Аннотация
The aim of this research was to assess the nutraceutical potential of Hura crepitans seed oil in food chemistry. For that, we determined the phytochemical composition, physicochemical parameters, mineral composition, as well as proximates of the oil under study. The yield of the oil obtained by the Soxhlet method was 47.8 ± 0.2%. According to the phytochemical screening result, the oil contained saponin, alkaloid, terpenoids, steroids, and cardenolides. Quantitative analysis of proximates for the Hura crepitans seed oil displayed 10.1 ± 0.4% of protein, 19.4 ± 0.1% of crude fibres, 14.5 ± 0.5% of carbohydrates, 5.3 ± 0.2% of water, and 2.9 ± 0.1% of ash. Some selected physicochemical parameters such as refractive index, pH, and specific gravity were 1.47; 5.2; and 0.80, respectively. Cloud point was 6.20°C. Metal content determination revealed the presence of mineral substances such as magnesium (119.51 ± 0.25 mg/kg), potassium (4.25 ± 0.04 ppm), iron (4.01 ± 0.03 mg/kg), manganese (3.66 ± 0.02 ppm), sodium (2.18 ± 0.02 ppm), calcium (0.11 ± 0.001 ppm), zinc (0.04 ± 0.001 µg/g), and cadmium (0.0028 ± 0.0004 mg/kg). However, such minerals as nickel and lead were not detected. The structural analyse was carried out based on physicochemical properties and spectroscopic data of the Hura crepitans seed oil. The results of the research proved the nutraceutical potential of this oil to use as raw materials in various areas of industry.Ключевые слова
Fatty acid, secondary metabolites, Soxhlet, Hura crepitans, proximate testВВЕДЕНИЕ
Recently, the use of plant oils in various industries has been consistently grown [1]. Hura crepitans seed oil is derived from a class of tree commonly known as the sandbox tree [2]. It originates from the tropical regions of North and South America and maintains its foliage throughout the year. It is also referred to as possumwood and jabillo aside sandbox tree in the earlier identified nomenclature [3].
Hura crepitans has can grow up to 30 m (100 ft) tall. It has large ovate leaves which can growth up to 2 ft wide in nature [4]. It is a typical rain forest tree with seed rich in oil [5, 6]. It is a prominent and salient medicinal plant widely used in sub-Saharan Africa [7]. H. crepitans seed oil has been reported to be essential raw material in biodiesel production [8]. It is also commonly called ‘Odan Mecca’ in Kabba part of the Kogi State and ‘Aroyin’ among Ilesha indigenes of the Osun State, both in the western region of Nigeria [9].
The woody fruits have resemblance with small pumpkin pods, with about thirteen seeds in each pod. This plant is used for the treatment of dermatitis, gastrointestinal disorders, and respiratory infections [10]. Oil obtained has been recognized to play the purgatory function. ‘Hurin’ is a non-color secretion from Hura crepitans which has been established by medicinal plant scientists as panacea for elephantiasis and leprosy [11].
Physiological and phenological reports [12] discovered this plant as edible food with diverse nutraceutical potentials aside utilization in biodiesel manufacturing [13]. Thus, this study deals with the evaluation of the seed oil of H. crepitans grown in the premises of the Covenant University, Nigeria. We analysed phytochemical compounds, physicochemical properties, as well as proximate and metal content compositions of the oil.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
Materials and measurements. The chemicals used were manufactured and supplied by Sigma-Aldrich Chemicals, USA. They included acetone, bromine water, chloroform, ethanol, conc. HCl, conc. sulfuric acid, n-hexane, magnesium ribbon, CuSO4 , and NaOH. The IR and UV analyses were carried out with a PMA-50 Bruker FT-IR spectrophotometer, manufactured by Bruker Optik GmbH, Germany, and a 840-208200 Genesys 10s uv-visible spectrophotometer, manufactured by Thermofisher Scientific, USA. A LI-AREF-186 refractometer, manufactured by LABARD Instruchem Private Limited, India, was applied to determine refractive index. All temperature measurements were carried out with Stuart SMP10 melting point apparatus supplied by Cole Parmer Company, Staffordshire, UK. A Buck scientific 210VGP atomic absorption spectrophotometer was utilized to quantitatively analyse the digested sample for metal content determination. A Perkin-Elmer 241 polarimeter (PerkinElmer Incorporation, California, USA) was used for optical rotation determination. Moisture content was determined by a RS232 DSH moisture analyser (W&J Instrument Company, China). The 1H (400 MHz) and 13C (100 MHz) NMR analysis were determined in CDCl3 with the help of a JEOL Delta NMR ECX 400 spectrometer (California, USA). The chemical shift values (δH and δc) were measured in part per million. When the oil extraction procedure finished, the n-hexane was evaporated with the help of a 0010004799 IKA® RV 10 Rotary evaporator (IKA-Werke GmbH and Co KG, Germany) to obtain the desired oil for further analysis.
Seed sample collection. Seeds of Hura crepitans were harvested in the early hours (from 7 a.m. to 9 a.m.) of the 15th May, 2017 on Covenant University campus, Nigeria, at 27 ± 2°C. From sandbox trees which were about eight years old, the matured and healthy fruits were randomly picked and thoroughly mixed together. These were then kept in a dark polyethylene bag and transported immediately to the laboratory for further processing. The plant species were identified and authenticated at the Department of Biological Sciences of the Covenant University.
Extraction and isolation. Extraction of the oil from the seed of Hura crepitans was done at Covenant University, Ota, Ogun State, Nigeria using the standard method [14, 15]. The dried seeds of Hura crepitans were thoroughly ground into powder to increase the extraction efficiency. 2 g of milled sample was weighed and carefully packed in filter paper and mounted on Soxhlet set-up and extracted with n-hexane for 6 hours. After that, we removed n-hexane from the mixture to ensure yellow oil, with yield of 47.79%.Spectral data for the seed oil sample
IR (KBr): 2,974; 2,822; 2,485; 1,750; 1,600; and 1,038 cmsup>–1;
1H NMR (400 MHz, CDCl3) δ: 5.35–5.30 (m, 5H), 4.31–4.26 (dd, J1 = 4.28 Hz, J2 = 8.00 Hz, 1H), 4.16–4.11 (dd, J1 = 5.96 Hz, J2 = 8.00 Hz, 1H), 2.79–2.74 (t, J = 12.72 Hz, 2H), 2.34–2.31 (m, 4H, 2·CH2), 2.03–1.99 (m, 6H), 1.60 (s, 3H), 1.29–1.24 (d, J = 12.72 Hz, 32H), 0.88 (m, 6H);
13C NMR (100 MHz, CDCl3) δ: 173.30; 172.81; 130.32; 130.11; 127.81; 68.80; 62.20; 34.11; 32.01; 31.62 (3·CH2); 29.80 (2·CH2); 29.40 (2·CH2); 29.20 (2·CH2); 27.30 (3·CH2); 25.70 (3·CH2); 24.90 (CH2); 22.70 (CH3); 14.21 (CH3); 13.50 (CH3) ppm;
MS: in m/z (rel. %): 508.97 (M+, 34.5%), 466.95 (100%), 354.40 (10.1%), 275.00 (22.5%), and 270.03 (35.2%).
Procedure for phytochemical test. Phytochemical screening was conducted using the standard procedure that we described earlier to reveal the secondary metabolites in Hura crepitans sample [16]. According to the result, the phytoconstituents in the sample were saponins, steroids, alkaloids, terpenoids, and cardenolides.
Test for terpenoids. 2 ml of chloroform was added to 5 ml the oil aliquot and stirred thoroughly. It was evaporated on the water bath and then heated up in the presence of 3 ml of concentrated H2SO4. Grey colour confirmed the presence of terpenoids.
Test for steroids. 2 ml of chloroform and 3 ml of concentrated H2SO4 were added to 5 ml of the oil. The appearance of red coloration within the lower chloroform portion depicted the presence of steroids.
Tannins testing. 10.00 ml of bromine water was cautiously added into 0.5 g of the oil. Decolorization of Br/H2O was a clear indication of the availability of tannins.
Saponins testing. 5.00 ml of distilled water and the oil aliquot were mixed vigorously in a test tube. The frothing was mixed with few drops of olive oil and mixed vigorously. Foam formation indicated the presence of saponins.
Flavonoids testing. Shinoda test was used to determine flavonoids. Several drops of concentrated HCl were added to the aliquot sample. Then magnesium ribbon turnings were slipped into the solution. The observation of pink coloration implied the presence of flavonoids. This was also confirmed by alkaline reagent test.
Test for alkaloids. An aliquot of the sample was spotted on a precoated thin layer chromatographic plate. The plate was sprayed with the Dragendorf reagent. The appearance of orange spot depicted the presence of alkaloids.
Test for steroid. 2 mg of ground seed sample was agitated in CHCl3. A solution of H2SO4 was carefully added through the test-tube wall to the CHCl3 layer; then sulphuric acid was added slowly by the sides of test tube.The appearance of red colour implied the presence of steroids.
Proximate analysis procedure. The nitrogen content was determined by the Micro-Kjeldahl technique, while multiplication of N (%) by conversion factor (6.25) afforded crude protein [16]. Moisture content and crude fibres were determined using standard methods [16, 17].
Determination of total carbohydrate. The determination of carbohydrate percentage of the oil sample was ascertained by another approach. The technique involved the taking of sum-total of crude protein, lipid, crude fibres, moisture, and ash from 100 [16].
Qualitative analysis. We used the Biuret test to determine protein content. 6% NaOH solution and few drops of 1% CuSO4 were added to the test solution in a sequential manner. Appearance of violet color in inference indicated the presence of protein is present [18].
In order to determine carbohydrate content, the Molisch’s test was used. Several drops of α-naphthol in alcohol were added into the extracted oil sample. This was followed by the addition of concentrated H2SO4 (1 ml) in a slowly and drop wisely manner through the side wall of the test-tube. Appearance of purple coloration that turned to violet in form of a ring at the junction of the resulting mixture depicted the presence of carbohydrates [19].
Determination of Total Ash. 5 g of the sample was weighed into a crucible which was previously dried to constant weight prior to use. The content was first heated gently over a low flame until charred, and transferred into a muffle furnace at 550°C until a white ash was formed. A standard procedure was adopted for both the ashing and calculation [11].
Mineral Content Analysis. Acid digestion method was used to prepare a solution of the sample in an acceptable format for the minerals content analysis according to a known procedure [15]. About 2 g of the seed samples were weighed and digested using double acid digestion with perchloric acid/nitric acid in ratio 1:1 using hot plate in the fume hood. When fume is invisible, the filtrate obtained was transferred into a 50 ml volumetric flask and prepared with distilled water prior to use. The solution was then analyzed for metals/elements such as Ca, Mg, Zn, Fe, Cd, Co, Pb, Ni, and Mn using AAS. Selective determination of K and Na was carried out with a PFP7 flame photometer (Jenway, UK).
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
Spectroscopic characterization. Since seed oils find a wide application to different industries, seeds Hura crepitans are of great interest in present research. In the course of the study into the seed oil from African shore, we have herein evaluated the valuable parameters and salient compositions of Hura crepitans seed oil [15, 16].
Physiological and phenological analyses revealed that during germination of oil seeds, the utilization of the storage fat was initiated by the stepwise hydrolysis of triacylglycerol to free fatty acids and glycerol [12]. The structural characteristics of the extracted seed oil of Hura crepitans was validated using spectroscopic means. The FT-IR spectral data of the compound was run from 4,000 to 400 cm–1 but showed no noticeable band above 3,000 cm–1. This implied that there was no N-H band and no OH band either free or hydrogen-bonded.
The highest absorption band was recorded at 2,974 cm–1 which was due to appearance of C-H of aliphatic followed by the second C-H aliphatic at the stretching vibrational frequency at 2,822 cm–1. The C = O of ester was noticed at 1,750 cm–1 and it was doubly established with C-O of alkoxyl functional at 1038 cm–1. The stretching vibration at 1,600 cm–1 was ascribed to the C = C functional moieties of alkene, and this value was in concordance with the literature value [14].
The 1H NMR analysis of the compound was run in deuterated chloroform using TMS as internal standard. The most downfield signal away from TMS was recorded as five proton multiplet at δH 5.35–5.30, after which one proton dd at δH 4.31–4.26 ppm with J values of 4.28 Hz and 8.00 Hz was recorded. Another 1H doublets of doublet appeared at δH 4.16-4.11 with J values of 5.96 Hz and 8.00 Hz. The chemically equivalent sixteen methylene protons resonated upfield as 32 protons doublet at δH 1.29–1.24 ppm. The presence of three methyl groups was confirmed by two signals in the 1H NMR spectrum as a 3H singlet (CH3) at δH 1.60 ppm and as a highly shielded 6H (2·CH3) multiplet at δH 0.88 ppm, which agreed with the value for the highly shielded methyl group earlier reported [20].
Furthermore, the
The mass spectrometric analysis revealed the molecular ion and base peaks to be at m/z 508.97 and 466.95 with intensities of 34.5% and 100%, respectively. The molecular ion peak was in agreement with the theoretical value for molar mass of the compound, and the base peak had m/z ratio which resulted in loss of a propyl radical (M – C3H7). Other daughter peaks due to some fragmentation patterns were noticed at m/z of 354.4; 275.0; and 270.0 with relative peak heights of 10; 22.5; and 35.0%, respectively. The uv-visible spectrum of the targeted sample in n-hexane showed the presence of four peaks with the wavelength λmax of 199 nm (log εmax 5.2), 210 nm (log εmax 4.9), 229 nm (log εmax 4.8), and 277 nm (log εmax 4.4) as shown in Table 1.
Phytochemical screening. In order to preview into potential bioactivity of the seed oil, the phytochemical screening was determined (Table 2). The phytochemical testing showed that cardenolides as well as saponins were present in low capacity, terpenoids and steroids in moderate capacity, and alkaloids and carbohydrates in high capacity. Saponins are glycosides with foaming ability [21]. Since saponins contain polycyclic aglycones which are attached to one or more sugar side chain, the presence of saponin in the Hura crepitans seed oil explained why it was rich in carbohydrates.
Table 2 demonstrates phytochemical substances in the Hura crepitans seed oil. As one can see from Table 2, Wagner and Dragendorff methods revealed the presence of alkaloids in high intensity. Steroid hormones are responsible for many osmoregulatory functions of physiological factors in human. Hence, availability of these secondary metabolites in moderate intensity indicates that adequate intake of this oil will aid hormonal boosting in vertebrates. The result of the phytochemical analysis also showed the absence of secondary metabolites which include tannins, flavonoids, phenols, phlabotannins, oxalates, and quinones. Since oxalates and cyanogenic glycosides contribute immensely to the removal of important nutritional component, the oil has great nutraceutical potential due to the absence of oxalate and quinones [14].
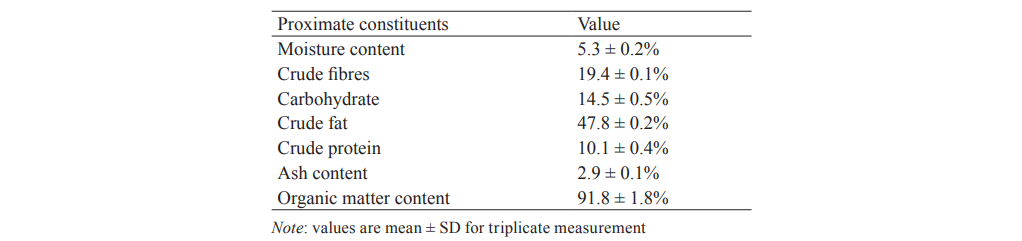
Proximate determination. Having obtained the oil via Soxhlet extraction technique, proximate determination results showed that Hura crepitans is rich in oil with percentage yield of 47.79%, as shown in Table 3 [14]. Crude fat yield was higher than the value (36.4%) previously reported by Ezeh et al. [22], closely related to the value (47.80%) reported in [23], but lesser when compared with 53.81% yield in the previous work of Abdulkadir et al. [11]. Nevertheless, the oil yield of Huran crepitans in this study was much higher than the value documented for soybean oil (18%) and groundnut oil (43%) [24].
It is very crucial to have a deep understanding of the nature of constituents which furnish energy as well as enhance digestible conditioning in food prior to effective preparation of commercially quality diets [16]. Over three decades now, curative medicine and therapeutic prevention against cardiovascular disorder is linked with balance diet intake [25]. Low moisture content (5.3 ± 0.2%) was an affirmation of the tendency for long shelf-life [11]. The proximate determination results also revealed that Hura crepitans was rich in carbohydrates (14.5 ± 0.5%), crude fibres (19.4 ± 0.1%), and crude proteins (10.1 ± 0.4%). The ash content was 2.9 ± 0.1%, which contributed to the total organic matter content (91.8 ± 1.8%). This ash value was within the expected limit (various reports on the ash show it does not exceed 5% in fresh food [26, 27]).
Njoku et al. investigated lipase properties of seed endosperm of Hura crepitans at the moment of germination. According to the results, its oil contained predominantly palmitic, stearic, oleic, and linoleic acids [12]. Idowu et al. reported that Hura crepitans contained oil and vitamins [28]. The high oil content in this research (47.8 ± 0.2%) implies the high level of essential vitamins.
The high carbohydrate content (14.5 ± 0.5%) ensured the valuable role of this substance for the enhancement of total well-being of man via the provision of energy for body metabolic processes [14]. The crude fibre content (19.4 ± 0.1%) will help in hasting digestion process [29]. Consistent and persistent intake of the recommended level of crude fibre helps the body to resist against numerous cardiovascular disorders [30].
The analysis also revealed that the crude protein was 10.1 ± 0.4% which implied that it was capable of ensuring the required cell division and growth to improve enzymatic action that is essential for hormonal balance in the body. We found that Hura crepitans seeds had remarkable efficiency as a means of boosting the energy load of the body. David et al. established that Hura crepitans seed oil possessed broad spectrum antimicrobial activity [30]. With MIC values of 1.25 mg/ml, the oil was effective against Pseudomonas aeruginosa (Z.O.I. = 5.5 mm), Salmonella typhi (Z.O.I. = 8.5 mm), Escherichia coli (Z.O.I. = 6.0 mm), and Enterobacter sp. (Z.O.I. = 6.0 mm) [30]. Thus, the high proximate profile together with the broad antimicrobial activities allowed us to conclude that Hura crepitans had great nutraceutical potential.
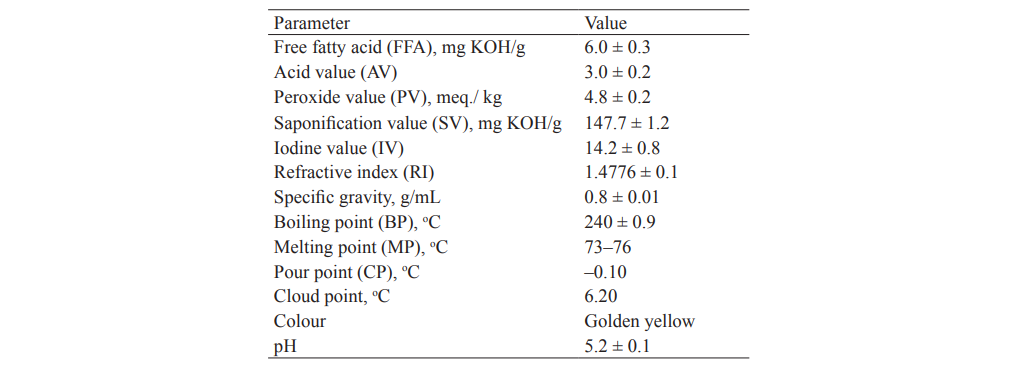
Physicochemical parameter determination. Table 4 shows the physicochemical properties of the Hura crepitans seed oil determined using standard methods. The parameters evaluated were refractive index (RI), specific gravity (SG), boiling point (BP), melting point (MP), cloud point (CP), etc.
The oil had low free fatty acid content (6.00) and acid value (3.00) which was an indicator that the oil was of good quality. The peroxide value was 4.80 (milliequivalent of O2/kg) and was within the stipulated limit by ASTM [31]. Ramadan [32] found that free fatty acid might be less suitable parameter to assess degradability of oil. This is because FFA content is unstable and can change due to oxidation and hydrolysis. However, PV is a good quality control parameter in this study since the lower PV value demonstrated that the oil had high quality, with no oxidative rancidity. The saponification value is 147.7 mg KOH/kg, which is an indicator of average molar mass of fatty acid. The acceptable range of SV for palm oil is 195–205 mg KOH/g according to Standard Organization of Nigeria [33]. Hence, the lower saponification value in this research, as compared with the standard value, clearly indicates that there are lesser ester bonds [34].
Moreover, iodine value (IV) for Hura crepitans was 14.23. It measures the degree of unsaturation in a fat or vegetable oil and determines how stable the oil is to oxidation [35]. A decrease in the total unsaturation of oils can indicate quality deterioration of stored oil [36]. Hence, the low IV in the oil under study might have contributed to a high stability of the oil during storage [34].
The refractive index of the oil was 1.478 and was in line with the American Standard of Test Material (ASTM) value (1.476–1.479). The boiling point (240°C), the pour point (–0.10°C), and cloud point (6.2°C) were in agreement with the values reported by O.O. Oniya et al. [13] who also investigated sandbox tree. Nonetheless, the cloud point, which had a value 6.2°C in this study, slightly varied from that obtained for peanut oil by Agarwal [37], which was 5°C. The specific gravity (0.88) at room temperature had the same value as in [38]. However, there was a slight variation in pH (5.2) from that in [22], where pH was 7.6. This might be due to different process condition.
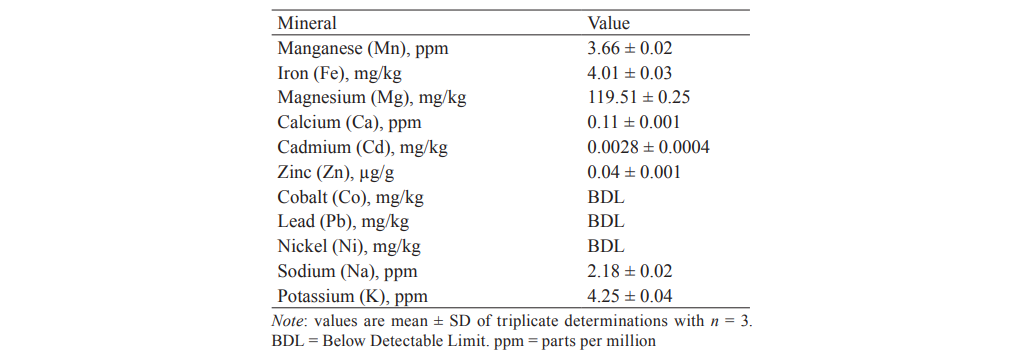
Mineral content determination. The determination of mineral substances in seed oil is an important factor for nutritional assessment and human health benefits that sums up to provide the nutraceutical potential of the oil [39]. To determine the mineral and metal contents in the Hura crepitans seed oil, we used the AAS technique. The results are represented in Table 5.
Since mg/kg and µg/g are equivalent to part per million (ppm), the latter will be used in the discussion for uniformity. Of eleven minerals determined, eight were detected while three elements were below the detectable limit of the machine used (Buck Scientific 210VGP AAS).
The order of the detectable minerals based on their relative abundance was as follows: Mg (119.51 ± 0.25 ppm) > K (4.25 ± 0.04 ppm) > Fe (4.01 ± 0.03 ppm) > Mn (3.66 ± 0.02 ppm) > Na (2.18 ± 0.02 ppm) > Ca (0.11 ± 0.001 ppm) > Zn (0.04 ± 0.001 ppm) > Cd (0.0028 ± 0.0004 ppm). Among the detected minerals, magnesium (Mg) was present in the highest concentration (119.51 ± 0.25 ppm), while cadmium (Cd) displayed the lowest value (0.0028 ± 0.0004 ppm). Such metals as lead (Pb), nickel (Ni), and cobalt (Co) were not detected because of their insignificant amount. This gives a considerable advantage to the consumption of this oil as metal lead is harmful for the body.
As for the other mineral substances, the amount of iron (Fe) contained in the Hura crepitans oil (4.02 ± 0.02 mg/kg) might assist in replacing lost blood via enrichment of hemoglobin. In addition, regular consumption of the oil would prevent anaemia disease in humans [15]. The manganese (Mn) value (3.66 mg/kg) was within the acceptable standard, which was 2–4 mg per day. Magnesium is a valuable element in animal feed and the human diet.
Calcium (Ca) is responsible for the formation of strong bone and teeth. In this study, calcium was detected in trace quantity (0.11 ± 0.001 ppm). Nevertheless, regular consumption of the Hura crepitans oil could reduce hypocalcaemia and osteoporosis resulted from calcium deficiency in the human skeletal system [40]. Sodium (Na) and potassium (K) content determination is of great importance because they are both intracellular and extracellular cations. The sodium content indicated that it would be able to control plasma volume, neuron transmission, and muscular contraction [16]. The values for magnesium, sodium, potassium, zinc, and calcium are in good agreement with those in Hura crepitans oil documented by Abdulkadir et al. [11].
The heavy metals such as nickel (Ni) and cobalt (Co) were not detected in the oil sample. It is noticeable that lead was not detected in the Hura crepitans oil as well, since lead showed toxic effect on living organisms. This implied that the oil was medically safe for consumption and could be used in the human diet.
ВЫВОДЫ
The seed of Hura crepitans has promising profile. Solvent-extracted oil from Hura crepitans was analysed for physicochemical parameters, mineral content, and phytochemical composition in order to assess the nutraceutical potential. The results of the research allow us to conclude that this oil can be used as an alternative source of dietary supplement and raw materials in animal feed. Structural analyse of the Hura crepitans seed oil was carried out based on its physicochemical parameters and spectroscopic data. The data reported in this research were obtained with the help of diverse standard methods. The Hura crepitans seed oil has potential for further investigations of its mechanical properties for technical applications.
КОНФЛИКТ ИНТЕРЕСОВ
The authors declare no conflict of interests.
БЛАГОДАРНОСТИ
The authors express their gratitude to the Covenant University for the support for this work.
СПИСОК ЛИТЕРАТУРЫ
- Montesano D., Blasi F., Simonetti M.S., Santini A., and Cossignani L. Chemical and nutritional characterization of seed oil from Cucurbita maxima L. (var. Berrettina) pumpkin. Foods, 2018, vol. 7, no. 3, pp. 30. DOI: https://doi. org/10.3390/foods7030030.
- Gbadamosi S.O. and Osungbade O.R. Effects of cooking and fermentation on the antinutrients, total phenolic contents and antioxidant properties of sandbox (Hura crepitans) seeds. International Food Research Journal, 2017, vol. 24, no. 4, pp. 1621–1627.
- Adewuyi A., Awolade P.O., and Oderinde R.A. Huran crepitans seed oil: An alternative feedstock for biodiesel pro- duction. Journal of Fuels, 2014, 8 pp. DOI: https://doi.org/10.1155/2014/464590.
- Oyelade J.O., Idowu D.O., Oniya O.O., and Ogunkunle O. Optimization of biodiesel production from sandbox (Hura crepitans L.) seed oil using two different catalysts. Energy Sources, Part A: Recovery, Utilization, and Environmental Effects, 2017, vol. 39, no. 12, pp. 1242–1249. DOI: https://doi.org/10.1080/15567036.2017.1320691.
- Okolie P.N., Uaboi-Egbenni P.O., and Ajekwene A.E. Extraction and quality evaluation of sandbox tree seed (Hura crepitans) Oil. World Journal of Agricultural Sciences, 2012, vol. 8, no. 4, pp. 359–365. DOI: https://doi.org/10.5829/ idosi.wjas.2012.8.4.1119.
- Oyeleke G.O., Olayiwola A.O., and Latona D.F. Chemical examination of sandbox (Hura crepitans) seed: Proximate, elemental and fatty acid profile. IOSR Journal of Applied Chemistry, 2012, vol. 1, no. 2, pp. 10–13. DOI: https://doi. org/10.9790/5736-0121013.
- Avoseh O.N., Ogunbajo L.O., Ogunwande I.A., Ogundajo A.L., and Lawal O.A. Anti-inflammatory activity of hexane and ethyl acetate extracts of Hura crepitans L. European Journal of Medicinal Plants, 2018, vol. 24, no. 1, pp. 1–6. DOI: https://doi.org/10.9734/EJMP/2018/41439.
- Oderinde R.A., Ajayi I.A., and Dewuyi A.A. Characterization of seed and seed oil of Hura crepitans and the kinetics of degradation of the oil during heating. Electronic Journal of Environmental, Agricultural and Food Chemistry, 2009, vol. 8, pp. no. 3, 201–208.
- Fowomola M.A. and Akindahunsi A.A. Nutritional Quality of Sandbox Tree (Hura crepitans Linn.). Journal of Me- dicinal Food, 2007, vol. 10, no. 1, pp. 159–164.
- Mshana N.R., Abbiw D.K., Addae-Mensah I., et al. Traditional Medicine and Pharmacopoeia: Contribution to the revision of ethnobotanical and Floristic Studies in Ghana. Organization of African Unity/Scientific, Technical & Re- search Commission Publ., 2000. 67 p.
- Abdulkadir M.N., Amoo I.A., and Adesina A.O. Chemical composition of Hura crepitans seed and antimicrobial ac- tivities of its oil. International Journal of Science and Research, 2013, vol. 2, no. 3, pp. 440–445.
- Njoku O.U., Ononogbu I.C., Nwaneri V.O., and Ugwuanyl J.O. Lipase activity in Hura crepitans seed endosperm during germination. Nigerian Journal of Botany, 1996, vol. 9, no. 21–26.
- Oniya O.O., Oyelade J.O., Ogunkunle O., and Idowu D.O. Optimization of solvent extraction of oil from sandbox kernels (Hura crepitans L.) by a response surface method. Energy and Policy Research, 2017, vol. 4, no. 1, pp. 36–43. DOI: https://doi.org/10.1080/23815639.2017.1324332.
- Owoeye T.F., Ajani O.O., Akinlabu D.K., and Ayanda O.I. Proximate composition, structural characterization and phytochemical screening of the seed oil of Adenanthera pavonina linn. Rasayan Journal of Chemistry, 2017, vol. 10, no. 3, pp. 807–814. DOI: https://doi.org/10.7324/RJC.2017.1031712.
- Ajani O.O., Owoeye T.F., Olasehinde G.I., et al. Characterization, proximate composition and evaluation of antimi- crobial activity of seed oil of Bauhinia tomentosa. Journal of Biological Sciences, 2016, vol. 16, no. 4, pp. 102–111. DOI: https://doi.org/10.3923/jbs.2016.102.111.
- Gul R., Jan S.U., Faridullah S., Sherani S., and Jahan N. Preliminary phytochemical screening, quantitative analysis of alkaloids, and antioxidant activity of crude plant extracts from ephedra intermedia indigenous to Balochistan. The 148 Scientific World Journal, 2017, p. 7. DOI: https://doi.org/10.1155/2017/5873648.
- AOAC. Association of Official Analytical Chemists. 21st Edn. Washington DC: AOCS Press Publ., 1999. 46 p.
- Suneetha V., Bishwambhar M., Gopinath R., et al. Screening and Identification of degradable products by pectin lyase producing Actinomycetes from katpadi and chittoor fruit industrial waste enriched soil samples. Asian Journal of Microbiology, Biotechnology and Environmental Sciences, 2012, vol. 14, no. 3, pp. 405–412.
- Gangwal A., Parmar S.K., and Sheth, N.R. Triterpenoid, flavonoids and sterols from Lagenaria siceraria fruit. Der Pharmacia Lettre, 2010, vol. 2, no. 1, pp. 307–317.
- Siyanbola T.O., Akinsola A.F., Obanla O.R., et al. Studies on the antibacterial and anticorrosive properties of syn- thesized hybrid polyurethane composites from castor seed oil. Rasayan Journal of Chemistry, 2017, vol. 10, no. 3, pp. 1003–1014. DOI: https://doi.org/10.7324/RJC.2017.1031811.
- Yahia E.M. Fruit and Vegetable Phytochemicals: Chemistry and Human Health. 2nd Ed. UK, Chichester: John Wiley and Sons Publ., 2018. 1149 p.
- Ezeh I.E., Umoren S.A., Essien E.E., and Udoh A.P. Studies on the utilization of Hura crepitans L. seed oil in the prepa- ration of alkyd resins. Industrial Crops and Products, 2012, vol. 36, no. 1, pp. 94–99. DOI: https://doi.org/10.1016/j. indcrop.2011.08.013.
- Oyekunle J.A.O. and Omode A.A. Chemical composition and fatty acid profile of the lipid fractions of selected Nige- rian indigenous oil seeds. International Journal of Food Properties, 2008, vol. 11, no. 2, pp. 273–281. DOI: https:// doi.org/10.1080/10942910701302598.
- Auta J. and Anwa E.P. Preliminary Studies on Albizzia lebbeck seeds. Proximate analysis and phytochemical screen- ing. Research Journal of Biological Sciences, 2007, vol. 2, no. 1, pp. 33–35.
- Stewart J., Manmathan G., and Wilkinson P. Primary prevention of cardiovascular disease: A review of con- temporary guidance and literature. JRSM Cardiovascular Disease, 2017, vol. 6, pp. 1–9. DOI: https://doi. org/10.1177/2048004016687211.
- Idowu D.O. and Abegunrin T.P. A study of some hydro-thermal properties of sandbox (Hura crepitans) seed. Agricul- tural Engineering International: CIGR Journal, 2014, vol. 16, no. 4, pp. 255–260.
- Chaudhary M. and Verma S.K. Analysis of the physicochemical properties of the processed fruits and vegetable pro- ducts. International Journal of Pharma and Bio Sciences, 2011, vol. 2, no. 4, pp. 660–666.
- Idowu D.O., Abegunrin T.P., Ola F.A., Adediran A.A., and Olaniran J.A. Measurement of some engineering prop- erties of sandbox seeds (Hura crepitans). Agriculture and Biology Journal of North America, 2012, vol. 3, no. 8, pp. 318–325.
- Ponnusamy S. and Vellaichamy T. Nutritional assessment, polyphenols evaluation and antioxidant activity of food resource plant Decalepis hamiltonii wight and arn. Journal of Applied Pharmaceutical Science, 2012, vol. 2, no. 5, pp. 106–110. DOI: https://doi.org/10.7324/JAPS.2012.2518.
- David O.M., Ojo O.O., Olumekun O.V., and Famurewa O. Antimicrobial activities of essential oils from Hura crep- itans (L.), Monodora myristica (Gaertn Dunal) and Xylopia aethiopica (Dunal A. Rich) seeds. British Journal of Applied Science & Technology, 2014, vol. 4, no. 23, pp. 3332–3341.
- Standard test method for oxidation onset temperature of hydrocarbons by differential scanning calorimetry. USA, Pennsylvania West Conshohocken: ASTM International Publ., 2002. pp. 734–738.
- Ramadan M.F., Amer M.M.A., and Sulieman, A.E.-R.M. Correlation between physicochemical analysis and radical scavenging activity of vegetable oil blends as affected by frying of French fries. European Journal of Lipid Science and Technology, 2006, vol. 108, no. 8, pp. 670–678. DOI: https://doi.org/10.1002/ejlt.200600058.
- Standards for Edible Refined Palm Oil and Its Processed Form. Standard Organization of Nigeria Publ., 2000. pp. 2–5.
- Zahir E., Saeed R., Hameed M.A., and Yousuf A. Study of physicochemical properties of edible oil and evaluation of frying oil quality by Fourier Transform-Infrared (FT-IR) Spectroscopy. Arabian Journal of Chemistry, 2014, vol. 10, pp. S3870–S3876. DOI: https://doi.org/10.1016/j.arabjc.2014.05.025.
- Asuquo J.E., Anusiem A.C.I., and Etim E.E. Extraction and characterization of rubber seed oil. International Journal of Modern Chemistry, 2012, vol. 1, no. 3, pp. 109–115.
- Perkin E.G. Effect of lipid oxidation on oil and food quality in deep frying. In: Angels A.J.S. (ed.) Lipid Oxidation in Food. Chapter 18. Washington DC: American Chemical Society Publ., 1992. pp. 310–321.
- Agarwal A.K. Vegetable oil versus diesel fuel: Development and use of biodiesel in a compression ignition engine. Teri Information Digest on Energy and Environment, 2005, vol. 8, pp. 191–204.
- Afolabi T.J., Onifade K.R., Akindipe V.O., and Odetoye T.E. Optimization of Benth seed oil (Parinari polyandra) using response surface methodology. British Journal of Applied Science & Technology, 2014, vol. 5, pp. 437–445.
- Nunes L.S., Barbosa J.T.P., Fernandes A.P., et al. Multi-element determination of Cu, Fe, Ni and Zn content in vegetable oils samples by high-resolution continuum source atomic absorption spectrometry and microemulsion sample preparation. Food Chemistry, 2011, vol. 127, no. 2, pp. 780–783. DOI: https://doi.org/10.1016/j.food- chem.2010.12.147.
- Beto J.A. Role of calcium in human aging. Clinical Nutrition Research, 2015, vol. 4, no. 1, pp. 1–8. DOI: https://doi. org/10.7762/cnr.2015.4.1.1.