Аннотация
The increasing shortage of fossil hydrocarbon fuel dictates the need to search for and develop alternative energy sources, including plant biomass. This paper is devoted to the study of the Miscanthus plants biomass potential and the analysis of technologies of its processing into products targeted at bioenergy, chemistry, and microbiology. Miscanthus is a promising renewable raw material to replace wood raw materials for the production of chemical, fuel, energy, and microbiological industries. Miscanthus is characterised by highly productive (up to 40 tons per one hectare of dry matter) C4-photosynthesis. Dry Miscanthus contains 47.1–49.7% carbon, 5.38–5.92% hydrogen, and 41.4–44.6% oxygen. The mineral composition includes K, Cl, N and S, which influence the processes occurring during biomass combustion. The total amount of extractives per dry substance lies in the range of 0.3–2.2 % for different extraction reagents. Miscanthus has optimal properties as an energy source. Miscanthus × giganteus pellets showed the energy value of about 29 kJ/g. For the bioconversion of plants into bioethanol, it is advisable to carry out simultaneous saccharification and fermentation, thus reducing the duration of process steps and energy costs. Miscanthus cellulose is of high quality and can be used for the synthesis of new products. Further research will focus on the selection of rational parameters for processing miscanthus biomass into products with improved physical and chemical characteristics: bioethanol, pellets, industrial cellulose, bacterial cellulose, carbohydrate substrate.Ключевые слова
Miscanthus, bioethanol, cellulose, raw materials, processingВВЕДЕНИЕ
Currently, the task of finding alternative energy sources that are environmentally safe and economically affordable is very urgent. Of particular interest are species of herbaceous plants with a high growth rate, characterised by high values of the above-ground vegetative mass growth and having practical application as an energy source [1].
An example is the genus Miscanthus plants, a practically inexhaustible source of renewable raw materials in the field of alternative energy. This is due not only to the chemical properties of their biomass, but also to high growth rates and enormous biological productivity, among other things in a temperate climate, which together make their use in Russia promising [2].
The main advantages of miscanthus biomass in comparison with other types of perennial grasses are associated with its higher productivity, resistance to adverse environmental conditions, increased lignin content and, consequently, increased calorific capacity.
In addition, the genus Miscanthus plants can be used to produce biologically active substances. Miscanthus extracts include fatty acids, sterols and other aromatic compounds. The main structures of phenolic compounds and sterols of the bark and core of Miscanthus × giganteus include vanilla acid, para-coumaric acid, vanillin, para-hydroxybenzaldehyde, syringaldehyde, campesterol, stigmasterol, β - sitosterol, stigmast-3,5-diene-7-one, stigmast-4-ene-3-one, stigmast-6-ene-3,5-diol, 7-hydroxy-β-sitosterol and 7-oxo-β-citerol [3].
Currently in the world there is an increase in cultivation of Miscanthus driven by the characteristic high growth rates and a high degree of its biological needs compliance with agro-climatic conditions.
The purpose of this review was to analyse the modern methods of processing Miscanthus plants for bio-ethanol production, technical and bacterial cellulose, as well as products for microbiological and biotechnological industry.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
The representatives of the genus Miscanthus (Miscanthus Anderss L.) graminoid family (Poaceae) were the materials of this research. We analysed botanical characteristics and geographical distribution of various studied plants, made chemical composition analysis, and summarised the main processing methods according to the sources of scientific literature. These resulted in the analysis of modern methods of obtaining products for fuel, energy, chemical and microbiological industries.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
The botanical characteristics and distribution of the genus Miscanthus plants. The Miscanthus family includes about 40 species of monocotyledonous herbaceous perennial, sustainable plants with long curved linear leaves and small buds that bloom in late summer or early autumn, and about 20 species of miscanthus proper (Fig. 1).
In the Russian Far East (Primorsky Krai, Sakhalin and the Southern Kuril Islands), there are three species of miscanthus: Miscanthus sinensis, Miscanthus sacchariflorus and Miscanthus purpurascens. It can grow in the climatic conditions of Central and Eastern Europe [4]. In the late 20th century there appeared new miscanthus genotypes adapted to growth in the Northern regions, including Russian territories.
For the countries of the European Union (EU) it is recommended to grow miscanthus in the continental climate zone and the North Mediterranean, where soil and climatic resources correspond to the requirements of the plants [5, 6].
In Europe the plants reach a height of 3–4 m, and the representatives of tropical and subtropical species may reach 5 m or more in warm and humid climatic conditions. In the Central part of Russia, according to research of scientists of the Russian Timiryazev State Agrarian University, miscanthus giant reaches the height of 2 m, in Western Siberia – 2.5 m, and 3.9 m in the middle Volga region. The stems are upright and resistant to lodging because of their considerable thickness. It reaches 6 cm in homeland regions (China, Japan, Russian Far East, USA East coast); in the Middle Volga regions plants of 1–4 years of life are 0.8–1.5 cm thick [7, 8].
Miscanthus does not impose high requirements to soil and can grow well on marginal and low density soils whose granulometric composition is dominated by sand fractions. In Ukraine it is cultivated on sod-podzolic type of soils; in the forest-steppe of Novosibirsk Priobye, Middle Volga and Moscow region – on gray forest soils [9, 10].
For optimal growth and development plants require certain thermal and water regimes. Miscanthus seed germination requires ≥ 20°C soil heating with soil moisture of 60–80% of full field moisture capacity. To resume the shoots and the active growing season on crops of previous years, the temperature of the air must be in the range of 20–25°C. As shown in the literature, physiological activity of the studied representatives of the genus is sharply reduced at temperatures below 6°С.
Optimal temperature for adequate photosynthesis is considered to be 28–32°С. In Eastern Europe this is enough to produce sufficiently high yields of biomass [11].
It is known that miscanthus belongs to the C4 plants, characterised by: optimum temperature for photosynthesis of 30–45°C, 40–80 mg/dm2h CO2 assimilation in full sunlight, more economical water consumption as compared to C3 plants (twice and more), high drought and heat resistance, salt tolerance. These lead to better assimilation activity and, consequently, biological productivity.
The vast majority of well-known scientific studies are devoted to the three species of the genus miscanthus: Miscanthus giganteus, Miscanthus sinensis and Miscanthus sacchariflorus, which are the most widespread in Russia and abroad [12].
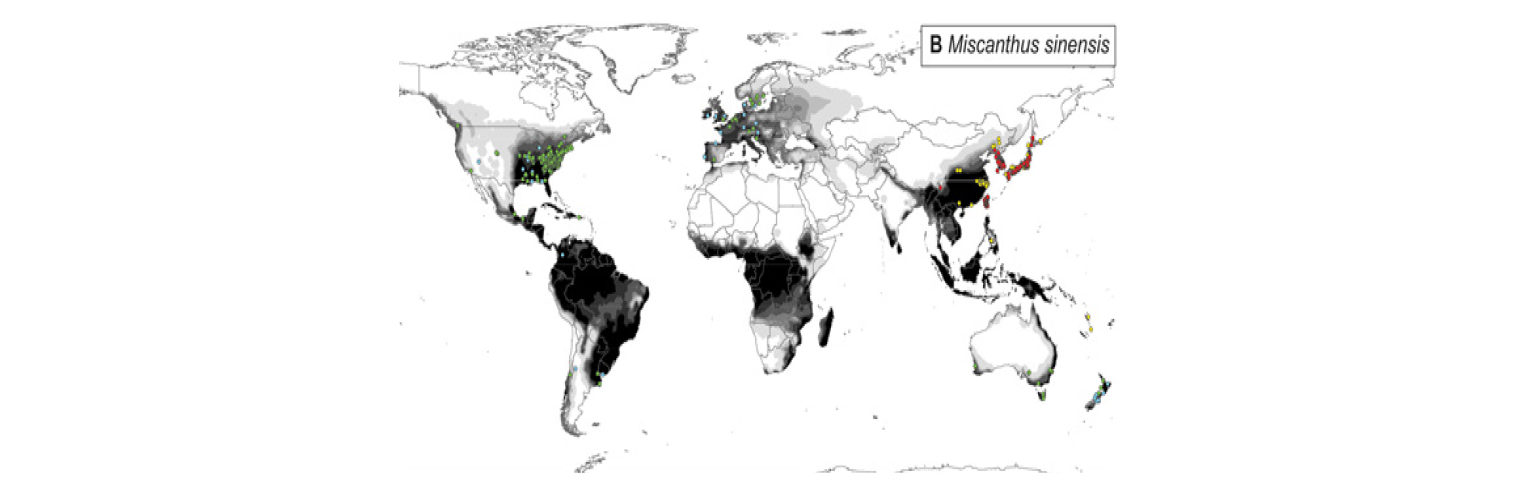
Chinese miscanthus (M. sinensis) is one of the most common types of ornamental grasses, named differently in different countries. For example, ‘Chinese silver grass’ or ‘magic grass’, sometimes ‘Chinese reeds’. In nature it is widespread in the Russian Far East up to the taiga zone, also in China, Korea, Japan. As an adventive species it occurs in many countries, e.g. USA, Brazil and African States (Fig. 2) [13].
M. sacchariflorus is a species growing on wet meadows, forest clearings and stony slopes in the Primorsky Krai, China, Korea, Japan, and also in the above-mentioned areas [14].
This also is a perennial long-stem herb up to 2 m in height. Stems are erect, thick and numerous. Leaves are rather rigid, linear, flat, long-acuminate, 7–18 mm wide, up to 60 cm long. Flowers are small, in spikelets with long, white, silky hairs. Inflorescence is 15–25 cm long, pinkish-silvery, white, fan-shaped, consisting of 8–20 spiciform branchlets with fruits. It blooms in late August–September. Flowering often occurs in July [15]. Not demanding to soil fertility.
Giant miscanthus (Miscanthus × giganteus), as described above, has a C4 photosynthetic path and provides high productivity of plant biomass. The genome of this species includes a triple set of chromosomes that do not divide during meiosis because gametes are not viable. As a rule, seeds are formed sterile, which significantly limit reproduction of the species, this is a barrier to the establishment of new fields.
The rhizome structure of giant miscanthus grows very slowly and decreases proliferation [16, 17]. Therefore, planting material is produced by cultivation of mother plantations (Queen cells), pre-multiplying it in vitro, or by rhizomes by targeted separation from plants of the previous planting year. This technology is limited by insufficient amount of seedling material and the lack of landing equipment [18, 19]. In this regard, high-quality uniform planting material can be grown in special nurseries with the use of modern biotechnologies.
Chemical composition and properties of genus Miscanthus plants. Analysis of miscanthus chemical composition biomass allows planning proper use of species. The three main components of lignocellulosic materials are: cellulose, hemicellulose and lignin. Their content in each organ is not equivalent and depends on the plant’s functional and physiological properties. The amount of cellulose in the stem, as a rule, is higher than that in the leaves. Lignin contains three-dimensional phenylpropyl-based polymer that provides structural rigidity and integrity, as well as prevents lingocellulose swelling [20–22].
Table 1 shows the differences in concentration of these substances in the cell wall in the three species growing on the territory of European countries, after 3–5 years of cultivation [14].
According to the data presented in the table, the types of miscanthus giganteus and sacchariflorus differ insignificantly in pulp content and hemicellulose/lignin ratio.
Table 2 shows the averages of biochemical composition of leaves, stems and a whole 4-year-old plant of Miscanthus varieties [4].
Data analysis indicates that the stem of miscanthus is the most suitable raw material for obtaining a large amount of high quality cellulose, as it has lower content of ash and lignin and a higher yield of the target product.
In the process of turning cellulose into ethanol such indicators as degree of polymerisation (n) and its crystallinity are of supreme value. The number of glucose units that make up one polymer molecule is called the degree of polymerisation.
X-ray diffraction and solid state 13 CP/MAS NMR spectroscopy are the two most commonly used methods of cellulose crystallinity determination. The crystallinity of cellulose for the species Miscanthus sinensis was measured by X-ray diffraction. Subject to the dimensions of the particles, differences in the index of cellulose crystallinity were revealed (Table 3) [23].
It is generally believed that cellulose crystalline regions are harder to decompose than amorphous domains, due to the strong intermolecular hydrogen bonds. For the studied species, the researchers found that the initial rate of cellulose hydrolysis increased with decreasing crystallinity [24, 25].
Unlike cellulose, hemicelluloses have lower degree of polymerisation, typically 50–300; they also have a branched structure and are amorphous. The predominant hemicellulose polymer for the miscanthus is the arabinoxylane, which contains a chain of 1.4-linked xylonic links. Sugar components in hemicellulose can participate in the formation of lignin-carbohydrate complexes (LCC) by covalent linkages between lignin and carbohydrates.
Despite considerable analytical studies aimed at the characterisation of the LCC, they still remain poorly defined, and their biosynthesis pathways require further study [26]. Distribution, structure and content of lignin is considered to be one of the important factors responsible for the recovery of lignocellulosic enzymatic degradation.
Considering miscanthus as fuel, the values of specific heat of combustion, mineral composition, ash content and content of volatile substances were analysed. The specific heat of combustion parameter is closely linked to the elemental composition and ash content. For Miscanthus × giganteus it ranges from 17 to 20 MJ/kg. Dry raw material contains on average 47.1–49.7% carbon, 5.38–5.92% hydrogen, and 41.4–44.6% oxygen. Mineral composition includes the following elements: K, Cl, N and S, which have an impact on the processes occurring during biomass combustion [27].
Increased content of K and Cl can reduce the melting point of ash and cause corrosion. High concentrations of N and S can lead to increased NOx and SO2 formation during combustion. Miscanthus mineral concentration varies depending on the type of plant, place of growth, time of harvest and even the type of fertilisation. Late harvest is the preferred fuel due to the lower content of K, Cl and N. Some studies provide the trace element composition of the miscanthus: S – 0.7–1.9 g/kg, Ca – 0.5–1.4 g/kg, Mg – 0.2–0.6 g/kg, P – 0.4–1.1 g/kg [28].
Ash content is an important parameter for fuel. The indicator represents the mass fraction of noncombustible residue (calculated as anhydrous weight) percentage, which results from mineral impurities of the fuel during its complete combustion. According to generalised data, miscanthus ash consists of 20–40% SiO2, 20–25% K2O, 5% P2O5, 5% CaO and 5% MgO. Its composition depends on the content of silt and clay in the soil. High ash content leads to the formation of slag and causes thermal process agglomeration, thereby lowering combustion efficiency of biomass plant [29].
Biomass high moisture content impedes its combustion, causing a problem of transportation. Moreover, in the process of wet fuel combustion, a large number of volatile side-products are released. Table 4 shows composition of volatile products, ash content, and molar internal energy (Ea) of miscanthus. The parameters presented are influenced by the harvest period, plant species, and climate [30].
In addition to biofuels production, the Miscanthus plants can be used for obtaining biologically active substances. The total amount of extractives based on the dry substance is redistributed in the range of from 0.3 to 2.2% with different extraction reagents. Also more than 20 hydroxycinnamic acids and their derivatives were discovered and described. The interest in these compounds is justified by the potential of plant phenols in the pharmaceutical industry. They can be used as antioxidant, antimicrobial, anti-inflammatory, anticancer biological active substances for manufacturing drugs to prevent HIV, thrombosis and atherosclerosis, reduce cholesterol, etc. [31, 32].
Features of processing Miscanthus raw materials for energy industry products. Miscanthus is the main energy culture, because it has the most optimal flow ratio of in/out energy content parameters [33]. Miscanthus as a lignocellulosic biomass with a low moisture content can be processed into fuel thermochemically. Figure 4 shows a simplified scheme of the two main ways of producing chemicals and fuels from thermal conversion of miscanthus biomass [34].
The first way is gasification, followed by Fischer-Tropsch synthesis, which requires large-scale installations. Large-scale installations cannot be adapted to biomass supply chain without biomass pyrolysis energy compaction before its long range transport. The second way is fast pyrolysis or biomass liquefaction, with the consequent quality biological oils increase in modified refrigerators [35].
Fast miscanthus pyrolysis was investigated in a fluidised bed reactor for production of bio-oil depending on the temperature (350–550°C), particle size (0.3 mm–1.3 mm), feed rate and gas flow rate. The highest bio-oil yield of 69.2% was observed at the temperature of 450°C. With increasing temperature the amount of oxygenates in bio-oil gradually decreased, and the amount of water and aromatics increased. The output of the bio-oil did not depend on particle size or feed speed. The use of gaseous products as a medium for fluidisation increased the yield of bio-oil. It was also shown that partial removal of sodium and potassium increases the yield of Miscanthus × giganteus volatile substances due to the formation of semi-coke [36].
Miscanthus gasification study was carried out in a fluidised layer using olivine as the primary catalyst. It was shown that miscanthus raw material produces about 1.1 m3/kg gas containing more than 40% of H2 and 24% CO. Gas outlet and H2 concentration increase with temperature while the yields of tar, semi-coke, CO, CO2 and CH4 decrease.
Experiments on miscanthus gasification were carried out in a circulating fluidised bed in the presence of oxygen, magnesite or olivine as a granular catalyst and kaolin as the additive to reduce agglomeration of the layer. Alkaline elements, mainly Na, K and Cl in the ash of miscanthus lead to agglomeration of the silicarich material in the fluidised bed. The use of magnesite as an additive or as a bed material leads to a significant increase in the hydrogen fraction volume in the gaseous product. Its maximum volume fraction can reach up to 40% during the gasification of biological material with a layer of magnesite. Magnesite has also shown excellent results in resin content reduction and increase in hydrogen/carbon dioxide ratio (H2:CO).
Thus, the analysis of scientific literature confirms the prospects of miscanthus as a source of energy. It can be briquetted or granulated. Combusted pellets from raw materials of miscanthus (Miscanthus × giganteus) demonstrated that the energy value of this product reaches 29 kJ/g. Meanwhile, low-temperature slow pyrolysis is energetically more favourable [37].
Features of processing plant biomass to bioethanol. The use of lignocellulosic biomass as a source of raw materials for the production of bioethanol has some complications, lying in its complex structure. It is established that the necessary preliminary chemical treatment of raw materials is needed. The process of raw materials bioconversion into bioethanol may include both separate hydrolysis and fermentation and simultaneous saccharification and fermentation, known, scientifically, as SHF and SSF processes, respectively.
One of the main advantages of SHF is the ability of enzyme preparations and microorganisms to operate under their optimal conditions. However, a disadvantage of stages continuous implementation is excessive length. For the purposes of optimisation, today the consistent process is faced with an alternative of SSF.
The advantage of this process is the carryingout of saccharification and fermentation in one reactor, shortening time process steps and reduction in energy consumption. It is also known that in the simultaneous process with introduction of bioethanol producers reducing substances begin to escape from the system, getting used for the synthesis of bioethanol. Thus, the equilibrium of the cellulose hydrolysis enzymatic reaction is continuously shifted toward the formation of reaction products (glucose), achieving saccharification intensification. However, one of the drawbacks of the simultaneous method is the difference in optimum temperatures needed for enzyme activity at saccharification stage (45–50°C) and for microorganisms cultivation (28–30°C) [9].
The main bioethanol producer in Russia is the yeast Saccharomyces cerevisiae, used in ethyl alcohol production both on food raw materials and hydrolysis media. In some sources these microorganisms are considered as bioethanol producers on hydrolysates of various types obtained from miscanthus raw materials.
For example, the paper by Baibakova shows the scheme of obtaining ethanol as a result of bioconversion using Saccharomyces serevisiae RNCIM Y-1693, isolated from the reactor of Kotlas (Arkhangelsk region) pulp and paper mill [28]. The peculiarity of the strain is its resistance to harmful impurities of hydrolysates. Optimal conditions for the strain are the temperature of 26–28°C and native active acidity of the extract of 4.5–4.7 pH. Earlier it was shown that this strain is resistant to lack of nutrients in the medium, products of its own metabolism and media obtained from cellulosecontaining raw materials by enzymatic hydrolysis. The raw material was subjected to preliminary chemical treatment by alkaline delignification, after which the products of alkaline delignification were converted into a solution of monosaccharides by enzymatic hydrolysis. Further, bioethanol was synthesised on the obtained media [29].
Another paper provides information that bioethanol is also obtained by converting the strain with Saccharomyces cerevisiae Y-1693, but a solution of nitric acid is used for pretreatment. In this case, bioethanol yield reached 70.9 % [28, 30, 31].
The use of consortium for enzymatic hydrolysate of miscanthus cellulose based on Pachysolen tannophilus and Saccharomyces cerevisiae strains is also described. The yield of ethanol amount to 44 % for P. Tannophilus RNCIM Y-1532 producer; to 62.5% for S. сerevisiae RNCIM Y-1693 of theoretically possible. With the combined use of cultures, the rate of fermentation increases by 10% compared to S. cerevisiae RNCIM Y-1693, but there is no increase in the proportion of ethanol yield. Joint use of strains was considered inappropriate [28, 31].
Bioconversion by enzyme preparations in combination with hydrolysis by dilute nitric acid at 90–96°C or alkaline delignification by 4% sodium hydroxide solution at 90–96°C is used for pretreatment of raw materials from miscanthus plants. Preparations ‘Cellolux-A’ (Sibbiopharm Ltd, Berdsk) and ‘Bruzime BGX’ (Polfa Tarchomin Pharmaceutical Works S. A., Poland) are used as cellulolytic enzymes.
‘Cellolux-A’ is positioned in the market as cellulase for non-starch polysaccharides fermentation, ‘Bruzime BGX’ – as hemicellulase [38]. As a result of enzymatic methods of miscanthus raw materials hydrolysis, bioethanol with a low content of ethers and fusel oils was obtained. There is no methanol in bioethanol obtained from miscanthus. However, saccharomycetes do not ferment pentoses, whose amount in hydrolysates can be significant (depending on the type of raw material and the method of hydrolysate obtaining), into ethanol. Several types of yeast are known to ferment xylose into ethanol: Pachysolen tannophilus, Candida shehatae, Candida tropicalis, Pichia stipitis, etc. To select a bioethanol producer, it is necessary to determine the specific rate of yeast biomass growth and the rate of substrate utilisation on synthetic media.
In addition to the use of wild strains, work is underway to obtain recombinant ones with increased capacity for bioconversion of raw materials. Thus, the patent CN 106701605 Huazhong Agricultural University presents a modified Saccharomyces cerevisiae SF4 yeast for efficient ethanol fermentation using xylose [32].
Specifics of processing plants into products for the chemical and microbiological industry. Beside the process of converting miscanthus raw materials to produce biofuels, a large amount of research is devoted to the production of cellulose fibres. Cellulose is widely used in modern industry, e.g. as a tablet excipient in pharmaceuticals, for the manufacture of fabrics, paper, plastics, explosives, etc.
The paper by Gismatulina describes obtaining cellulose from miscanthus of Soranovski variety (Miscanthus sinensis Andersson) by the nitrate method featuring two consecutive stages of processing the crushed material with diluted solutions of nitric acid, then sodium hydroxide [33].
The cellulose obtained by the nitrite method is characterised by high quality: the mass fraction of α-cellulose is 96.1%, the degree of polymerisation is 970, the ash content and mass fraction of lignin are 0.11 and 0.65%, respectively, the mass fraction of pentosans is 0.8%. Miscanthus cellulose is similar in quality to cotton cellulose. With these parameters, it can be successfully used for the synthesis of cellulose ethers and other valuable products.
In another paper, miscanthus samples are cellulose from the leaf and stem of miscanthus obtained separately by two methods (nitrite and combined) [34]. The nitrite method consists in cooking raw material in a dilute solution of nitric acid at atmospheric pressure, followed by treatment with a dilute solution of sodium hydroxide. Thus, cellulose obtained from the stem by the nitrite method has a better quality than that from the leaf. This is reflected in high values of α-cellulose content (94.4% vs. 91.7%) and degree of polymerisation (800 vs. 580), and also low values of noncellulosic compounds mass fraction: ash – 0.07% vs. 1.01%, acid-insoluble lignin – 0.45% vs. 1.51%.
Celluloses obtained by the combined method demonstrate the same regularity: cellulose from the stem is characterised by higher quality than that from the leaf. The data show high value of polymerization degree – 1040 vs. 640 and low noncellulosic compounds mass fraction: ash – 0.14% vs. 0.75%, acid-insoluble lignin – 0.88% vs. 4.12%, pentosans – 6.38% vs. 8.53%. The cellulose obtained by the nitrite method may be suitable for chemical modifications, including nitration. The cellulose obtained by the combined method can be used in paper industry [31].
The use of miscanthus as a medium for bacteria cultivation can be carried out without the targeted production of simple sugars for use in the food, feed and pharmaceutical industries, as well as a substrate for bacterial growth. Some studies on bacterial celluloses production present media based on incomplete miscanthus hydrolysates.
Thus, Gladysheva describes obtaining bacterial cellulose by bioconversion of Medusomyces gisevii bacteria on a synthetic nutrient medium, including sucrose, black tea extract, starch hydrolysate, enzymatic miscanthus hydrolysate [35]. Cultivation was carried out in static conditions at 25–29°C for 13 days.
Gismatulina also used miscanthus raw materials to obtain a nutrient medium for the growth of bacteria producing bacterial cellulose [36, 37]. Pre-hydrolysis was carried out with 0.2% solution of nitric acid at 90–95°C for 1 h. Nitric acid treatment was carried out with 4% nitric acid solution at 90–95°C for 6 h. Washing was performed successively with 1% sodium hydroxide solution, and then 1 % nitric acid solution. The resulting pulp was pressed with a vacuum filter, washed to a neutral reaction of washing water, dried to a moisture content of 7–10 %.
The raw material for the experiments was ground to a particle size of 10–15 mm. It was established that optimal conditions of the principal and longest stage of obtaining cellulose by the combined method (alkaline treatment) are: sodium hydroxide concentration, 4%; temperature, 90–98°C; duration, 6 h. Cellulose extraction under such conditions allows obtaining the maximum yield of the target product – 35–40% with α-cellulose content of 87.0–90.3%, degree of polymerisation 950–990, residual lignin content of 2.0 to 3.0%, ash content of 0.3–0.4%, and pentosan content of 3.0 to 8.0%.
Cellulose isolated from miscanthus by the combined method is a promising substrate for enzymatic hydrolysis, with the degree of its conversion was 91–93% by weight of the substrate. High quality indicators of the substrate allow predicting the effectiveness of its use for the subsequent bacterial cellulose biosynthesis.
Also miscanthus raw materials can provide organic acids, alcohols and adsorbents. The paper describes obtaining formic acid from lignocellulose or its major components, which comprises two successive stages:
– acid-catalysed depolymerisation (polysaccharides hydrolysis, delignification);
– subsequent monomers (monosaccharides, phenolic derivatives) oxidation into formic acid. A high yield of formic acid equal to 45% was obtained [23].
Organosolv method of cooking miscanthus raw materials can also deliver ferulic, vanilla and paracumaric acids, sterines, among which the main factions are β-sitosterol, 7-oxo-β-sitosterol, stigmasterol and campesterol. However, this method has not become widespread, as sterol derivates are oxidized during preliminary treatment with organic solvents.
ВЫВОДЫ
Furthering lignocellulose biomass integrated processing by chemical and/or biotechnological methods into a range of competitive products and energy is a modern and fundamental area of industrial biotechnology developing in industrial countries.
The conducted botanical properties analysis of chemical composition and modern methods of processing miscanthus species biomass proved that it was a promising renewable wood-substituting raw material for products of chemical, fuel, energy, and microbiological industries. Further research will focus on the selection of rational parameters of processing miscanthus biomass into valuable products with improved physical and chemical characteristics, such as bio-ethanol, pellets, technical cellulose, bacterial cellulose, and carbohydrate-containing substrate.
КОНФЛИКТ ИНТЕРЕСОВ
The authors declare no conflict of interest.
ФИНАНСИРОВАНИЕ
The survey lies within a framework of fundamental research project No. 19-416-390001 ‘Scientific and technological foundations of Miscanthus plants biomass processing into products for the fuel and energy, chemical and microbiological industry’.
СПИСОК ЛИТЕРАТУРЫ
- Prosekov AY, Ivanova SA. Food security: The challenge of the present. Geoforum. 2018;91:73–77. DOI: https://doi.org/10.1016/j.geoforum.2018.02.030.
- Gushina VA, Volodkin AA, Ostroborodova NI, Agapkin ND, Letuchiy AV. Peculiarities of growth and development of introduction of miscanthus gi-ganteus in the conditions of forest-step zone in Middle Volga. The Agrarian Scientific Journal. 2018;(1):10–13. (In Russ.).
- Volobaev VP, Larionov AV, Kalyuzhnaya EE, Serdyukova ES, Yakovleva S, Druzhinin VG, et al. Associations of polymorphisms in the cytokine genes IL1β (rs16944), IL6 (rs1800795), IL12b (rs3212227) and growth factor VEGFA (rs2010963) with anthracosilicosis in coal miners in Russia and related genotoxic effects. Mutagenesis. 2018;33(2):129–135. DOI: https://doi.org/10.1093/mutage/gex047.
- Gismatulina YuA. Comparative chemical composition of five miscanthus var. ‘Soranovskiy’ harvests: whole plant, leaf, and stem. Advances in current natural sciences. 2016;(4):23–26. (In Russ.).
- Sarkar A, Asaeda T, Wang QY, Rashid MH. Arbuscular mycorrhizal influences on growth, nutrient uptake, and use efficiency of Miscanthus sacchariflorus growing on nutrient-deficient river bank soil. Flora. 2015;212:46–54. DOI: https://doi.org/10.1016/j.flora.2015.01.005.
- Tamura K, Sanada Y, Shoji A, Okumura K, Uwatoko N, Anzoua KG, et al. DNA markers for identifying interspecific hybrids between Miscanthus sacchariflorus and Miscanthus sinensis. Grassland Science. 2015;61(3):160–166. DOI: https://doi.org/10.1111/grs.12089.
- Zhang J, Yang SY, Huang YJ, Zhou SB. The tolerance and accumulation of Miscanthus Sacchariflorus (maxim.) benth., an energy plant species, to cadmium. International Journal of Phytoremediation. 2015;17(6):538–545. DOI: https://doi.org/10.1080/15226514.2014.922925.
- Tamura K, Uwatoko N, Yamashita H, Fujimori M, Akiyama Y, Shoji A, et al. Discovery of Natural Interspecific Hybrids Between Miscanthus Sacchariflorus and Miscanthus Sinensis in Southern Japan: Morphological Characterization, Genetic Structure, and Origin. Bioenergy Research. 2016;9(1):315–325. DOI: https://doi.org/10.1007/s12155-015-9683-1.
- Baybakova OV. Study into simultaneous saccharification and fermentation for bioethanol production by the example of miscanthus and oat hulls. Fundamental research. 2016;(6–1):14–18. (In Russ.).
- Sarkar A, Asaeda T, Wang QY, Kaneko Y, Rashid MH. Response of Miscanthus sacchariflorus to zinc stress mediated by arbuscular mycorrhizal fungi. Flora. 2017;234:60–68. DOI: https://doi.org/10.1016/j.flora.2017.05.011.
- Grams J, Kwapinska M, Jedrzejczyk M, Rzenicka I, Leahy JJ, Ruppert AM. Surface characterization of Miscanthus × giganteus and Willow subjected to torrefaction. Journal of Analytical and Applied Pyrolysis. 2019;138:231–241. DOI: https://doi.org/10.1016/j.jaap.2018.12.028.
- Gismatulina YuA. Chemical composition study of sb ras miscanthus variety harvested in 2013. Fundamental research. 2014;(1):47–60. (In Russ.).
- Ilʹyasov SG, Cherkashin VA. Poluchenie i svoystva shchelochnogo lignina iz miskantusa kitayskogo [Production of alkaline lignin from miscanthus Chinese and its properties]. Polzunovsky vestnik. 2014;(4–2):137–142. (In Russ.).
- Redcay S, Koirala A, Liu JD. Effects of roll and flail conditioning systems on mowing and baling of Miscanthus × giganteus feedstock. Biosystems Engineering. 2018;172:134–143. DOI: https://doi.org/10.1016/j.biosystemseng.2018.06.009.
- Makarova EI, Budaeva VV. Bioconversion of non-food cellulosic biomass. Part 1. Proceedings of Universities. Applied Chemistry and Biotechnology. 2016;6(2)(17):43–50. (In Russ.). DOI: https://doi.org/10.21285/2227-2925-2016-6-2-43-50.
- Dabkowska K, Alvarado-Morales M, Kuglarz M, Angelidaki I. Miscanthus straw as substrate for biosuccinic acid production: Focusing on pretreatment and downstream processing. Bioresource Technology. 2019;278:82–91. DOI: https://doi.org/10.1016/j.biortech.2019.01.051.
- Gismatulina YuA, Budaeva VV. Chemical composition of five Miscanthus sinensis harvests and nitric-acid cellulose therefrom. Industrial Crops and Products. 2017;109:227–232. DOI: https://doi.org/10.1016/j.indcrop.2017.08.026.
- Hoover A, Emerson R, Ray A, Stevens D, Morgan S, Cortez M, et al. Impact of Drought on Chemical Composition and Sugar Yields From Dilute-Acid Pretreatment and Enzymatic Hydrolysis of Miscanthus, a Tall Fescue Mixture, and Switchgrass. Frontiers in Energy Research. 2018;6. DOI: https://doi.org/10.3389/fenrg.2018.00054.
- Plazek A, Dubert F, Kopec P, Krepski T, Kacorzyk P, Micek P, et al. In vitro-propagated Miscanthus × giganteus plants can be a source of diversity in terms of their chemical composition. Biomass and Bioenergy. 2015;75:142–149. DOI: https://doi.org/10.1016/j.biombioe.2015.02.009.
- Lanzerstorfer C. Chemical composition and properties of ashes from combustion plants using Miscanthus as fuel. Journal of Environmental Sciences. 2017;54:178–183. DOI: https://doi.org/10.1016/j.jes.2016.03.032.
- Morgun IA, Andreeva LS. Kapelʹnoe oroshenie kak faktor intensifikatsii vegetativnogo razmnozheniya miskantusa [Drip irrigation as a factor in the intensification of miscanthus vegetative propagation]. Vestnik Belorusskoy gosudarstvennoy selʹskokhozyaystvennoy akademii [Bulletin of the Belarussian state agricultural Academy]. 2016;(4):93–95. (In Russ.).
- Ashman C, Awty-Carroll D, Mos M, Robson P, Clifton-Brown J. Assessing seed priming, sowing date, and mulch film to improve the germination and survival of direct-sown Miscanthus sinensis in the United Kingdom. Global Change Biology Bioenergy. 2018;10(9):612–627. DOI: https://doi.org/10.1111/gcbb.12518.
- Brosse N, Dufour A, Meng XZ, Sun QN, Ragauskas A. Miscanthus: a fast-growing crop for biofuels and chemicals production. Biofuels Bioproducts and Biorefining-Biofpr. 2012;6(5):580–598. DOI: https://doi.org/10.1002/bbb.1353.
- Lee S, Han J, Ro HM. Interpreting the pH-dependent mechanism of simazine sorption to Miscanthus biochar produced at different pyrolysis temperatures for its application to soil. Korean Journal of Chemical Engineering. 2018;35(7):1468–1476. DOI: https://doi.org/10.1007/s11814-018-0054-4.
- Bondar VS, Fursa AV. Economic ground for technologies of plant biomass growing and processing into solid fuels. The Economy of Agro-Industrial Complex. 2015;245(3):22 – 27. (In Russ.).
- Schafer J, Sattler M, Iqbal Y, Lewandowski I, Bunzel M. Characterization of Miscanthus cell wall polymers. Global Change Biology Bioenergy. 2019;11(1):191–205. DOI: https://doi.org/10.1111/gcbb.12538.
- Skiba EA. Determination procedure for biological goodness of hydrolyzates from cellulosic biomass using Saccharomyces cerevisiae Y-1693 strain. Proceedings of Universities. Applied Chemistry and Biotechnology. 2016;6(1)(16):34–44. (In Russ.).
- Baybakova OV. Fermentation of enzymatic hydrolyzate of miscanthus cellulose by Pachysolen tannophilus Y-1532 and Saccharomyces cerevisiae Y-1693. Fundamental research. 2014;(9–5):949–953. (In Russ.).
- Skiba EA, Mironova GF. Advantages of combining biocatalytic stages in bioethanol synthesis from cellulosic biomasses. Proceedings of Universities. Applied Chemistry and Biotechnology. 2016;6(4)(19):53–60. (In Russ.). DOI: https://doi.org/10.21285/2227-2925-2016-6-4-53-60.
- Baibakova OV, Skiba EA. Biotechnological view of ethanol biosynthesis from miscanthus. Vavilov Journal of Genetics and Breeding. 2014;18(3):564–571. (In Russ.).
- Baibakova OV. Bioconversion of miscanthus lignocellulosic substrate into ethanol. Fundamental research. 2015; (2–13):2783–2786. (In Russ.).
- Peng L, Hao B, Xia T. Transgenic engineering Saccharomyces cerevisiae SF4 for efficiently fermenting ethanol using xylose. Patent CN 106701605. 2017.
- Gismatulina YuA. Quality of pulp obtained by the dilute nitric-acid method from miscanthus harvested in 2013. Fundamental research. 2015;(2–18):3948–3951. (In Russ.).
- Gismatulina YuA, Budaeva VV, Veprev SG, Sakovich GV, Shumny VK. Cellulose from various parts of Soranovskii miscanthus. Vavilov Journal of Genetics and Breeding. 2014;18(3):553–53. (In Russ.).
- Gladysheva EK. Bacterial cellulose x-ray study results. Fundamental research. 2015;(7–2):240–244. (In Russ.).
- Gismatulina YuA. Masshtabirovanie azotnokislogo sposoba polucheniya tsellyulozy iz miskantusa [Gismatulina Scaling nitrate method of obtaining cellulose from miscanthus]. Polzunovsky vestnik. 2015;(4–2):108–111. (In Russ.).
- Gismatulina YuA. Chemical pretreatment of miscanthus for subsequent bacterial cellulose synthesis. Fundamental research. 2017;(9–2):284–289. (In Russ.).
- Budaeva VV, Skiba EA, Baybakova OV, Makarova EI, Orlov SE, Kukhlenko AA, et al. Kinetics of enzymatic hydrolysis of lignocellulosic materials at different concentrations of substrat. Catalysis in Industry. 2015;(5):60–66. (In Russ.). DOI: https://doi.org/10.18412/1816-0387-2015-5-60-66.