Аннотация
Safe and healthy nutrition has a beneficial effect on human well-being. Various foods, such as berries, are known to inhibit cancer-promoting pre-proliferative signals. Among European fruit and berry crops, raspberries demonstrate one with the widest ranges of biologically active substances. Extraction remains a reliable method of obtaining biologically active substances from plant materials. The research objective was to obtain a semi-finished raspberry product by using microwave and ultrasonic processing and to study its antioxidant, anti-carcinogenic, sensory, physico-chemical, and microbiological properties. The raspberry extracts were obtained by maceration, ultrasound treatment, and microwave processing. After that, the samples underwent a comparative analysis of their antioxidant properties. The ultrasonic method gave the best results. A set of experiments made it possible to define the optimal technological modes for the extraction process: ethanol = 50%, ultrasonic radiation = 35 kHz, temperature = 40 ± 5°C, time = 120 min, water ratio = 1:10. A set of experiments on cell cultures demonstrated that the raspberry extract was able to reduce the expression of the anti-inflammatory COX-2, iNOS, and IL-8 genes. Hense, we recommend further studies of the effect of the raspberry extract on the induced expression of COX-2, iNOS, and IL-8. In addition, its anticarcinogenic properties have to be studied in vivo.Ключевые слова
Extraction of plant materials, phenolic substances, PRC-analysis, expression of anti-inflammatory genes, inhibition, ultrasound, microvavesВВЕДЕНИЕ
In many ways, human well-being is associated with safe and healthy food. Food safety is often understood as the absence of harmful microorganisms and chemicals, including synthetic additives, while its healthiness is often identified with naturalness and freshness [1]. The increasing demand for safe and natural food has become a major problem for food science and industry over the past decades [2]. Despite the use of various traditional and modern antibacterial agents, as well as chemical preservatives, about a third of the population of industrialised countries suffer from foodborne diseases [3]. Meanwhile, more and more consumers demand fresh, ready-to-eat, minimally processed foods that contain neither chemical preservatives nor synthetic additives [4]. For modern foods, it is not enough to be a source of energy: modern food has to be functional, e.g. to possess antioxidant or anti-carcinogenic properties. This trend has triggered multiple studies in the field of food processing, as well as an active search for alternative natural supplements with a wide spectrum of physiological properties [5].
In the process of evolution, plants developed natural mechanisms of defense against microbial infections and other harmful environmental factors. Plants are known to produce antimicrobial peptides, lectins, polyphenols, terpenoids, essential oils, and other biologically active compounds. According to some studies, phenolic substances obtained from berries can act as a new type of food components that can inhibit a wide range of pathogens, e.g. Salmonella, Escherichia, and Staphylococcus [6].
Cancer is a serious social problem in many parts of the world [7]. According to the World Health Organization, cancer is responsible for about 7.6 mln (13%) of the 59 mln annual deaths. By 2030, cancer mortality worldwide is expected to reach 11.8 mln per year. Most cancer deaths are caused by five basic behavioral and eating habits, including a low intake of fruits and vegetables. Bad eating habits, obesity, and sedentary lifestyle have been proven to increase the risk of oncological diseases [8]. Therefore, a healthy diet is one of the most important changes in one’s lifestyle that can reduce the risk of cancer [9].
Recent epidemiological and basic studies have demonstrated the anticarcinogenic properties of fruit components. They can inhibit pro-proliferative signals that cause or stimulate the growth of tumors or cancer cells [10].
Raspberries are one of the major fruit and berry crops in Europe [11]. They can be used both fresh and processed [12].
Raspberry polyphenols consist mainly of anthocyanins and tannins [13]. Raspberries are a rich source of cyanidin glycoside. A high content of ellagitannin releases free ellagic acid during hydrolytic processes [14]. These components are responsible for about 60% of the antioxidant potential [15]. According to Landele, raspberries owe their antioxidant, antimicrobial, and anti-inflammatory properties to ellagic acid and ellagotanin [16]. Thus, raspberries and their semi-finished products can prevent many diseases, if included in foods.
Raspberries contain a wide range of phytonutrients with antioxidant, antitumor, anti-neurodegenerative, and anti-inflammatory properties [17, 18]. The following substances are responsible for the hemotherapeutic and prophylactic components: vitamins C and E, folic acid, calcium, selenium, β-carotene, α-carotene, lutein, such polyphenols as ellagic acid, ferulic acid, p-coumaric acid, anthocyanins, quercetin, and kaempferol, and such phytosterols as β-sitosterol and stigmasterol [19].
A raspberry ethanol extract suppresses cell proliferation in squamous cell carcinoma without altering cell viability and inhibits the expression of vascular endothelial growth factor. In addition, it also inhibits nitric oxide synthase activity and indicates apoptosis and terminal differentiation [20]. These data suggest that raspberry extract can be used for chemoprophylaxis in people with oral cavity dysplasia.
Solvent extraction can be used to obtain polyphenols [21]. Unfortunately, it has several drawbacks. For instance, it requires a larger amount of organic solvents and a longer extraction time. Moreover, the solvent can have a negative effect on human health and degrade the target compounds. However, there are several alternative methods that can either eliminate or reduce these disadvantages [22].
There are several factors that affect the efficiency of extraction of biologically active components from plant materials. They include technology, solvent type, time, temperature, material vs. solvent ratio, etc. However, it is technology that remains crucial. The traditional extraction technologies, such as Soxhlet extraction and maceration, require a lot of solvent, time, and energy, but are popular and effective. However, new extraction technologies are being actively introduced, e.g. ultrasonic, microwave, infrared, and fluid supercritical extractions. They are energy saving and environmentally friendly, according to one of the latest books on the extraction of biological active substances from plant and animal raw materials [23]. Still, an optimal extraction technology should be simple, safe, reproducible, inexpensive, and suitable for industrial use [24].
Ultrasonic (US) extraction is a fairly cheap method that requires minimal hardware design [25]. It destroys cell walls (lysis) and disintegrates individual cellular structures and the cell as a whole, which increases the number of components that enter the extract. US produces a mechanical effect: the solvent penetrates into the matrix of berries, thus increasing the area of the contact surface between the solid and the liquid phases [26]. Moreover, US waves can cause some undesirable chemical processes that can change the chemical composition, degrade the target compounds, and cause free radicals in gas bubbles [27]. Therefore, a set of experiments is required to define the optimal extraction conditions, i.e. time, temperature, power, and ultrasonic frequency.
Microwave (MW) radiation is another possible way to increase extraction efficiency [28]. MW radiation is a popular means of extraction, as far as low-molecular compounds from plant raw material are concerned.
The research objective was to obtain a semi-finished raspberry product using MW and US processing, as well as to study its antioxidant, anti-carcinogenic, sensory, physico-chemical, and microbiological properties.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
The experiments were performed on the premises of the Department of Technology and Catering at the Samara State Technical University (Samara, Russia). The anti-inflammatory and cytostatic, or cytotoxic, properties were determined in the N.N. Blokhin National Medical Research Oncology Center (Moscow, Russia).
The research featured a variety of fresh raspberries (Rúbus idáeus L.) harvested in the Samara region (53°12′N - 50°06′E) in 2017. The raspberries were provided by the Research Institute of Horticulture and Medicinal Plants ‘Zhigulyovskie Sady’ (Samara, Russia).
Determination of the antioxidant properties indicators.
Chemicals and reagents. The experiment used ethanol and distilled water. The Folin-Ciocalteu reagent (FCR) and the gallic acid were provided by the Fluka company (Germany). The DPPH (2,2-diphenyl-1-picrylhydrazyl), sodium nitrite, aluminum chloride, sodium carbonate, and linoleic acid were ordered from Sigma-Aldrich, Inc. The 2,4,6-tri(2-pyridyl)-s-triazine (TPTZ) was purchased from Fluka Chemicals (Spain). Other chemicals included hydrochloric acid, potassium chloride, acetic acid, sodium acetate, sodium phosphate, ferric chloride (II), ferric chloride (III), and ammonium rodanide.
Phenolic compounds. The content of total phenols was estimated using a modified version of the FCR method [29]. Gallic acid was used as a standard: an aqueous solution of gallic acid (200 mg in 1000 cm3) was diluted with distilled water to obtain the concentrations appropriate for the calibration curve. The experiment involved 0.50 cm3 of the analysed substance or standard gallic acid, 4.00 cm3 of distilled water, 0.25 cm3 of FCR reagent, and 0.25 cm3 of a saturated aqueous solution of sodium carbonate. The samples were shaken and kept in the dark at room temperature for 30 min. The absorption coefficient was determined at 725 nm with a spectrophotometer. Results were expressed in mg equivalent of gallic acid per 100 g of dry weight. The experiment was performed in triplicate.
Flavonoids. The content of flavanoids was determined using a modified method described Demidova et al [30]. 0.50 cm3 of the analysed substance or standard catechin solution was put in a 10 cm3 measuring tube. After that, 2.50 cm3 of distilled water was added at the time zero followed by 0.15 cm3 of a 5% aqueous solution of sodium nitrate. After 5 minutes, 0.30 cm3 of a 10% aqueous solution of aluminum chloride was added and kept for another 5 min. The absorption coefficient was measured at 510 nm. The content of flavonoids was expressed in mg equivalent of catechin per 100 g of dry weight. The experiment was performed in triplicate.
Anthocyanins. To define the total content of anthocyanins, the absorption coefficient was measured at two different pH values (1.0 and 4.5) at 515 and 700 nm [31]. The content of anthocyanins was expressed in mg equivalent of cyanidin-3-glycoside per 100 g of dry matter. The experiment was performed in triplicate.
Antioxidant activity in the linoleic acid system. The antioxidant activity in the linoleic acid system was determined according to the method described Karabegovic [32]. 0.5 cm3 of ethanol, 0.5 cm3 of distilled water, 1 cm3 of linoleic acid, and 2 cm3 of phosphate buffer (pH 7.0) were added to 1.0 cm3 of the analysed substance. The mixture was kept at 40°C for 120 h. Then an aliquot part (0.1 cm3) was isolated from the mixture. After that, 9.7 cm3 of 75% ethanol and 0.1 cm3 of a 30% ammonium rhodanide solution were added to the aliquot and allowed to stand for 4 min. Subsequently, 0.1 cm3 of ferric chloride (II) solution was added to the mixture (0.2 M in 3.5% of HCl). A spectrophotometer was used to measure the optical density of the mixture at 500 nm. The control sample contained all the reagents but the extract. The antioxidant activity was expressed in percent of inhibition of linoleic acid oxidation. The experiment was performed in triplicate.
Antioxidant activity by DPPH. The antioxidant properties of the samples were measured using the method described Cheigh et al [33]. The method is based on the ability of the antioxidants of the raw material to bind the stable chromogen radical of 2,2-diphenyl-1-picrylhydrazyl (DPPH). 4 mg of DPPH was dissolved in 100 cm3 of ethanol. The aliquots were dissolved in 100 cm3 of distilled water in the quantities of 0.05, 0.10, 0.40, 0.80, 1.00, and 5.00 cm3. Then, 0.2 cm3 of each solution was added to 2.0 cm3 of the DPPH solution at 20°C and kept in the dark for 30 min. The transmittance was determined at 517 nm. The antiradical activity was expressed as the concentration of the original object in mg/cm3, at which 50% of the radicals were bound. The experiment was performed in triplicate.
FRAP method. The restoring force of the analysed substance was determined by the FRAP method [34]. A freshly prepared FRAP solution included 10 cm3 of acetate buffer (pH 3.6), 1 cm3 of a 10% solution of ferric chloride (III) and 1 cm3 of TPTZ solution (2,4,6-tripyridyl-s-triazine) (10 mmol/L TPTZ in 40 mmol/1000 cm3 of HCl). The solution was kept at 37°C for 10 min. After that, 3.0 cm3 of distilled water and 1 cm3 of FRAP solution were added to the analysed substance (0.1 cm3). The mixture was allowed to stand at 37°C for 4 min. The optical density was measured at 593 nm. The restoring force was determined according to the calibration graph and expressed in mmol of Fe2+/1 kg of the raw material. The experiment was performed in triplicate.
The sensory properties of the raspberry extract were defined according to State Standard 8756.1-2017.
The microbiological studies of the semi-finished product were performed according to State Standards 31659-2012 and State Standard 30712-2001 in licenced testing laboratory No. ROSS RU.0001.510137.
The physical and chemical properties were determined according to State Standards 34128-2017 and State Standards 34127-2017. The content of ethanol in the raspberry extract was determined according to State Standard ISO 2448-2013. The experiments were performed in triplicate.
Statistical data processing. The statistical processing of the results was performed with the help of Student’s t-test to determine M ± m, where M is the mean value, m is the standard error of the mean (the standard deviation √n) was defined using the Microsoft Excel software.
Determination of potential anticarcinogenic properties indicators.
Anti-inflammatory drugs are known to produce an inhibitory effect on the pro-inflammatory pathways of cells, including COX2, iNOS, and IL-8. It is currently considered a proven fact that these drugs exert an anticarcinogenic effect in vivo. That is why the present study featured these very genes and the effect of raspberry extract on them to determine the potential anticanceragenic activity of the product.
The study used HCT-116 colon cancer cell line [35]. The cells were cultured at 37°C in standard DMEM medium containing 5% fetal calf serum (PAA, Australia) and gentamicin (50 U/cm3) (PanEko, Russia) and in 5% CO2.
Cell viability study (MTT-test). The cells were dispersed into 96-well plates (BDMicro-FinePlus, USA). There were 3×103 cells in 190 μl of culture medium. After that, the cells were incubated for 24 h. Serial dilutions of raspberries were prepared on the day of the experiment. The cells were incubated with the extract for 72 h at concentrations of 0.03125–2% (v/v). Then 20μL of the MTT reagent solution were added in the ratio of 5 mg/cm3 (PanEko, Russia) in Hanks salt solution (PanEko, Russia). The solution was allowed to uncubate at 37°C for 2 h until it turned violet. The formazan was then dissolved in 200 μL of dimethyl sulfoxide (DMSO, PanEko, Russia) and incubated at 37°C. After the formazan crystals had completely dissolved, the optical density of the wells was measured at a wavelength of 570 nm using a MultiScan MCC 340 multiwell spectrophotometer (Labsystems, USA). The data were presented as the optical density of the experimental samples vs. that of the control sample. The optical density in the control sample was taken for 100%. The cells in the control sample were incubated in a 1% ethanol solution.
RNA isolation. The total cellular RNA was isolated using an RNA isolation kit. The RNA concentration was determined with a spectrophotometer according to the optical density of the solution at a wavelength of 260 nm. The absence of impurities in the sample was stated by the ratio of the optical density of the solution at a wavelength of 260 nm and 280 nm.
Reverse transcription reaction. Reverse transcription was used to obtain cDNA. 1μg of RNA was mixed with 0.4 μg of random hexamer oligonucleotides, denatured at 25°C, and cooled on ice. The reverse transcription mixture included: 2 units of reverse transcriptase MMLV, a suitable buffer, 2 mM of dithiothreitol, 0.5 units of ribonuclease inhibitor, 0.5 mM of dNTP, and ≤ 20 μL of distilled water. The reaction lasted 1 h at 37°C. After that, reverse transcriptase was inactivated at 95°C for 5 min, which stopped the reaction. After adding 80 μL of distilled water, the aliquots were used for real-time PCR amplification with specific primers.
Quantitative real-time PCR analysis. After the reverse transcription reaction, the samples were diluted 1:10 with sterile deionised water to obtain working dilutions of cDNA. 5 μL of the cDNA working solution was added to 20 μL of the reaction mixture that contained SYBR Green Master Mix, 500 nM of the reverse primers and 500 nM of direct primers. A Bio-Rad iQ5 PCR analyser was used to perform a real-time quantitative PCR analysis. The amplification programme was as follows: 95°C – 10 min, 40 cycles (95°C – 15 s, 60°C – 30 s, 72°C – 30 s). The relative change in the expression of the mRNA was calculated using the ΔΔCt method. The ΔΔCt was determined by subtracting the average ΔCt of the control sample from the ΔCt of the experimental samples [36]. For each gene, a PCR analysis was performed in triplicate, and the melting curves were obtained for each primer pair to confirm their specificity. To analyse the melting curves, the temperature was raised from 55°C to 95°C at a pace of 0.5°C. The ribosomal protein gene L27 (Rpl27) was used for control.
The primers for cDNA amplification were designed using the Primer-Bank database and the Oligo 6 software [37]. Table 1 shows the primer sequences.
Statistical data processing. Statistical processing of the results performed with the help of Student’s t-test to determine M ± m, where M is the mean value, m is the standard error of the mean (standard deviation √n) was defined using the Microsoft Excel software.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
The research compares the antioxidant properties of raspberry extracts obtained by maceration, ultrasonic treatment, and microwave processing. All the extracts were obtained using 50% ethanol, while the raw material vs. solvent ratio was 1:10 (w/v).
The maceration extract was obtained by storing the raw material and the solvent at 40°C for 120 min.
The US extract was obtained using an Elmasonic S 15H device at a frequency of 50 kHz at 40°C for 120 min.
The MW extract was obtained by using microwave irradiation with a irradiation rate of 90 W for 1 min.
Figure 1 shows the total content of phenols, flavonoids, and anthocyanins.
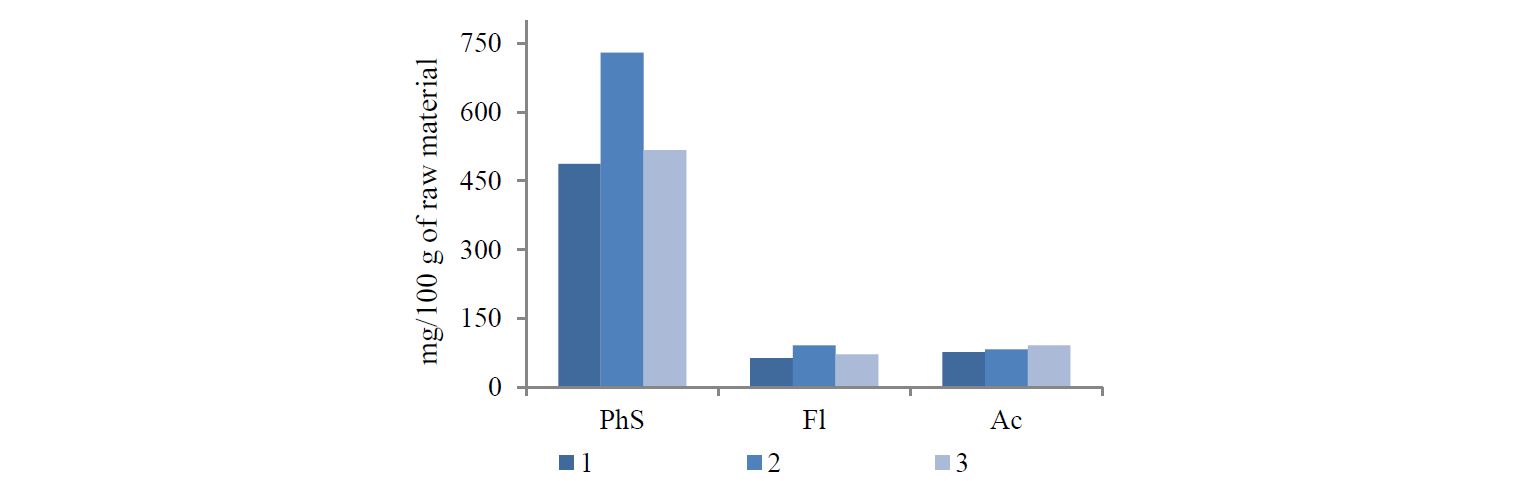
US radiation resulted in the biggest content of phenolic substances: it increased by 1.50 times as compared with classical maceration. MW radiation produced nothing but minor changes: the content of total phenolic substances in the extract increased by 1.06 times.
The US and MW processing also increased the extraction of flavonoids by 1.44 and 1.13 times, respectively.
All the methods showed nearly the same content of anthocyanins in the extracts.
Thus, both US and MW methods increased the content of biologically active substances in the raspberry extracts. US extraction proved to have the greatest impact on the content of phenolic substances and flavonoids, while the content of anthocyanins remained almost the same in different types of extraction.
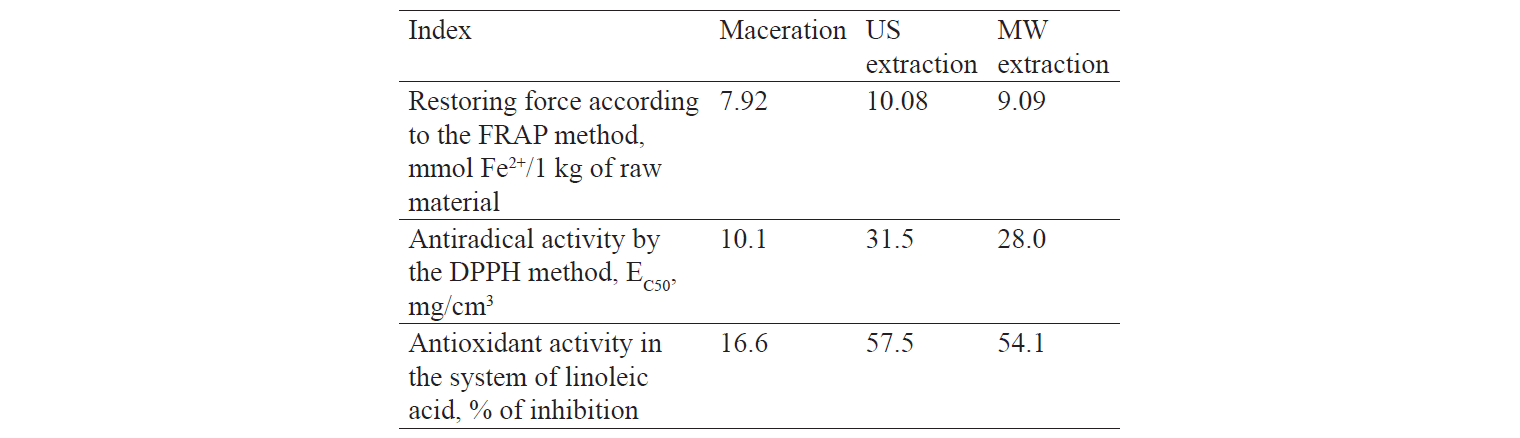
Table 2 demonstrates the antioxidant properties of the raspberry extracts.
The inhibitory effect of DPPH free radicals increased by 1.15 and 1.27 during MW and US extractions, respectively.
The restoring force of the US extract increased as compared with MW and maceration extracts.
In addition, US extraction increased the ability of the raspberry extract to inhibit linoleic acid by 3.46 times.
Similarly, additional treatment with US or MW radiation increased the antioxidant properties of the semi-finished products, if compared with classical maceration.
Thus, US processing is necessary to obtain a raspberry extract with a high content of physiologically active substances and high antioxidant properties.
The study introduces a optimal conditions for raspberry extract production. The new technological scheme is given in Fig. 2.
The experimental data made it possible to define the best technological modes: ethanol = 50%, US radiation frequency = 35 kHz, temperature = 40 ± 5°C, time = 120 min, raw materials vs. solvent ratio = 1:10. A circulation vacuum evaporator concentrated the extract until the content of soluble solids was 65% and the mass fraction of ethanol was ≤ 1.0%.
The extract was then analysed by sensory, microbiological, physicochemical, and antioxidant properties (Table 3).
Chronic inflammation is one of the main etiological factors that trigger certain types of cancer. As a result, some anti-inflammatory drugs, e.g. ibuprofen, have an anti-carcinogenic effect on colon cancer.
The main objective of this research was to study the anti-inflammatory properties of the raspberry extract. A set of experiments was conducted to study its effect on the expression of the genes of individual components of the anti-inflammatory pathway. A colon cancer cell line was studied by the RT-PCR method to measure the effect of non-toxic extract doses on the expression of the following genes: cyclooxygenase 2 (COX-2), induced NO synthase (iNOS) and interleukin 8 (IL-8) [38]. The antiinflammatory effect of the raspberry extract indicates its potential anticarcinogenic activity.
The functional activity of the COX-2 gene is directly related to inflammation. This gene is expressed bymacrophages, synoviocytes, fibroblasts, smooth vascular muscles, chondrocytes, and endothelial cells after they have been induced with cytokines or growth factors. COX-2-induced prostaglandins – directly or indirectly – enhance the production of the enzyme according to the positive feedback mechanism [39]. Inhibition of COX-2 is considered as one of the main mechanisms of the anti-inflammatory activity of nonsteroidal antiinflammatory drugs (NSAIDs). Selective inhibition of this cyclooxygenase can minimise various side effects observed during the inhibition of cyclooxygenase 1. COX-2 plays an important role in the development of inflammatory processes and carcinogenesis in the gastrointestinal tract. An increased COX-2 expression was observed in 85% of gastrointestinal tumours, which also correlated with low survival. Animal studies showed that deleting COX-2 or treating animals with selective COX-2 inhibitors reduced the number, size, and multiplicity of tumours. COX-2 causes tumour progression as it induces the expression of anti-apoptotic proteins of the Bcl-2 family, which leads to apoptosis resistance in the future [37].
IL-8 is known as a T-cell chemotactic factor and a neurophil activating factor (NAF) [40, 41]. It belongs to the group of chemokines, which provide chemotaxis in the area of inflammation of neutrophils, monocytes, eosinophils, and T-cells. IL-8 possesses pronounced pro-inflammatory properties. It causes the expression of intercellular adhesion molecules and enhances neutrophil adherence to endothelial cells and subendothelial matrix proteins. Hence, it is an important mediator of inflammatory response [42]. IL-8 is produced by macrophages, lymphocytes, epithelial cells, fibroblasts, and epidermal cells. IL-8 also regulates pro-inflammatory angiogenesis. This cytokine enhances the expression of vascular endothelial growth factor A (VEGF-A) by endothelial cells and increases the expression of vascular growth receptor 2 (VEGFR2) [43].
iNOS expression is regulated by pro-inflammatory cytokines (tumour necrosis factor-alpha (TNF-α), interleukin-1β (IL-1β), interferon-γ (IFN-γ), hypoxia, oxidative stress, and, according to recent studies, by Hsp70 heat shock protein. Inhibition of iNOS results from the suppression of the pro-inflammatory and proliferative pathways NF-κB and JAK-STAT [44].
The expression of these genes can denote the presence or absence of the anti-inflammatory effect of the extracts on colon cells. This research did not study the anticarcinogenic properties of the extract components; however, its results may indicate the feasibility of in vivo experiments to determine the anticarcinogenic properties of the raspberry extract.
We performed an MTT test to define the cytotoxicity of the raspberry extract. A wide range of concentrations (0.03125–2%, v/v) showed that the raspberry extract has a cytotoxic effect on colon cancer cells HCT-116 (Table 4).
Next, a non-toxic concentration of the raspberry extract was used to define the working concentration. It was used to study the effect of the extract on the expression level of COX-2, iNOS, and IL-8. Working concentrations used were 0.0625 and 0.03125% (v/v).
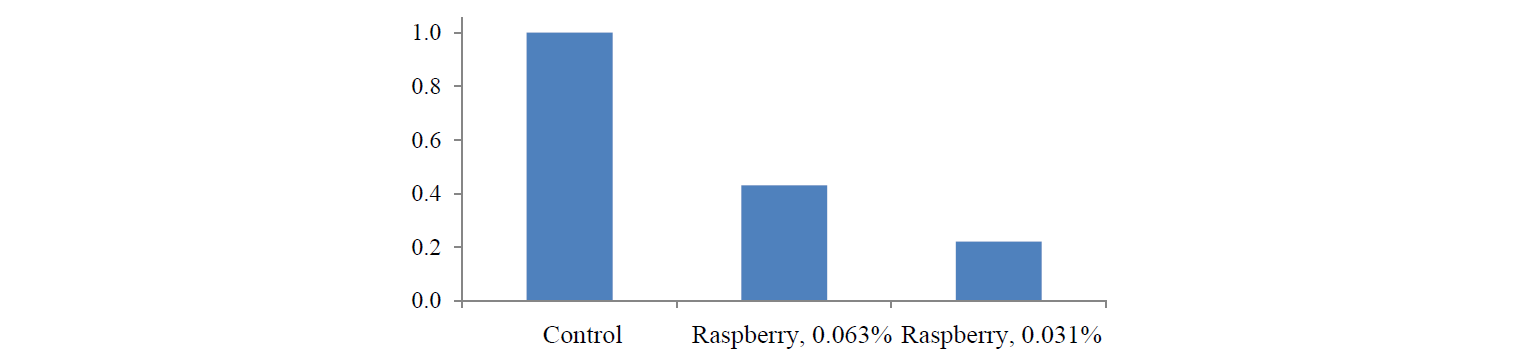
A PCR analysis of COX-2 expression was performed after colon cancer cells of the HCT-116 line had undergone a proper treatment. The analysis showed that the raspberry extract had an inhibitory effect on the expression of this gene. The effect of the extract on COX-2 expression depended on the dose. Figure 3 shows that when the concentration of the extract was 0.063%, COX-2 expression fell down to 43%, i.e. by 2.3 times. When the concentration of the extract was 0.031%, it fell down to 22%, i.e. by 4.5 times.
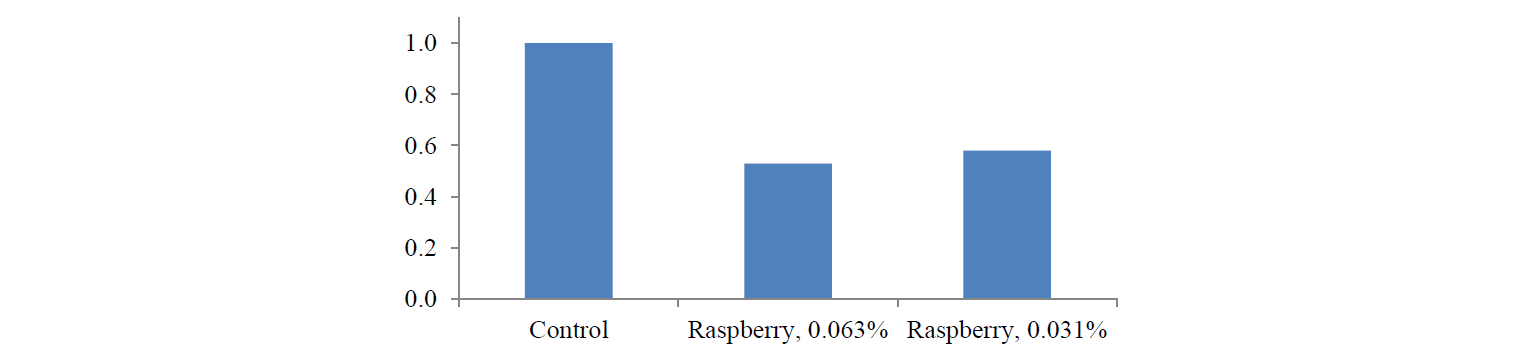
Figure 4 shows some dependencies revealed by the analysis of iNOS expression. When treating the cells with the raspberry extract, both concentrations resulted in a decrease in iNOS expression by almost 2 times: 47% and 42% for concentrations of 0.063% and 0.031 %, respectively.
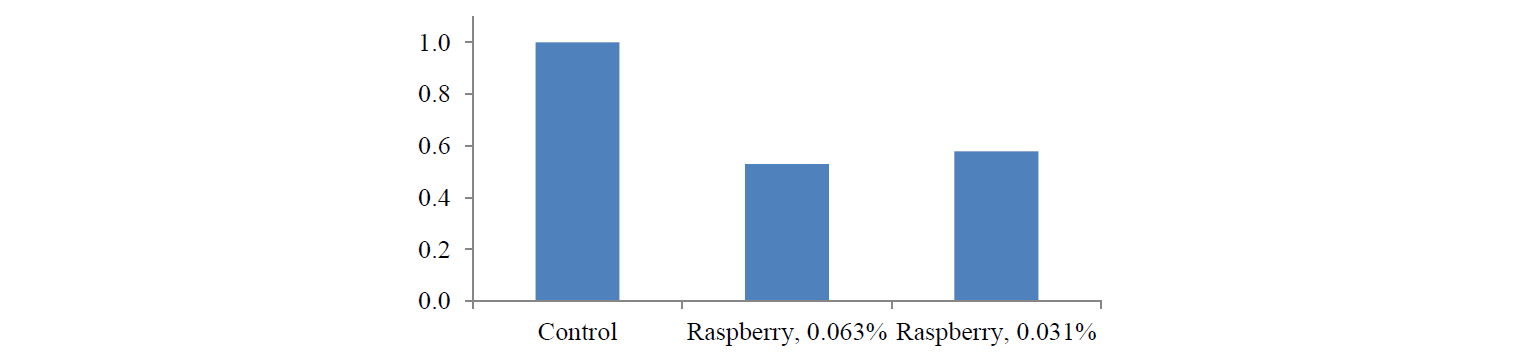
The PCR analysis showed that the raspberry extract also inhibited IL-8 expression. When HCT-116 cells were treated with the raspberry extract at the concentration of 0.063%, it inhibited IL-8 expression by 54%, while the concentration of 0.031% inhibited IL-8 expression by 42%. Figure 5 shows the effect of the raspberry extract on IL-8 expression.
ВЫВОДЫ
The research results made it possible to draw the following conclusions:
(1) US or MW treatment improved the extraction process and increased the content of biologically active cells and their antioxidant properties. US extraction had a greater impact on the content of phenolic substances and flavonoids, whereas the content of anthocyanins remained almost the same after different types of extraction.
(2) The experimental data made it possible to define the optimal technological parameters: ethanol = 50%, US radiation = 35 kHz, temperature = 40 ± 5°C, time = 120 min, raw materials vs. solvent ratio = 1:10.
(3) The study defined the sensory, physical, and chemical quality and safety indicators for raspberry extracts, which did not contradict with the national regulatory documentation.
(4) The raspberry extract was found able to reduce the expression of pro-inflammatory COX-2, iNOS, and IL-8 genes. This semi-finished product can be recommended for further studies of the effect it has on induced COX-2, iNOS, and IL-8 expression, as well as for in vivo studies of its anticarcinogenic activity.
КОНФЛИКТ ИНТЕРЕСОВ
The authors declare that there is no conflict of interest related to this article.
СПИСОК ЛИТЕРАТУРЫ
- Taylor TM. Handbook of Natural Antimicrobials for Food Safety and Quality. USA: Elsevier Science, 2014. 442 p.
- Bobinaite R, Viškelis P, Šarkinas A, Venskutonis PR. Phytochemical composition, antioxidant and antimicrobial properties of rasberry fruit, pulp, and marc extracts. CyTA – Journal of Food. 2013;11(4):334–342. DOI: https://doi.org/10.1080/19476337.2013.766265.
- Wallace CA, Sperber WS, Mortimore SE. Food Safety for the 21st Century: Managing HACCP and Food Safety Throughout the Global Supply Chain. Wiley; 2018. 477 p. DOI: https://doi.org/10.1002/9781444328653.
- Kapetanakou AE, Skandamis PN. Applications of active packaging for increasing microbial stability in foods: natural volatile antimicrobial compounds. Current Opinion in Food Science. 2016;12:1–12. DOI: https://doi.org/10.1016/j.cofs.2016.06.001.
- Boo H-O, Hwang S-J, Bae C-S, Park S-H, Heo B-G, Gorinstein S. Extraction and characterization of some natural plant pigments. Industrial Crops and Products. 2012;40(1):129–135. DOI: https://doi.org/10.1016/j.indcrop.2012.02.042.
- Tian Y, Liimatainen J, Puganen A, Alakomi H-L, Sinkkonen J, Yang B. Sephadex LH-20 fractionation and bioactivities of phenolic compounds from extracts of Finnish berry plants. Food Research International. 2018;113:115–130. DOI: https://doi.org/10.1016/j.foodres.2018.06.041.
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer Journal for Clinicians. 2012;62(1):10–29. DOI: https://doi.org/10.3322/caac.20138.
- Zanini S, Marzotto M, Giovinazzo F, Bassi C, Bellavite P. Effects of Dietary Components on Cancer of the Digestive System. Critical Reviews in Food Science and Nutrition. 2015;55(13):1870–1885. DOI: https://doi.org/10.1080/10408398.2012.732126.
- Kushi LH, Doyle C, McCullough M, Rock CL, Demark-Wahnefried W, Bandera EV, et al. American Cancer Society guidelines on nutrition and physical activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. CA Cancer Journal for Clinicians. 2012;62(1):30–67. DOI: https://doi.org/10.3322/caac.20140.
- Zhao T-T, Jin F, Li J-G, Xu Y-Y, Dong H-T, Liu Q, et al. Dietary isoflavones or isoflavone-rich food intake and breast cancer risk: A meta-analysis of prospective cohort studies. Clinical Nutrition. 2019;38(1):136–145. DOI: https://doi.org/10.1016/j.clnu.2017.12.006.
- Teng H, Fang T, Lin Q, Song H, Liu B, Chen L. Red raspberry and its anthocyanins: Bioactivity beyond antioxidant capacity. Trends in Food Science and Technology. 2017;66:153–165. DOI: https://doi.org/10.1016/j.tifs.2017.05.015
- Golubtsova YuV. Physical and chemical indicators and merchandasing assessment of wild strawberry, gooseberry, cherry, raspberry, banana, wild rose and kiwi. Foods and Raw Materials. 2017;5(1):154–164. DOI: https://doi.org/10.21179/2308-4057-2017-1-154-164.
- Zhidyokhina TV. Industrial assortment of raspberry and its productivity in the black-earth region. The Bulletin of KrasGAU. 2015;109(10):131–135. (In Russ.).
- Miret JA, Munné-Bosch S. Abscisic acid and pyrabactin improve vitamin C contents in raspberries. Food Chemistry.2016;203:216–223 DOI: https://doi.org/10.1016/j.foodchem.2016.02.046.
- Zhang L, Li J, Hogan S, Chung H, Welbaum GE, Zhou K. Inhibitory effect of raspberries on starch digestive enzyme and their antioxidant properties and phenolic composition. Food Chemistry. 2015;119:592–599. DOI: https://doi.org/10.1016/j.foodchem.2009.06.063.
- Landele JM. Ellagitannis, ellagicacid and their derived metabolites: A review about source, metabolism, functions and health. Food Research International. 2011;44(5):1150–1160. DOI: https://doi.org/10.1016/j.foodres.2011.04.027.
- Luchina NA. Technological Properties of Raspberries Grown in the Novosibirsk Region. Food Industry. 2015;(8):22–24. (In Russ.).
- Zhbanova EV. Biochemical characterization of fruits from raspberry vor gene pool under the circumstances of the Central Chernozem Zone (Michurinsk). Collection of scientific works SNBG. 2017;144–1:182–186. (In Russ.).
- Xiao T, Guo Z, Bi X, Zhao Y. Polyphenolic profile as well as anti-oxidant and anti-diabetes effects of extracts from freeze-dried black raspberries. Journal of Functional Foods. 2017;31:179–187. DOI: https://doi.org/10.1016/j.jff.2017.01.038.
- Terletskaya VA, Rubanka EV, Zinchenko IN. Influence of technological factors on the process of black chokebery extraction. Food Processing: Techniques and Technology. 2013;31(4):127–131. (In Russ.).
- Polyakov VA, Abramova IM, Zenina GP, Aristarhova TYu, Stekanova GV. Ripple Effect on the Extraction Process in the Preparation of Fruit Drinks Fortified for Distillery. Beer and beverages. 2016;(6):42–45. (In Russ.).
- Khramtsov AG, Evdokimov IA, Lodygin AD, Budkevich RO. Technology development for the food industry: a conceptual model. Foods and Raw Materials. 2014;2(1):22–26. DOI: https://doi.org/10.12737/4121.
- Meireles MAA. Extracting Bioactive Compounds for Food Products. Theory and Applications. Bova Raton: CRC Press; 2009. 464 p.
- Vongsak B, Sithisarn P, Mangmool S, Thongpraditchote S, Wongkrajang Y, Gritsanapan W. Maximizing total phenolics, total flavonoids contents and antioxidant activity of Moringa oleifera leaf extract by the appropriate extraction method. Industrial Crops and Products. 2013;44:566–571. DOI: https://doi.org/10.1016/j.indcrop.2012.09.021.
- Khoei M, Chekin F. The ultrasound-assisted aqueous extraction of rice bran oil. Food Chemistry. 2016;194:503–507. DOI: https://doi.org/10.1016/j.foodchem.2015.08.068.
- Nipornram S, Tochampa W, Rattanatraiwong P, Singanusong R. Optimization of low power ultrasound-assisted extraction of phenolic compounds from mandarin (Citrus reticulata Blanco cv. Sainampueng) peel. Food Chemistry. 2018;241:338–345. DOI: https://doi.org/10.1016/j.foodchem.2017.08.114.
- Espada-Bellido E, Ferreiro-González M, Carrera C, Palma M, Barroso CG, Barbero GF. Optimization of the ultrasound-assisted extraction of anthocyanins and total phenolic compounds in mulberry (Morus nigra) pulp. Food Chemistry. 2017;219:23–32. DOI: https://doi.org/10.1016/j.foodchem.2016.09.122.
- Markin VI, Cheprasova MIu, Bazarnova NG. Basic directions of use microwave radiation in the processing of plant raw material (review). Chemistry of plant raw material. 2014;(4):21–42. (In Russ.). DOI: https://doi.org/10.14258/jcprm.201404597.
- Cai M, Hou W, Lv Y, Sun P. Behavior and rejection mechanisms of fruit juice phenolic compounds in model solution during nanofiltration. Journal of Food Engineering. 2017;195:97–104. DOI: https://doi.org/10.1016/j.jfoodeng.2016.09.024.
- Demidova AV, Makarova NV. Influence of blanching on the physical and chemical properties and antioxidant activity of fruit raw materials cherries, plums, blank chokeberry, strawberry. Food industry. 2016;(2):40–43. (In Russ.).
- Strycova AD, Makarova NV. Frozen Berries – Effective Antioxidant for Whole Year. Food industry. 2013;(3):28–31. (In Russ.).
- Karabegovic IT, Stojičević SS, Veličković DT, Todorović ZB, Nikolić NT, Lazić ML. The effect of different extraction techniques on the composition and antioxidant activity of cherry laurel (Prunus laurocerasus) leaf and fruit extracts. Industrial Crops and Products. 2014;54:142–148. DOI: https://doi.org/10.1016/j.indcrop.2013.12.047.
- Cheigh C-I, Chung E-Y, Chung M-S. Enhanced extraction of flavanones hesperidin and narirutin from Citrus unshiu peel using subcritical water. Journal of Food Engineering. 2012;110(3):472–477. DOI: https://doi.org/10.1016/j.jfoodeng.2011.12.019.
- M’hiri N, Ioannou I, Mihoubi Boudhrioua N, Ghoul M. Effect of different operating conditions on the extraction of phenolic compounds in orange peel. Food and Bioproducts Processing. 2015;69:161–170. DOI: https://doi.org/10.1016/j.fbp.2015.07.010.
- Yao C-Y, Yang J-Y, Xu Z-L, Wang H, Lei,H-T, Sun Y-M, et al. Indirect Competitive Enzyme-Linked Immunosorbent Assay for Detection of Tylosin in Milk and Water Samples. Chinese Journal of Analytical Chemistry. 2018;46(8):1275–1281. DOI: https://doi.org/10.1016/S1872-2040(18)61106-5.
- Pal V, Saxena A, Singh S, Kumar S, Goel AK. Development of a TaqMan Real-Time Polymerase Chain Reaction Assay for Detection of Burkholderia mallei. Journal of Equine Veterinary Science. 2017;58:58–63. DOI: https://doi.org/10.1016/j.jevs.2017.08.008.
- PCR Primers for Gene Expression Detection and Quantification [Internet]. [cited 2018 Sep 01]. Available from: http://pga.mgh.harvard.edu/primerbank/.
- Luo , Zhang H. The role of proinflammatory pathways in the pathogenesis of colitis-associated colorectal cancer. Mediators of Inflammation. 2017;2017. DOI: https://doi.org/10.1155/2017/5126048.
- Nile SH, Ko EY, Kim DH, Keum Y-S. Screening of ferulic acid related compounds as inhibitors of xanthine oxidase and cyclooxygenase-2 with anti-inflammatory activity. Revista Brasileira de Farmacognosia. 2016;26(1):50–55. DOI: https://doi.org/10.1016/j.bjp.2015.08.013.
- Rennert K, Steinborn S, Gröger M, Ungerböck B, Jank A-M, Ehgartner J, et al. A microfluidically perfused three dimensional human liver model. Biomaterials. 2015;71:119–131. DOI: https://doi.org/10.1016/j.biomaterials.2015.08.043.
- Paccaud JP, Schifferli JA, Baggiolini M. NAP-1/IL-8 induces up-regulation of CR1 receptors in human neutrophil leukocytes. Biochemical and Biophysical Research Communications. 1990;166(1):187–192. DOI: https://doi.org/10.1016/0006-291X(90)91929-M.
- Souza GR, Cunha TM, Silva RL, Lotufo CM, Verri WA, Funez MI, et al. Involvement of nuclear factor kappa B in the maintenance of persistent inflammatory hypernociception. Pharmacology, Biochemistry and Behavior. 2015;134:49–56. DOI: https://doi.org/10.1016/j.pbb.2015.04.005.
- Alfaro C, Sanmamed MF, Rodríguez-Ruiz ME, Teijeira Á, Onate C, González Á, et al. Interleukin-8 in cancer pathogenesis, treatment and follow-up. Cancer Treatment Reviews. 2017;60:24–31. DOI: https://doi.org/10.1016/j.ctrv.2017.08.004.
- Vannini F, Kashfi K, Nath N. The dual role of iNOS in cancer. Redox Biology. 2015;6:334–343. DOI: https://doi.org/10.1016/j.redox.2015.08.009.