AI, Alyaev YuG, Zezerov EG, Belushkina NN, et al. Vyyavlenie mikrometastazov raka predstatelʹnoy zhelezy s pomoshchʹyu sovmeshchyonnykh reaktsiy obratnoy transkriptsii i polimeraznoy tsepnoy reaktsii [Identification of prostate cancer micrometastases using combined reverse transcription and polymerase chain reactions]. Problems of Biological, Medical and Pharmaceutical Chemistry. 2002;(3):28–32. (In Russ.).
- M’Zali F-H, Gascoyne-Binzi DM, Heritage J, Hawkey PM. Detection of mutations conferring extended-spectrum activity on SHV β-lactamases using polymerase chain reaction single strand conformational polymorphism (PCRSSCP). Journal of Antimicrobial Chemotherapy. 2006;37(4):797–802. DOI: https://doi.org/10.1093/jac/37.4.797.
- Fredericks DN, Relman DA. Application of Polymerase chain reaction to the diagnosis of infectious diseases. Clinical Infectious Diseases. 1999;29(3):457–488.
- Rubel MS, Dubrovina IA, Miasnikov VA, Lipaeva PV, Sinegubova EO. Comparative characteristics of modern express methods of diagnosing infectious diseases base on isothermal PCR technology. Vestnik Rossiiskoi Voennomedicinskoi Academii. 2018;S1:160–163. (In Russ.).
- Kolotova AA, Vasileva OYu, Goroshko PV. Polymerase chain reaction as the research technique in molecular and genetic diagnostics. Scientific Review. Pedagogical Sciences. 2019;5–2:46–51. (In Russ.).
- Zernov NV, Guskova AA, Skoblov MYu. New approach for diagnostic of Facioscapulohumeral muscular dystrophy based on PCR. Medical Genetics. 2019;18(7)(205):3–9. (In Russ.). DOI: https://doi.org/10.25557/2073-7998.2019.07.3-9.
- Limanskii A. Visualization of amplicons after polymerase chain reaction. Biophysics. 2005;50(6):1019–1024. (In Russ.).
- Chernukha IM. Application of “-omics” technologies for the analysis of raw meat and products. All about meat. 2012;(6):34–38. (In Russ.).
- Belousova EV, Yurchak ZA, Chernukha IM, Kryuchenko EV. New standards on testing methods of meat products. All about meat. 2017;2:58–60. (In Russ.).
- Vostrikova NL, Chernukha IM. Izuchenie proteoma myshechnoy tkani zhivotnykh s ispolʹzovaniem bioinformatiki [The study of the animal muscle tissue proteome using bioinformatics]. Aktualʹnaya biotekhnologiya [Actual Biotechnology]. 2018;26(3):322–327. (In Russ.).
- Vostrikova NL, Kulikovskii AV, Chernukha IM, Kovalev LI, Savchuk SA. Determination of muscular tissue proteins by 2D electrophoresis and time-of-flight mass spectrometry. Journal of Analytical Chemistry. 2017;72(10):932–943. (In Russ.). DOI: https://doi.org/10.7868/S0044450217100103.
- Tyulkin SV, Vafin RR, Muratova AV, Khatypov II, Zagidullin LR, Rachkova EN, et al. Development of a method for PCR-RFLP on the example of dgat1 gene in cattle. Fundamental research. 2015;(2–17):3773–3775. (In Russ.).
- Kushnareva TV, Maksema IG. Aspects of the application of rt-PCR with hantaan-, amur- and seoul-hantaviral infections. Modern problems of science and education. 2015;(3):560. (In Russ.).
- Mazitov MR, Valiullina IR, Kulagina LYu. Using PCR in real time for the detection of resistance genes problematic gram-negative bacteria. Practical Medicine. 2015;89(4–2):76–78. (In Russ.).
- Zavyalova EA, Kandrina NYu, Lomakina NF, Gulyukin MI. Indication and indification of dangerous and guarantine fish diseases based on reverse transcription (RT) -polymerase chain reaction (PCR). Rybovodstvo i rybnoe khozyaystvo [Fish-farming and fisheries]. 2015;(3):21–25. (In Russ.).
- Tamakhina AYa, Kozhokov MK. Biosecurity and methods of falsification of meat products. Izvestia of Kabardino-Balkarian State Agrarian University named after V.M. Kokov. 2017;16(2):53–58. (In Russ.).
- Osipova NI. Opredelenie vidovoy prinadlezhnosti myasa metodom izoehlektrofokusirovaniyam [Determination of species of meat by isoelectric focusing method]. Veterinariya. Referativnyy zhurnal [Veterinary Medicine. Abstract journal]. 2011;(2):299. (In Russ.).
- Kizina IE. Information content of polymerase chain reaction for diagnostics, evaluation of course and results of onychomycosis treatment. Biomedical and Biosocial Anthropology. 2015;(25):169–172. (In Ukr.).
- Tan LL, Ahmed SA, Ng SK, Citartan M, Raabe CA, Rozhdestvensky TS, et al. Rapid detection of porcine DNA in processed food samples using a streamlined DNA extraction method combined with the SYBR Green real-time PCR assay. Food Chemistry. 2020;309. DOI: https://doi.org/10.1016/j.foodchem.2019.125654.
- Balakrishna K, Sreerohini S, Parida M. Ready-to-use single tube quadruplex PCR for differential identification of mutton, chicken, pork and beef in processed meat samples. Food Additives and Contaminants – Part A Chemistry, Analysis, Control, Exposure and Risk Assessment. 2019;36(10):1435–1444. DOI: https://doi.org/10.1080/19440049.2019.1633477.
- Shehata HR, Naaum AM, Chen S, Murphy T, Li J, Shannon K, et al. Re-visiting the occurrence of undeclared species in sausage products sold in Canada. Food Research International. 2019;122:593–598. DOI: https://doi.org/10.1016/j.foodres.2019.01.030.
- Skouridou V, Tomaso H, Rau J, Bashammakh AS, El-Shahawi MS, Alyoubi AO, et al. Duplex PCR-ELONA for the detection of pork adulteration in meat products. Food Chemistry. 2019;287:354–362. DOI: https://doi.org/10.1016/j.foodchem.2019.02.095.
- Mansouri M, Khalilzadeh B, Barzegari A, Shoeibi S, Isildak S, Bargahi N, et al. Design a highly specific sequence for electrochemical evaluation of meat adulteration in cooked sausages. Biosensors & bioelectronics. 2020;150. DOI: https://doi.org/10.1016/j.bios.2019.111916.
- Danilov AM, Bazhenova BA, Danilov MB, Gerasimov AV. Study of lysate activity to modificate collagene raw materials to use in sausage mixture. Foods and Raw Materials. 2018;6(2):256–263. DOI: https://doi.org/10.21603/2308-4057-2018-2-256-263.
- Safaei F, Abhari K, Khosroshahi NK, Hosseini H, Jafari M. Optimisation of functional sausage formulation with konjac and inulin: using D-Optimal mixture design. Foods and Raw Materials. 2019;7(1):177–184. DOI: https://doi.org/10.21603/2308-4057-2019-1-177-184.