Аннотация
Many cheese manufacturers still have not utilized cheese whey that damages to the environment as it is directly been drained into waters. Cheese whey can be used as active packaging material to prolong the shelf-life of food products. Fermented cheese whey contains bioactive peptides which are able to improve the functional properties of cheese whey as an antimicrobial agent. The combination of cheese whey with polysaccharides, lipid, and other additional ingredients can improve the physical characteristics of the active packaging in the form of edible film. Around 20-45% of plasticizer will expose the film formed. Cheese whey with agro-industrial waste starch-based formulation can be used as an alternative way to produce an antimicrobial edible film as an active packaging. The film has shown acceptable physical characteristics and high antimicrobial activity, which makes it possible to extend the shelf life of food products. An advanced process, for example, the use of transglutaminase enzyme and Candida tropicalis mutant, is also effective. The result of that is the formation of the essential compound which can improve the active packaging quality. The utilisation of cheese whey and agro-industrial waste based on starch contributes significantly to the environmental conservation.Ключевые слова
Whey, protein, shelf-life, packaging, antimicrobial, edible film, fermentation, environmentВВЕДЕНИЕ
Cheese production process has significant impact on the environment. One of damaging factor is the disposal of cheese by-product. Cheese making process produces large amounts of by-product called cheese whey, which is almost 90% of used milk [1]. It implies if one batch of cheese production uses 100 L of milk, 80–90 L of cheese whey will be produced [2]. Although it is wasted, 30% of cheese whey still has been utilised as animal feed and fertiliser, while the rest has thrown away to the rivers or seas [3]. Cheese whey is able to damage them environment due to its characteristics. Cheese whey has high biochemical oxygen demand (BOD) and chemical oxygen demand (COD), which is more then 35000 and 60000 ppm, respectively [4]. Thus, 4000 L of whey from the cheese industry can damage the environment to the same extent as faecal waste from 1900 humans [5].
On the other hand, whey has a valuable chemical composition and contains 55% of total nutrients in milk [6]. Whey contains (w/w): 93.7% of water, 0.1–0.5% fat, 0.8% protein, 4.9% lactose, 0.5–0.8% ash, and 0.1–0.4% lactic acid [2]. Functionally, the beneficial effect of whey on the human health is due to immunoglobulin and glycoprotein, such as lactoferrin and transerin, as well as enzymes – lysozyme and lactoperoxidase. All of these components contribute to human immunity and have an antimicrobial activity against allergy reaction [7, 8].
Cheese whey has been utilised in various ways. About 70% of whey is processed into whey powder that can be used in pastry, ice cream, sweets, glazes, sugar dressing, jams, and melted cheese industry [3, 9]. Whey is used as a food ingredient because of its gelling, emulsifying, antimicrobial properties, good solubility, viscosity, nutritional value, as well as the ability to reduce allergenicity [2, 10]. Unfortunately, it is difficult to utilise whey for cheese manufactures [11]. One of the causes is high cost drying process of whey. Therefore, the search of alternative whey processing is of great interest.
Organic compounds of whey are a potential biomass to be utilised as bio-energy. Bioethanol can be made from whey through fermentation by using Kluyveromyces fragilis var marxianus [11–13]. Lactose, whose content in whey is 4.5–5%, acts as a carbon source for ethanol fermentation. The fermentation also can result in various bioproducts, such as ethanol, biogas (methane), organic acids (acetic, propionic, lactic, citric, and gluconic), amino acids (glutamic, lysine, and threonine), vitamins (B12 and B2), polysaccharides (gum, dextran, and gellan), lipids, enzymes (polygalacturonase), and others (calcium magnesium acetate, butanol, and glycerol) [11].
The fermentation of whey leads to other compounds which have high functional use, such as bioactive peptides. Native whey has minor bioactive compounds such as lysozyme, lactoperoxidase and lactoferrin that are reported to have antimicrobial activity towards pathogenic bacteria [7]. Fermentation causes protein hydrolysis – by a microorganism which releases bioactive peptides from protein molecules or by a digestive enzyme, such as proteases [14, 15].
Cheese whey can also be utilised as biodegradable packaging material, such as edible film. Such a film is safe to consumers and environmentally friendly. It is also expected to extend the shelf life of food products because it protects them from gases, such as oxygen, carbon dioxide, and ethylene, as well as from water loss [16–18].
The use of the edible film as a food packaging material is expected to reduce plastic waste. Annually, Indonesia produces 3.22 million metric tons of plastic waste. It is the second largest plastic waster after China that produces 8.82 million metric tons. The use of plastic has rapidly increased since the development of plastic commercialisation in the 1930s and 1940s. It reached 288 million metric tons of global plastic resin production in 2012 [19].
The high rate of population growth has caused an increase in food demand. This has resulted in an increasing use of plastic, which contributes to the economic benefits [20, 21]. Food industry still widely uses non-degradable plastic as a food packaging material. However, non-degradable plastic, such as polyethylene (PE), has an immensely slow degradation time under natural environmental conditions [22–24]. Thus, food industry indirectly affects the environment.
The utilisation of cheese whey to produce bioproducts, such as edible films, would be a potential course of action to protect the environment. Cheese whey fermentation can be applied to enhance the antimicrobial effect and the packaging ability of the edible film produced in order to extend the shelf life of packaged food. The aim of this paper was to review the potential of fermented cheese whey in the produce of edible films and active packaging systems.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
The paper was written with non-research methodology based on literature reviews from various sources.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
Fermented cheese whey. The protein content in milk is 3.5% which is, in turn, composed of 80% of casein (α-, β-, and k-caseins) and 20% of whey proteins (β-lactoglobulin, α-lactalbumin, and others). Several proteins in milk have an antimicrobial effect as shown in Table 1 [31, 33–38]. Whey contains biological active substances, such as enzymes, trace elements, and immunoglobulins which contribute to the good health [25].
Fermented whey is known to exert antimicrobial properties. For example, Bacillus licheniformis can produce 3200 AU/mL of bacteriocins from 70 g/L of cheese whey. This amount can count over 4000 AU/mL if cheese whey increases up to 120.4 g/L (with initial pH of 7 and incubation temperature of 26–37°C), [26]. Bacteriocins are a bacterial peptides which are able to inhibit or kill microorganisms [27]. Other lactic acid bacteria, such as Lactococcus lactis, Lactobacillus casei, and Leuconostoc mesenteroides, also can produce bacteriocins in response to whey fermentation [28–30].
Besides bacteriocins, fermented whey contains bioactive peptides which also have antimicrobial activity. Bioactive peptides defined as inactive fragments of precursor protein sequences. Proteolytic enzymes can release the fragments, and they can interact with selected receptors and regulate the body’s physiological function [31]. Table 2 demonstrates bioactive peptides contained in whey protein [50–55]. β-lactolobulin, α-lactoalbumin, immunoglobulin, bovine serum albumin, bovine lactoferrin, lactoperoxidase and minor proteinaceous, such as glycomacropeptide, are released from k-casein during enzymatic cheese making [32].
Along with antimicrobial effect, whey bioactive peptides act as immunomodulatory agents that regulate cell-mediated and humoral immune functions [31]. In addition, bioactive peptides inhibit angiotensinconverting enzyme (ACE) that splits angiotensin I to angiotensin II, an active peptide hormone. These peptides are able to inhibit ACE and control the increase in blood pressure [39]. Opioid peptides influence the central or peripheral nervous system that involved in hypotension, reduced appetite, fluctuating body temperature and alteration of sexual behaviour [40, 41]. Also, peptides with antioxidant activity which can protect the cell from free radicals has been detected [42].
Bioactive peptides can be released in three ways: gastrointestinal digestion (in vivo), fermentation (in vivo), and hydrolysis (in vitro) [31]. Release of bioactive peptides in gastrointestinal tract is the result of enzymatic action. The enzymes are pepsin, trypsin, or chymotrypsin. Pepsin, which is produced from pepsinogen by hydrochloric acid (HCl), converted protein to peptides and amino acids [43]. Other enzymes, such as alcalase and thermolysin, can also stimulate gastrointestinal digestion to produce ACE inhibitory peptides, as well as anti-bacterial, anti-oxidative, immunomodulatory, and opioid peptides [44–49].
Other ways to produce bioactive peptides are microbial fermentation and hydrolysis. For microbial fermentation, such LAB as Lactococcus lactis and Lactobacillus helveticus are used. Microbes will use distinct intracellular peptidases including endopeptidases, amino-peptidase, di-peptidase, and tri-peptidase [33]. Hydrolysis of protein molecules can is performed by proteinases, which leads to the release of bioactive peptides. Proteinases are obtained from the secretion of the bacterial and fungal sources [31].
Bioactive peptides produced from fermented whey can be purified to enhance their functional activities. Stepwise filtration can be carried out to extract bioactive peptides after fermentation. Afterwards, the extract is centrifuged at 7000 rpm for 10 min in a refrigerated centrifuge to obtain supernatant. The supernatant is filtered through 0.45 μm and then through 0.22 μm syringe filters. Ultrafiltrate of bioactive peptides of 10 kDa and 5 kDa in size can be obtained after passing through the 10 kDa and 5 kDa MWCO membranes [56].
Edible film production. Food products usually have a short shelf-life. In order to prolong it, most of manufacturers uses food packaging. A packaging system should protect the product from contamination during handling, storage, and sale until it reaches retailers and consumers [57]. Non-degradable packaging still has widely applied by food industry. The US Environmental Protection Agency (EPA) reports that 31% of municipal solid waste (MSW) is packaging waste [58]. Edible films as a packaging material can be an effective solution of reducing waste because of their degradable characteristics [57].
The ideal edible film has high water holding ability; controls gas exchanges; inhibits solute transport, organic vapour transfers, as well as oil and fat migration; improves mechanical properties of food to simplify handling and carriage; has neutral sensory characteristics, improving sensory properties of food products [57]. Edible films should consist of components produced mainly from edible biopolymers and food grade additives. The additives should meet safety requirements to food additives and to be at least GRAS (Generally Recognized as Safe) [59].
There are two ways to create an edible film: wet process and dry process. Wet process applies dispersion in a solvent such as water, alcohol, or mixture of water and alcohol, or other solvents. The film-forming solution is then casted and dried to obtain films. The dry process does not require any solvent. It can be produced by compression, molding, or extrusion [60, 61]. The film-making and coating processes include melting and solidification of solid fats, waxes, and resins; conservation of hydrocolloid; complex conservation of two hydrocolloids; and thermal gelation or coagulation by heating [57]. Therefore, the dry process usually needs more equipment, which results in higher cost compared to the wet process.
In edible film production, the incorporation of a certain additive is possible to form an advanced system called active packaging [62]. The additive compound enhances shelf-life and stability of the product, as well as improves its microbiological safety and sensory attributes [63]. The following additives can be used in edible films: flavouring agents, spices, antimicrobial substances, antioxidants, pigments, light absorbers, salts, etc. Antioxidants and antimicrobial additives are commonly used in order to prevent spoilage and thus enhance safety. Antimicrobial agents, being used in active packaging, can overcome the hurdles of uncontrolled migration and interaction of an active compound of various natural antioxidants used directly in food [64, 65].
Comparison of characteristics from various film bases. Edible film or even active packaging usually use polysaccharide, protein, lipid, or composite base to make a film forming solution. Thus, fermented whey can be one of multifunctional ingredients and act as a filmforming base and an antimicrobial agent.
Researchers have focused on the use of composite based films to explore the complement advantages of each component [64, 66]. A composite based film can be both one-layer and multiple-layer. The matrix of hydrophilic and hydrophobic lipid, which is called bi-layer composite system, has better functional characteristics than pure hydrocolloid films. However, one of disadvantages of bi-layers composite systems is longer preparation process. It requires two casting and two drying stages, which has made these laminated films less popular in food industry [67]. In order to enhance holding properties of active packaging, scientists have studied its mechanical properties (Table 3). These are transparency, oxygen permeability, carbon dioxide permeability, water vapor permeability, emulsion stability, and glass transition temperature.
Generally, lipid films have the less structural integrity compared to protein or polysaccharide films [68]. The use of lipids in edible films has resulted in heterogenous film structure that has an impact on discontinuities in the polymer and production of a strong emulsion matrix [69]. A composite film based on polysaccharide has the greatest mechanical properties, which allows its using in gastronomy. Along with protein added, polysaccharide film is an optimal active packaging.
Besides the film based component, the composition of the edible film is also an important factor. Plasticizer is one of substantial components to create a flexible film by reducing interaction between intermolecular starch [70]. Examples of plasticizer are polyol groups such as glycerol, xylitol, sorbitol, mannitol, and sucrose [18, 70]. Xanthan gum and carrageenan are also promising plasticizers which provide the product with strength and durability with great sensory properties. They demonstrate high stabilizing ability and resistance to water, [71]. The plasticizer is able to reduce intermolecular bonds between amylose, amylopectin, and amylose-amylopectin in the starch matrix and replace them with hydrogen bonds between plasticizer and starch. This reduces brittle and enhances flexibility [68].
The flexibility of the film depends on the concentration of the plasticizer in fthe ilm-forming solution. High or low concentrations would result in anti-plasticization. For example, glycerol in the amount of over 30% used in the starch-based film is the case. It will result in a decrease in such characteristics as elongation at break. It was established that strong interaction between plasticizer and other molecules blocked the macromolecular mobility [70, 81]. On the other hand, if the plasticizer concentration is too low, the film formed will be brittle and hard to handle. Generally, the optimal concentration of plasticizer is 20–45% [82].
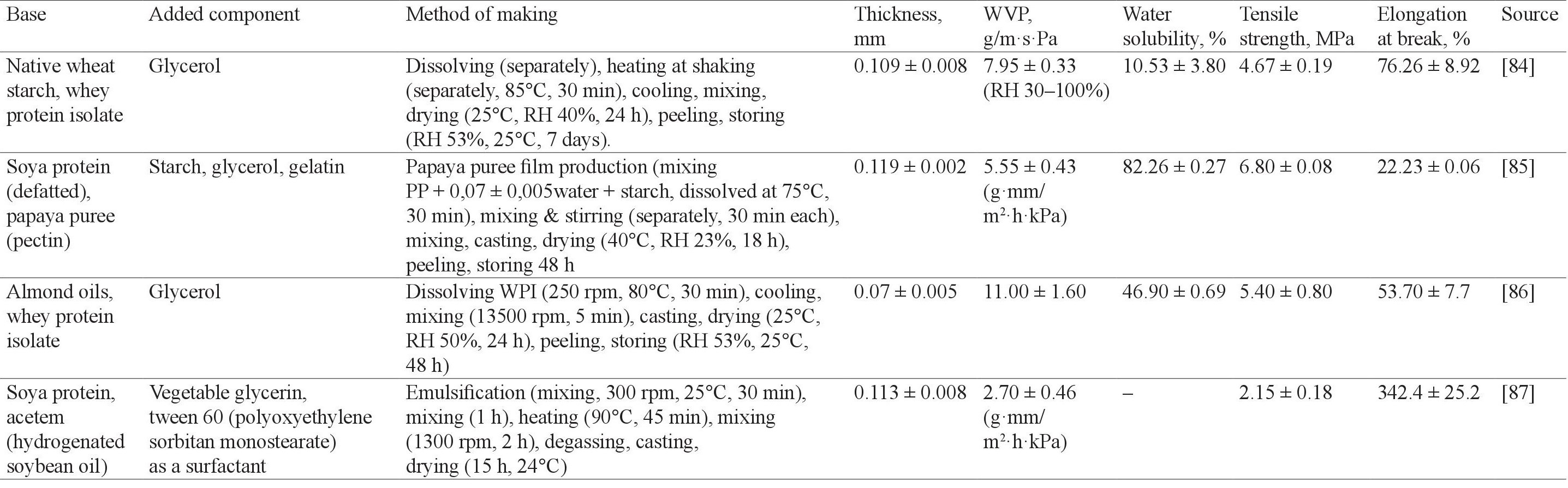
The potential of fermented whey for active packaging composite. The composite of edible film can be made to complement each single material-based film characteristics. Protein is usually used as one of material-based because of its nutritional value [83]. The comparison of the composite edible film using proteinbased is shown in Table 4. From all of the sources of protein, soya and whey have been mostly used.
As a food barrier capability, the addition of oil has resulted in a lower WVP, which is showed in a comparison of soya protein with oil and with pectin. Thereby, we can conclude that a composite protein film with oil has better barrier properties. However, the water solubility of whey protein with starch is higher than that of soya protein with pectin. In terms of mechanical properties, whey protein with starch provides a better result than soy protein with pectin. However, some other parameters cannot be compared because of the difference between the film production and the analysis method. Based on the description above, it is possible to conclude that the whey protein is more effective than soya protein to produce the edible film.
Functional characteristics of fermented whey make it promising raw material for active packaging. Bioactive peptides from fermented whey have had their ability to act as an antimicrobial agent; immunomodulatory peptides regulate cell-mediated and humoral immune functions; ACE inhibitory peptides lower blood pressure; and opioid peptides are effective against hypotension, lack of appetite, etc., as well as exhibit antioxidant properties, protecting cells from free radicals [31].
Several bioactive peptides derived from whey protein are also known for its capability to enhance the defence towards various pathogenic bacteria and yeast. Their antimicrobial activity can inhibit the growth of such microorganisms effectively. The incorporation of bioactive peptides into film is more effective way to lower the concentration of microorganisms than direct using them in food. Thus, it allow avoiding unwanted flavours and odor of food [88].
In terms of the characteristics, a good characteristic can be achieved if the protein contained in the cheese whey is mixed with starch that contained polysaccharides. With the addition of plasticizer, this composite based film will have good mechanical properties as well as the barrier ability to prolong food shelf-life.
Advanced process to create composite fermented cheese whey film. Nowadays, an advanced process to create an edible composite is based film-forming solution. The film with cellulose, whey and sunflower oil based are stirred with Ultraturrax homogenizer at 11000 rpm for 3 min to decrease the droplet sizes of oil. As a result, the smaller droplet sizes can disperse uniformly in the hydrocolloid matrix. Therefore, the penetration of water into film will be harder, which will result in better water vapour transfer. The combination of the degassed method under vacuum (80 kPa) and a vacuum pump for 5 min will result in the film production with tensile strength of 8.59 MPa, elongation at break 35.94%, WVP 3.211 g/m·s·Pa, and transparency of 3,637 % [79].
There are several methods available to increase the stability and the quality of characteristics of active packaging. The addition of enzyme is one of methods to enhance the film quality in the complex edible film. The presence of Transglutaminase (TGase) has caused enzymatic cross-linking in P/P soluble electrostatic aggregates. Thus, TGase can strongly produce composite bioplastics by escalating the mechanical and barrier characteristics. Supramolecular structure of P/P complex as enzyme substrate is crucially influencing pH of a film-forming solution. With the addition of TGase, film characteristics at pH (pH complexation around 3.25–5.5, when soluble P/P complexes occur) create better characteristics than higher pH. The pH can significantly increase tensile strength and elongation at break, and reduce Young’s modulus and WVP [89].
For the usage of fermented cheese whey in the active packaging, there is an advanced process that can be added in the fermentation process. Candida sp. is one of the yeast that has already found in Serro Minas, a cheese from Brazil [90]. Recently, a study for identifying the indigenous yeast that contained in homemade mozzarella whey has also found that Candida sp. contained in mozzarella whey and Greek fermented whey [91]. It means that Candida sp. is naturally contained in cheese whey and can live to ferment the whey [92]. Candida spp. is also known as the most massive yeasts to produce xylitol with 63–70% w/w yields. According to several studies, C. tropicalis mutant maximises the xylitol production, reaching 100% yields [93]. It is known that xylitol is one of sugar alcohol that can be utilized in the film production as a plasticizer [18]. Fermented cheese whey can act as an antimicrobial agent and natural plasticizer.
Prospects of the use of edible films and active packaging with cheese whey. The edible film can be an effective solution to reduce plastic waste of food packaging. Addition of several antimicrobials can also be used to prolong the shelf life to reach a proper packaging system which is similar to the plastic packaging. Thus, fermented cheese whey as a base ingredient of composite film system is able to meet this requirement. Despite some disadvantages of protein, its combination with other ingredients make is possible to obtain an excellent film with required characteristics.
Besides various modification of film manufacture, the cost in creating edible film must be taken into account. The edible film should be cost-effective compared to plastic, paper, or any other packaging that can harm the environment. Thus, advancing the edible film production is important to make film characteristics as high as characteristics of plastic packaging.
The simplified process of cheese whey fermentation using indigenous yeast can also increase the antimicrobial properties of the fermented cheese whey. In the future, advancing film manufacture process from fermented cheese whey can be one of massive ways to create modern environmentally-friendly packaging.
ВЫВОДЫ
Cheese whey, a by-product of cheese-making process, has several functional effects, including inedible film formation. Bioactive peptides contained in native cheese whey can be enhanced by fermentation to generate high antimicrobial activity. In addition, a composite edible film can be produced from fermented whey and starch to gain good mechanical characteristics as well as a good barrier to prolong food shelf-life. The utilisation of fermented cheese whey as an edible film material allows obtaining an active packaging system with high antimicrobial activity.
КОНФЛИКТ ИНТЕРЕСОВ
The authors declare no conflict of interest.
БЛАГОДАРНОСТИ
Authors thank the Ministry of Research, Technology and Higher Education of The Republic of Indonesia that funded the research through ‘Penelitian Tesis Magister’, 2019. This article’s publication is supported by the United States Agency for International Development (USAID) through the Sustainable Higher Education Research Alliance (SHERA) Program for Universitas Indonesia’s Scientific Modeling, Application, Research, and Training for City-centered Innovation and Technology (SMART CITY) Project, Grant #AID-497-A-1600004, Sub Grant #IIE-00000078-UI-1.
СПИСОК ЛИТЕРАТУРЫ
- Utama GL, Kurnani TBA, Sunardic, Balia RL. Reducing cheese-making by-product disposal through ethanol fermentation and the utilization of distillery waste for fertilizer. International Journal of GEOMATE. 2017;13(37):103–107. DOI: https://doi.org/10.21660/2017.37.2737.
- Božanic R, Barukcic I, Jakopovic LK, Tratnik L. Possibilities of Whey Utilisation. Austin Journal of Nutrition and Food Sciences. 2014;2(7):1036–1042.
- Jelen P. Whey Processing. Utilization and Products. In: Roginski H, editor. Encyclopedia of Dairy Science. Academic Press; 2002. pp. 2739–2745. DOI: https://doi.org/10.1016/B0-12-227235-8/00511-3.
- Smithers GW. Whey and whey proteins-From ‘gutter-to-gold’. International Dairy Journal. 2008;18(7):695–704. DOI: https://doi.org/10.1016/j.idairyj.2008.03.008.
- Tunick MH. Whey Protein Production and Utilization: A Brief History. In: Onwulata CI, Huth PJ, editors. Whey Processing, Functionality and Health Benefits. John Wiley & Sons; 2009. pp. 1–13. DOI: https://doi.org/10.1002/9780813803845.ch1.
- Andrade RP, Melo CN, Genisheva Z, Schwan RF, Duarte WF. Yeasts from Canastra cheese production process: Isolation and evaluation of their potential for cheese whey fermentation. Food Research International. 2017;91:72–79. DOI: https://doi.org/10.1016/j.foodres.2016.11.032.
- Maruddin F, Malaka R, Hajrawati, Taufik M. Antibacterial Activity of Fermented Whey Beverage by Products from Buffalo Dangke. Materials of the Buffalo International Conference 2013; 2013; Makassar. Makassar: University of Hasanuddin; 2013. p. 329–334.
- Balia RL, Fleet GH. Growth of Yeasts Isolated from Cheeses on Organic Acids in the Presence of Sodium Chloride. Food Technology and Biotechnology. 1999;37(2):73–79.
- Caric M. Technology and Milk Products, Dried and Concentrated. Beograd: Naucna knjiga;1990.
- Tratnik L. The role of whey in functional dairy food production. Mljekarstvo: journal for dairy production and processing improvement. 2003;53(4):325–352.
- Guimarães PMR, Teixeira JA, Domingues L. Fermentation of lactose to bio-ethanol by yeasts as part of integrated solutions for the valorisation of cheese whey. Biotechnology Advances. 2010;28(3):375–384. DOI: https://doi.org/10.1016/j.biotechadv.2010.02.002.
- González Siso MI. The biotechnological utilization of cheese whey: A review. Bioresource Technology. 1996;57(1):1–11. DOI: https://doi.org/10.1016/0960-8524(96)00036-3.
- Pesta G, Meyer-Pittroff R, Russ W. Ulitization of Whey. In: Oreopoulou V, Russ W, editors. Utilization of By-Products and Treatment of Waste in the Food Industry. Boston Springer; 2007. pp. 193–207. DOI: https://doi.org/10.1007/978-0-387-35766-9_10.
- Hayes M, Ross RP, Fitzgerald GF, Hill C, Stanton C. Casein-derived antimicrobial peptides generated by Lactobacillus acidophilus DPC6026. Applied and Environmental Microbiology. 2006;72(3):2260–2264. DOI: https://doi.org/10.1128/AEM.72.3.2260-2264.2006.
- Clare DA, Swaisgood HE. Bioactive milk peptides: A prospectus. Journal of Dairy Science. 2000;83(6):1187–1195. DOI: https://doi.org/10.3168/jds.S0022-0302(00)74983-6.
- Sánchez-Ortega I, García-Almendárez BE, Santos-López EM, Amaro-Reyes A, Barboza-Corona JE, Regalado C. Antimicrobial Edible Films and Coatings for Meat and Meat Products Preservation. Scientific World Journal. 2014;2014. DOI: https://doi.org/10.1155/2014/248935.
- Zerihun M, Worku T, Sakkalkar SR. Development and Characterization of Antimicrobial Packaging Films. Journal of Ready to Eat Food. 2016;3(2):13–24.
- Akhtar J, Omre PK, Ahmad Azad ZRA. Edible Coating for Preservation of Perishable Foods: A Review. Journal of Ready to Eat Food. 2015;2(3):81–88.
- Jambeck JR, Geyer R, Wilcox C, Siegler TR, Perryman M, Andrady A, et al. Plastic waste inputs from land into the ocean. Science. 2015;347(6223):768–771. DOI: https://doi.org/10.1126/science.1260352.
- Godfray HCJ, Beddington JR, Crute IR, Haddad L, Lawrence, Muir JF, et al. Food security: the challenge of feeding 9 billion people. Science. 2010;327(5967):812–818. DOI: https://doi.org/10.1126/science.1185383.
- Lamont WJ. Plastics: Modifying the microclimate for the production of vegetable crops. HortTechnology. 2005;15(3):477–481.
- Krueger MC, Harms H, Schlosser D. Prospects for microbiological solutions to environmental pollution with plastics. Applied Microbiology and Biotechnology. 2015;99(21):8857–8874. DOI: https://doi.org/10.1007/s00253-015-6879-4.
- Restrepo-Flórez J-M, Bassi A, Thompson MR. Microbial degradation and deterioration of polyethylene – A review. International Biodeterioration and Biodegradation. 2014;88:83–90. DOI: https://doi.org/10.1016/j.ibiod.2013.12.014.
- Steinmetz Z, Wollmann C, Schaefer M, Buchmann C, David J, Tröger J, et al. Plastic mulching in agriculture. Trading short-term agronomic benefits for long-term soil degradation? Science of the Total Environment. 2016;550:690–705. DOI: https://doi.org/10.1016/j.scitotenv.2016.01.153.
- Kitts DD, Weiler K. Bioactive proteins and peptides from food sources. Applications of bioprocesses used in isolation and recovery. Current Pharmaceutical Design. 2003;9(16):1309–1323. DOI: https://doi.org/10.2174/1381612033454883.
- Cladera-Olivera F, Caron GR, Brandelli A. Bacteriocin-like substance production by Bacillus licheniformis strain P40. Letters in Applied Microbiology. 2004;38(4):251–256. DOI: https://doi.org/10.1111/j.1472-765X.2004.01478.x.
- Klaenhammer TR. Bacteriocins of lactic acid bacteria. Biochimie. 1988;70(3):337–349. DOI: https://doi.org/10.1016/0300-9084(88)90206-4.
- De Vuyst L. Nutritional factors affecting nisin production by Lactococcus lactis subsp. actis NIZO 22186 in a synthetic medium. Journal of Applied Bacteriology. 1995;78(1):28–33. DOI: https://doi.org/10.1111/j.1365-2672.1995. tb01669.x.
- Vignolo GM, de Kairuz MN, de Ruiz Holgado AAP, Oliver G. Influence of growth conditions on the production of lactocin 705, a bacteriocin produced by Lactobacillus casei CRL 705. Journal of Applied Bacteriology. 1995;78(1):5–10. DOI: https://doi.org/10.1111/j.1365-2672.1995.tb01665.x.
- Krier F, Revol-Junelles AM, Germain P. Influence of temperature and pH on production of two bacteriocins by Leuconostoc mesenteroides subsp. mesenteroides FR52 during batch fermentation. Applied Microbiology and Biotechnology. 1998;50(3):359–363. DOI: https://doi.org/10.1007/s002530051304.
- Mohanty DP, Mohapatra S, Misra S, Sahu PS. Milk derived bioactive peptides and their impact on human health - A review. Saudi Journal of Biological Sciences. 2016;23(5):577–583. DOI: https://doi.org/10.1016/j.sjbs.2015.06.005,
- Madureira AR, Pereira CI, Gomes AMP, Pintado ME, Xavier Malcata F. Bovine whey proteins – Overview on their main biological properties. Food Research International. 2007;40(10):1197–1211. DOI: https://doi.org/10.1016/j.foodres.2007.07.005.
- Mohanty D, Jena R, Choudhury PK, Pattnaik R, Mohapatra S, Saini MR. Milk Derived Antimicrobial Bioactive Peptides: A Review. International Journal of Food Properties. 2016;19(4):837–846. DOI: https://doi.org/10.1080/10942912.2015.1048356.
- Ibrahim HR, Matsuzaki T, Aoki T. Genetic evidence that antibacterial activity of lysozyme is independent of itscatalytic function. FEBS Letters. 2001;506(1):27–32. DOI: https://doi.org/10.1016/S0014-5793(01)02872-1.
- Vorland LH, Ulvatne H, Rekdal O, Svendsen JS. Initial binding sites of antimicrobial peptides in Staphylococcus aureus and Escherichia coli. Scandinavian Journal of Infectious Diseases. 1999;31(5):467–473. DOI: https://doi.org/10.1080/00365549950163987.
- Recio I, Visser S. Two ion-exchange chromatographic methods for the isolation of antibacterial peptides from lactoferrin: In situ enzymatic hydrolysis on an ion-exchange membrane. Journal of Chromatography A. 1999;831(2):191–201. DOI: https://doi.org/10.1016/S0021-9673(98)00950-9.
- Hoek KS, Milne JM, Grieve PA, Dionysius DA, Smith R. Antibacterial activity in bovine lactoferrin-derived peptides. Antimicrobial Agents and Chemotherapy. 1997;41(1):54–59.
- Van Der Kraan MIA, Groenink J, Nazmi K, Veerman ECI, Bolscher JGM, Nieuw Amerongen AV. Lactoferrampin: a novel antimicrobial peptide in the N1-domain of bovine lactoferrin. Peptides. 2004;25(2):177–183. DOI: https://doi.org/10.1016/j.peptides.2003.12.006.
- Korhonen H, Pihlanto A. Technological options for the production of health-promoting proteins and peptides derived from milk and colostrum. Current Pharmaceutical Design. 2007;13(8):829–843. DOI: https://doi.org/10.2174/138161207780363112.
- Molina PE, Abumrad NN. Metabolic effects of opiates and opioid peptides. Advances in Neuroimmunology. 1994;4(2):105–116. DOI: https://doi.org/10.1016/S0960-5428(05)80005-1.
- Dziuba J, Minkiewicz P, Nałȩcz D, Iwaniak A. Database of biologically active peptide sequences. Nahrung – Food.1999;43(3):190–195. DOI: https://doi.org/10.1002/(SICI)1521-3803(19990601)43:3<190::AIDFOOD190> 3.0.CO;2-A.
- Abuja PM, Albertini R. Methods for monitoring oxidative stress, lipid peroxidation and oxidation resistance of lipoproteins. Clinica Chimica Acta. 2001;306(1–2):1–17. DOI: https://doi.org/10.1016/S0009-8981(01)00393-X.
- Korhonen H, Pihlanto A. Food-derived bioactive peptides – opportunities for designing future foods. Current Pharmaceutical Design. 2003;9(16):1297–1308. DOI: https://doi.org/10.2174/1381612033454892.
- Vermeirssen V, Van Camp J, Verstraete W. Bioavailability of angiotensin I converting enzyme inhibitory peptides. British Journal of Nutrition. 2004;92(3):357–366. DOI: https://doi.org/10.1079/BJN20041189.
- Mohanty DP, Tripathy P, Mohapatra S, Samantaray DP. Bioactive potential assessment of antibacterial peptide produced by Lactobacillus isolated from milk and milk products. International Journal of Current Microbiology and Applied Sciences. 2014;3(6):72–80.
- Suetsuna K, Ukeda H, Ochi H. Isolation and characterization of free radical scavenging activities peptides derived from casein. Journal of Nutritional Biochemistry. 2000;11(3):128–131. DOI: https://doi.org/10.1016/S0955-2863(99)00083-2.
- Rival SG, Boeriu CG, Wichers HJ. Caseins and Casein Hydrolysates. Antioxidative Properties Peroral Calcium Dosage of Infants. Acta Medica Scandinavica. 2001;55:247–255.
- Gauthier SF, Pouliot Y, Maubois J-L. Growth factors from bovine milk and colostrum: composition, extraction and biological activities. Lait. 2006;86(2):99–125. DOI: https://doi.org/10.1051/lait:2005048.
- Pihlanto-Leppälä A, Koskinen P, Paakkari I, Tupasela T, Korhonen HJT. Opioid whey protein peptides obtained by membrane filtration. IDF Bulletin. 1996;311:36–38.
- Shah NP. Effects of milk-derived bioactives: An overview. British Journal of Nutrition. 2000;84:S3–S10.
- Korhonen H, Pihlanto-Leppälä A, Rantamäki P, Tupasela T. The functional and biological properties of whey proteins: prospects for the development of functional foods. Agricultural and Food Science in Finland. 1998;7(2):283–296.
- Park YW, Nam MS. Bioactive Peptides in Milk and Dairy Products: A Review. Korean Journal for Food Science of Animal Resources. 2015;35(6):831–840. DOI: https://doi.org/10.5851/kosfa.2015.35.6.831.
- Lucarini M. Bioactive Peptides in Milk: From Encrypted Sequences to Nutraceutical Aspects. Beverages. 2017;3(3):41–50. DOI: https://doi.org/10.3390/beverages3030041.
- Beucher S, Levenez F, Yvon M, Corring T. Effect of caseinomacropeptide (CMP) on cholecystokinin (CCK) release in rat. Reproduction Nutrition Development. 1994;34(6):613–614. DOI: https://doi.org/10.1051/rnd:19940611.
- Neelima, Sharma R, Rajput YS, Mann B. Chemical and functional properties of glycomacropeptide (GMP) and its role in the detection of cheese whey adulteration in milk: A review. Dairy Science and Technology. 2013;93(1):21–43. DOI: https://doi.org/10.1007/s13594-012-0095-0.
- Kumari S, Vij S. Effect of Bioactive Peptides Derived from Fermented Whey Based Drink Against Food Borne Pathogens. International Journal of Current Microbiology and Applied Science. 2015;4(3):936–941.
- Umaraw P, Verma AK. Comprehensive review on application of edible film on meat and meat products: An ecofriendly approach. Critical Reviews in Food Science and Nutrition. 2017;57(6):1270–1279. DOI: https://doi.org/10.1080/10408398.2014.986563.
- Municipal Solid Waste in the United States: 2005 Facts and Figures Executive Summary. United States Environmental Protection Agency. 2006. 165 p.
- Sothornvit R, Krochta JM. Water Vapor Permeability and Solubility of Films from Hydrolyzed Whey Protein. Journal of Food Science. 2000;65(4):700–705.
- Pommet MA, Redl A, Guilbert S, Morel MH. Intrinsic influence of various plasticizers on functional properties and reactivity of wheat gluten thermoplastic materials. Journal of Cereal Science. 2005;42(1):81–91. DOI: https://doi.org/10.1016/j.jcs.2005.02.005.
- Liu L, Kerry JF, Kerry JP. Effect of food ingredients and selected lipids on the physical properties of extruded edible films/casings. International Journal of Food Science and Technology. 2006;41(3):295–302. DOI: https://doi.org/10.1111/j.1365-2621.2005.01063.x.
- Day BPF. Active Packaging. In: Coles R, McDowell D, Kirwan MJ, editors. Food Packaging Technology. Boca Raton: CRC Press; 2003. pp. 282–302.
- Khalil MS, Ahmed ZS, Elnawawy AS. Evaluation of the Physicochemical Properties and Antimicrobial Activities of Bioactive Biodegradable Films. Jordan Journal of Biological Sciences. 2013;6(1):51–60. DOI: https://doi.org/10.12816/0000259.
- Kuswandi B, Wicaksono Y, Jayus, Abdullah A, Heng LY, Ahmad M. Smart packaging: Sensors for monitoring of food quality and safety. Sensing and Instrumentation for Food Quality and Safety. 2011;5(3–4):137–146. DOI: https://doi.org/10.1007/s11694-011-9120-x.
- Morsy MK, Khalaf HH, Sharoba AM, El-Tanahi HH, Cutter CN. Incorporation of Essential Oils and Nanoparticles in Pullulan Films to Control Foodborne Pathogens on Meat and Poultry Products. Journal of Food Science. 2014;79(4):M675–M684. DOI: https://doi.org/10.1111/1750-3841.12400.
- Debeaufort F, Quezada-Gallo J-A, Voilley A. Edible films and coatings: Tomorrow’s packagings: A review. Critical Reviews in Food Science and Nutrition. 1998;38(4):299–313. DOI: https://doi.org/10.1080/10408699891274219.
- Galus S, Kadzińska J. Food applications of emulsion-based edible films and coatings. Trends in Food Science and Technology. 2015;45(2):273–283. DOI: https://doi.org/10.1016/j.tifs.2015.07.011.
- Gontard N, Marchesseau S, Cuq J-L, Guilbert S. Water vapour permeability of edible bilayer films of wheat gluten and lipids. International Journal of Food Science & Technology. 1995;30(1):49–56. DOI: https://doi.org/10.1111/j.1365-2621.1995.tb01945.x.
- Fabra MJ, Talens P, Chiralt A. Tensile properties and water vapor permeability of sodium caseinate films containing oleic acid–beeswax mixtures. Journal of Food Engineering. 2008;85(3):393–400. DOI: https://doi.org/10.1016/j.jfoodeng.2007.07.022.
- Sanyang ML, Sapuan SM, Jawaid M, Ishak MR, Sahari J. Effect of Plasticizer Type and Concentration on Tensile, Thermal and Barrier Properties of Biodegradable Films Based on Sugar Palm (Arenga pinnata) Starch. Polymers. 2015;7(6):1106–1124. DOI: https://doi.org/10.3390/polym7061106.
- Bykov DE, Eremeeva NB, Makarova NV, Bakharev VV, Demidova AV, Bykova TO. Influence of Plasticizer Content on Organoleptic, Physico-Chemical and Strength Characteristics of Apple Sauce-Based Edible Film. Foods and Raw Materials. 2017;5(2):5–14. DOI: https://doi.org/10.21603/2308-4057-2017-2-5-14.
- Hopkins EJ, Chang C, Lam RSH, Nickerson MT. Effects of flaxseed oil concentration on the performance of a soy protein isolate-based emulsion-type film. Food Research International. 2015;67(1):418–425. DOI: https://doi.org/10.1016/j.foodres.2014.11.040.
- Ortega-Toro R, Jiménez A, Talens P, Chiralt A. Effect of the incorporation of surfactants on the physical properties of corn starch films. Food Hydrocolloids. 2014;38:66–75. DOI: https://doi.org/10.1016/j.foodhyd.2013.11.011.
- Hambleton A, Debeaufort F, Bonnotte A, Voilley A. Influence of alginate emulsion-based films structure on its barrier properties and on the protection of microencapsulated aroma compound. Food Hydrocolloids. 2009;23(8):2116–2124. DOI: https://doi.org/10.1016/j.foodhyd.2009.04.001.
- Navarro-Tarazaga ML, Massa A, Pérez-Gago MB. Effect of beeswax content on hydroxypropyl methylcellulose-based edible film properties and postharvest quality of coated plums (Cv. Angeleno). LWT – Food Science and Technology. 2011;44(10):2328–2334. DOI: https://doi.org/10.1016/j.lwt.2011.03.011.
- Ayranci E, Tunc S. The effect of fatty acid content on water vapour and carbon dioxide transmissions of cellulosebased edible films. Food Chemistry. 2001;72(2):231–236. DOI: https://doi.org/10.1016/S0308-8146(00)00227-2.
- Bourtoom T. Edible protein films: Properties enhancement. International Food Research Journal. 2009;16(1).
- Pérez-Gago MB, Krochta JM. Lipid particle size effect on water vapor permeability and mechanical properties of whey protein/beeswax emulsion films. Journal of Agricultural and Food Chemistry. 2001;49(2):996–1002. DOI: https://doi.org/10.1021/jf000615f.
- Rubilar JF, Zúñiga RN, Osorio F, Pedreschi F. Physical properties of emulsion-based hydroxypropyl methylcellulose/whey protein isolate (HPMC/WPI) edible films. Carbohydrate Polymers. 2015;123:27–38. DOI: https://doi.org/10.1016/j.carbpol.2015.01.010.
- Zahedi Y, Ghanbarzadeh B, Sedaghat N. Physical properties of edible emulsified films based on pistachio globulin protein and fatty acids. Journal of Food Engineering. 2010;100(1):102–108. DOI: https://doi.org/10.1016/j.jfoodeng.2010.03.033.
- Sahari J, Sapuan SM, Ismarrubie ZN, Rahman MZA. Physical and chemical properties of different morphological parts of sugar palm fibres. Fibres and Textiles in Eastern Europe. 2012;91(2):21–24.
- Laohakunjit N, Noomhorm A. Effect of Plasticizers on Mechanical and Barrier Properties of Rice Starch Film. Starch/Staerke. 2004;56(8):348–356. DOI: https://doi.org/10.1002/star.200300249.
- Cao N, Fu Y, He J. Preparation and physical properties of soy protein isolate and gelatin composite films. Food Hydrocolloids. 2007;21(7):1153–1162. DOI: https://doi.org/10.1016/j.foodhyd.2006.09.001.
- Basiak E, Galus S, Lenart A. Characterisation of composite edible films based on wheat starch and whey-protein isolate. International Journal of Food Science and Technology. 2015;50(2):372–380. DOI: https://doi.org/10.1111/ijfs.12628.
- Tulamandi S, Rangarajan V, Rizvi SSH, Singhal RS, Chattopadhyay SK, Saha NC. A biodegradable and edible packaging film based on papaya puree, gelatin, and defatted soy protein. Food Packaging and Shelf Life. 2016;10:60–71. DOI: https://doi.org/10.1016/j.fpsl.2016.10.007.
- Galus S, Kadzińska J. Whey protein edible films modified with almond and walnut oils. Food Hydrocolloids.2016;52(1):78–86. DOI: https://doi.org/10.1016/j.foodhyd.2015.06.013.
- Otoni CG, Avena-Bustillos RJ, Olsen CW, Bilbao-Sáinz C, McHugh TH. Mechanical and water barrier properties of isolated soy protein composite edible films as affected by carvacrol and cinnamaldehyde micro and nanoemulsions. Food Hydrocolloids. 2016;57:72–79. DOI: https://doi.org/10.1016/j.foodhyd.2016.01.012.
- McClements DJ. Encapsulation, protection, and release of hydrophilic active components: potential and limitations of colloidal delivery systems. Advances in Colloid and Interface Science. 2015;219:27–53. DOI: https://doi.org/10.1016/j.cis.2015.02.002.
- Di Pierro P, Rossi Marquez G, Mariniello L, Sorrentino A, Villalonga R, Porta R. Effect of Transglutaminase on the Mechanical and Barrier Properties of Whey Protein/Pectin Films Prepared at Complexation pH. Journal of Agricultural and Food Chemistry. 2013;61(19):4593–4598. DOI: https://doi.org/10.1021/jf400119q.
- Cardoso VM, Borelli BM, Lara CA, Soares MA, Pataro C, Bodevan EC, et al. The influence of seasons and ripening time on yeast communities of a traditional Brazilian cheese. Food Research International. 2015;69:331–340. DOI: https://doi.org/10.1016/j.foodres.2014.12.040.
- Utama GL, Kurnani TBA, Sunardi, Balia RL. The Isolation and Identification of Stress Tolerance Ethanol-fermenting Yeasts from Mozzarella Cheese Whey. International Journal on Advanced Science, Engineering and Information Technology. 2016;6(2):252–257. DOI: https://doi.org/10.18517/ijaseit.6.2.752.
- Balia RL, Kurnani TBA, Utama GL. Selection of Mozzarella Cheese Whey Native Yeasts with Ethanol and Glucose Tolerance Ability. International Journal on Advanced Science, Engineering and Information Technology. 2018;8(4):1091–1097. DOI: https://doi.org/10.18517/ijaseit.8.4.5869.
- Ko BS, Rhee CH, Kim JH. Enhancement of xylitol productivity and yield using a xylitol dehydrogenase genedisrupted mutant of Candida tropicalis under fully aerobic conditions. Biotechnology Letters. 2006;28(15):1159–1162. DOI: https://doi.org/10.1007/s10529-006-9068-9.