Аннотация
Coffee pulp is the first waste product obtained during the wet processing of coffee beans. Coffee pulp makes up nearly 40% of the total weight of the coffee cherry. Coffee pulp contains 25.88% of cellulose, 3.6% of hemicel- luloses, and 20.07% of lignin. Coffee pulp is considered as an ideal substrate of lignocellulose biomass for micro- bial fermentation to produce such value-added products as ethanol. In this study, we used alkaline pre-treatment of the coffee pulp with NaOH (0.2 g/g biomass) in a microwave system at 120°C during 20 min. This method gave the best results: 71.25% of cellulose remained, and 46.11% of hemicellulose and 76.63% of lignin were removed. After that, the pre-treated biomass was hydrolyzed by Viscozyme Cassava C (enzyme loading was 19.27 FPU/g) at 50°C for 72 hours. The results showed that the highest reducing sugars and glucose concentration after hydrolysis were 38.21 g/l and 30.36 g/l, respectively. Then, the hydrolysis solution was fermented by S. cerevisiae (3.108 cells/ml) at 30°C for 72 hours. The highest concentration of ethanol obtained was 11.28 g/l. The result illustrated that, available and non- edible as it is, coffee pulp could be a potential feedstock for bioethanol production in Vietnam.Ключевые слова
Bioethanol, coffee pulp, Coffea robusta, lignocellulose biomass, hydrolysis, pre-treatmentВВЕДЕНИЕ
Vietnam is currently the world’s largest exporter of Robusta coffee, as well as the world’s second-largest exporter of coffee beans after Brazil. In 2016, the total production of coffee beans in Vietnam was about 1,636,500 tons. About 450,000 tons of dried coffee pulp is produced here annually. Coffee pulp is mainly used as a fuel for fruit/coffee beans drying or as a compost and fertilizer on coffee plantations, which causes serious environmental pollution.
All over the world, there have been many researches on the use of coffee pulp. For instance, feeding and digestibility studies were conducted in concrete ponds to evaluate the use of coffee (Coffea robusta) pulp as a partial and total replacement for yellow maize in low-cost diets for catfish [1]. The research evaluated the effect of adding coffee husks to animal feed as a substitute for a mixture of corn grain, husks, and cobs. In addition, there have been many studies on how coffee solid wastes can be used. For example, Flammulina velutipes mushroom can be cultivated on coffee spent-ground and coffee husk [2]. Coffee husk can be used as a carbon source for citric acid production in a solid-state fermentation system [3] or for wastewater treatment [4]. However, researchers are more concerned with producing ethanol from coffee pulp using chemical methods [5]. The problem is that these methods remain limited and eco-unfriendly as coffee pulp hydrolysis requires acid and alkali, which means expensive sophisticated equipment. In addition, coffee pulp has a high concentration of carbohydrates and, thus, can be used as a potential raw material for bioethanol production [6]. Besides, recent studies indicate that residue utilization has an excellent potential for bioethanol production, given that it does not involve costs related to raw material growth. Furthermore, it is estimated that ethanol production from agricultural residues could be sixteen times higher than the current production [7].
Vietnam possesses large quantities of coffee pulp hat need utilization. This is also in line with the current global trends to seek alternative renewable energy sources to replace traditional fossil fuels and solve the problem of environmental pollution and climate change. Thus, the present study offers a good solution for these problems.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
Materials. Robusta coffee pulp was collected at Pong Drang commune, Krong Buk district, Dak Lak province, Vietnam. The berries were of bright-red colour, ripe, neither crushed nor moldy. After harvesting, the pulp was removed and dried at 65°C until the moisture content was 5–8%. After that, the pulp was crushed and sieved; the diameter of the powder was 0.5–1 mm. Finally, the powder was packaged in plastic bags and stored under ambient conditions.
Analytical methods. The moisture content was analyzed according to AOAC method 934.06.
The total ash content was determined by using AOAC method 942.05.
The analysis of total fat was performed by using AOAC method 948.16.
The quantitative analysis of caffeine was performed by using a Genesys UV-Vis Spectrometer (Genesis 10S) [8].
The total polyphenol content in the extracts was determined according to the Folin-Ciocalteu colorimetric method with some modifications [9].
The micro-Lowry method [10] was used to determine the protein content.
The calcicum pectate method was applied to determine the pectin content [11].
Phenol sulphuric acid was used to estimate the total reducing sugars (TRS) using maltose as standard [12].
The reducing sugars (RS) in the hydrolysate were measured by using the DNS method adapted from Miller [13].
A Clever Check blood glucose meter (model TD 4230, Germany) [14] was used to determine the monomeric sugars (glucose).
The cellulose, hemicellulose, and lignin contents were determined by the crude fibre analysis [15].
The ethanol concentration was determined with the help of a Genesis UV-Vis Spectrometer (Genesis 10S) [16].
Pre-treatment method. 50 g of the dried coffee pulp was treated by 500 ml of sodium hydroxide solution (0.2 g NaOH/g biomass). After that, the mixture was pre-treated at 195W and 120°C for 20 min in the microwave system. The pre-treated biomass was recovered by filtration and washed with 1,000 mL of hot water (70°C) to remove the remaining lignin and alkaline substances according to the method offered by Chen et al. (2007) [17]. Then the pre-treated residue was pressed to remove excess water and dried at 65°C until moisture content stabilized between 5% and 8%. The concentrations of cellulose, hemicellulose, and lignin remaining in the pre-treated material were calculated by the following equation:
![]()
where Rx is the percentage of cellulose (RC), hemicellulose (RH), or lignin (RL) remaining in the pre-treated pulp, %; Ai is the amount of the constituent in the initial dried coffee pulp, g; and A is the amount of the constituent after the pre-treatment of the dried coffee pulp, g.
Hydrolysis method (enzyme loading). 5 mL of Viscozyme Cassava C preparation, 150 ml of 0.05 mol/l citrate buffer (pH 4.8), and 15 g (equivalent to 10% of dry material per 100 ml of solution, w/v) of pressed pre-treated dried pulp were mixed in a flask. The containers were incubated in a thermal shaker at 50°C and 150 rpm for 72 hours. After that, the material from each treatment was centrifuged at 2,500 rpm for 10 min [19]. The supernatant was removed to determine RSs, total reducing sugars (TRSs), and glucose concentrations. The control samples were not treated by heat and alkaline. The yield from the enzymatic hydrolysis process, %, was calculated using the following equation. Only the cellulose present in the pre-treated coffee pulp was taken into account:
![]()
where Ge is the glucose concentration at the end of the enzymatic hydrolysis, g glucose/l [18]; Gw is the glucose concentration without enzyme treatment, g glucose/l; and Cp is the cellulose concentration in the pre-treated material, g cellulose/l.
Fermentation method. After the hydrolysis, the solution was divided into equal portions of 250 ml each and put in an Erlenmeyer flask. Then (NH4)2SO4 (1 g/l), K2HPO4 (0.1 g/l) and MgSO4.7H2O (0.2 g/l) were added into the solution. The medium was autoclaved at 121°C for 20 min and cooled at room temperature. Fermentation was carried out in an Erlenmeyer flask with 3.108 cells/ml of S. cereviciae at 30°C, 120 rpm, and pH of 5 [20]. The yeast was collected from the Laboratory of the Food Technology Department at the Industrial University of Ho Chi Minh City. Ethanol concentration was analyzed by using a Genesis UV-Vis Spectrometer at different fermentation times:
![]()
where EC is ethanol concentration at the end of fermentation, g/l; Gb is glucose concentration at the beginning of the fermentation, g/l; Ge is glucose concentration at the end of the fermentation, g/l. The percentage of the theoretical ethanol yield was calculated as follows:
![]()
where 0.51 is the maximum theoretical ethanol yield when converting 1g glucose to ethanol.
Statistical analysis. All treatments in this study were conducted in triplicate, and 95% of confidence level was applied for the data analysis. ANOVA was used by the one-way analysis of variance, and Statgraphics software (Centurion XV) was used to determine the statistical differences between the treatments.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
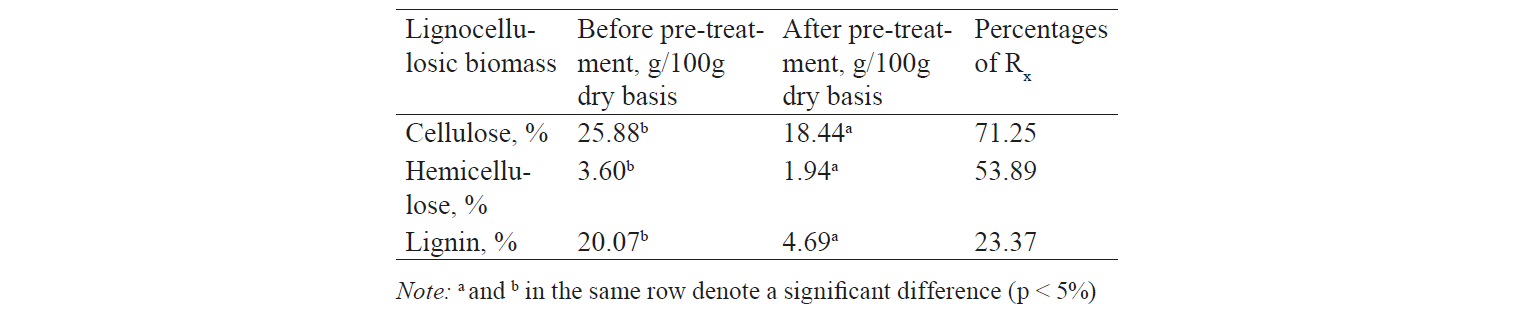
Characteristics of the solid fraction of coffee pulp. Table 1 shows that the cellulose and lignin content in the coffee pulp (Robusta coffea) was 25.88% and 20.07%, respectively. These results were higher than those received by Bonilla-Hermosa et al., Elias, and Menezes et al. However, the hemicellulose content was similar with the result obtained by Elias (1979) [18, 22, 23]. These differences can be explained by the fact that the previous studies used Arabica, whereas the present research was based on Robusta coffee.
Coffee husks and pulp are comprised of the outer skin and the attached residual pulp, and these solid residues are obtained after de-hulling of the coffee cherries during dry or wet processing, respectively [4]. The coffee pulp only included outer skin and fruit pulp. The sticky coffee husk included skin, fruit pulp, and, perhaps, an insignificant amount of pectin and parchment. Therefore, the total sugars (28.7%) and the reducing sugar content (24.25%) of the sticky coffee husk were higher than those of the coffee pulp (9.7 and 9.63%) [22, 25].
According to Palonen and Hetti [26], lignocellulose biomass is a major structural component of woody plants and other plants, such as grass, rice, and maize. The major constituents of lignocellulose are cellulose, hemicellulose, and lignin. The crude fibre in coffee pulp included: 25.88% of cellulose, 3.6% of hemicelluloses, and 20.07% of lignin. The content of cellulose in the coffee pulp was similar to that in rice husk (24.3%) [27] but lower than in wheat straw (38.2%) [28] and bagasse (38%) [29]. However, there was also a similar proportion between the cellulose content in the coffee pulp (equivalent to 52.23%, g cellulose/100g crude fibre) and the typical proportion of lignocellulose (40–60%) [30]. Therefore, coffee pulp is also considered a source of lignocellulose biomass, which can be used in the production of bioethanol (second generation ethanol production).
Alkali pretreatment. According to Sun and Cheng [31], the pre-treatment process has a number of advantages: it reduces cellulose crystallinity, removes lignin and hemicellulose, and increases the porosity of the materials. Pre-treatment should meet a number of requirements:
– it cannot produce by-products that are inhibitory to the subsequent hydrolysis and fermentation processes;
– it cannot result in a loss or degradation of carbohydrate;
– it should improve the formation of sugars or the ability to subsequently form sugars by enzymatic hydrolysis;
and
– it has to be cost-effective. Currently, pre-treatment of lignocellulosic materials can be chemical, physical, physico-chemical, and biological. As for materials that are rich in lignin, alkaline pre-treatment method seems to be the most efficient one.
The efficiency of pre-treatment depends entirely on the type of alkalis, concentration, time, and temperature of the pre-treatment process. To increase the efficiency of lignin removal, the above factors need to be increased. However, the increase in these factors means more cellulose loss. In this study, the coffee pulps were pre-treated with 0.2 g NaOH/g biomass at 120°C for 20 min in a microwave system. The results showed that 71.25% of cellulose was retained, while 46.11% of hemicellulose and 76.63% of lignin were removed. Although the result was not high, the conversion efficiency could not be regarded as low.
It was necessary to go through the next stages (hydrolysis and fermentation) to evaluate the ethanol conversion efficiency. According to [18], when coffee pulp was pre-treated with 4% NaOH (w/v) at 121°C for 25 min, the commercial efficiency removal of lignin and hemicellulose was 78,41% and 55.85%, respectively, while 69.18% of cellulose was obtained. Wang and Cheng [32] pre-treated coastal Bermuda grass with sodium hydroxide (1% NaOH) and calcium hydroxide (0.1 g Ca(OH)2) (in g/dry biomass) at 121°C during 30 min and obtained about 75% and less than 20% of lignin removal, respectively. In addition, the results of Kim and Holtzapple [33] showed that the optimal conditions of pre-treatment for corn stover were 0.5 g Ca(OH)2/g raw biomass (55°C). After 4 weeks of pre-treatment, 97.7% of cellulose remained while 32.3% of hemicellulose and 66.9% of lignin were removed. This result showed that the effect of Ca(OH)2 was better than that of NaOH because it was cheaper and produced a larger amount of cellulose. However, hydrolysis and fermentation need to be conducted to evaluate its effectiveness mainly because the final product is ethanol.
Fig. 1 shows that NaOH treatment of lignocellulosic materials caused swelling, which increased the internal surface area, decreased the degree of polymerization and crystallinity, separated the structural linkages between lignin and carbohydrates, and disrupted the lignin structure [34]. The mechanism of alkaline hydrolysis is believed to be in the saponification of intermolecular ester bonds which crosslink xylan hemicelluloses and other components, e.g. lignin and other hemicellulose. The porosity of the lignocellulosic materials increases with the removal of the crosslinks [35]. In addition, Fig. 1 also shows the difference between the raw and the pre-treated samples. The raw samples were rigid, very compact, and non-porous, while the pre-treated sample showed an increase in porosity and a greater surface area, which was attributed to the removal of lignin and hemicellulose.
The alkali pre-treatment can cause redistribution and solubilization of lignin, as well as an increase in the hydrolyzation of cellulose and the porosity of substrates. However, alkali pre-treatment combined with microwave pre-treatment could significantly increase the cellulose hydrolyzation by enzyme because of a larger contact area of cellulose and enzyme [36]. It also causes linkage degradation between cellulose, hemicelluloses, and lignin. In addition, this pre-treatment process has a low temperature and a low alkali concentration, which leads to a lower sugar degradation, and no yeast inhibitor agents are released, so ethanol fermentation yield is higher than when no microwave pre-treatment was conducted. This viewpoint corresponds with the results obtained by Zhao et al. [36], who combined alkali pre-treatment and microwave pre-treatment of rice hulls. As a result, the reducing sugar increased by 14% if compared with the samples that underwent no microwave pre-treatment. The results obtained by Xu et al. [37] showed that the ethanol yield from fermentation process was nearly 6 times higher than that obtained from the untreated material.
Enzymatic hydrolysis. To assess the effectiveness of alkaline pre-treatment, a certain percentage of hemicellulose and lignin was removed. The ability of enzymatic hydrolysis to produce a particular amount of glucose had to be taken into account. In addition, to evaluate the effectiveness of the pre-treatment process, a control sample was established. The control samples (not pre-treated with alkali) were also hydrolyzed simultaneously with the test sample under the same conditions. However, the hydrolysis effect was completely different (Table 3).
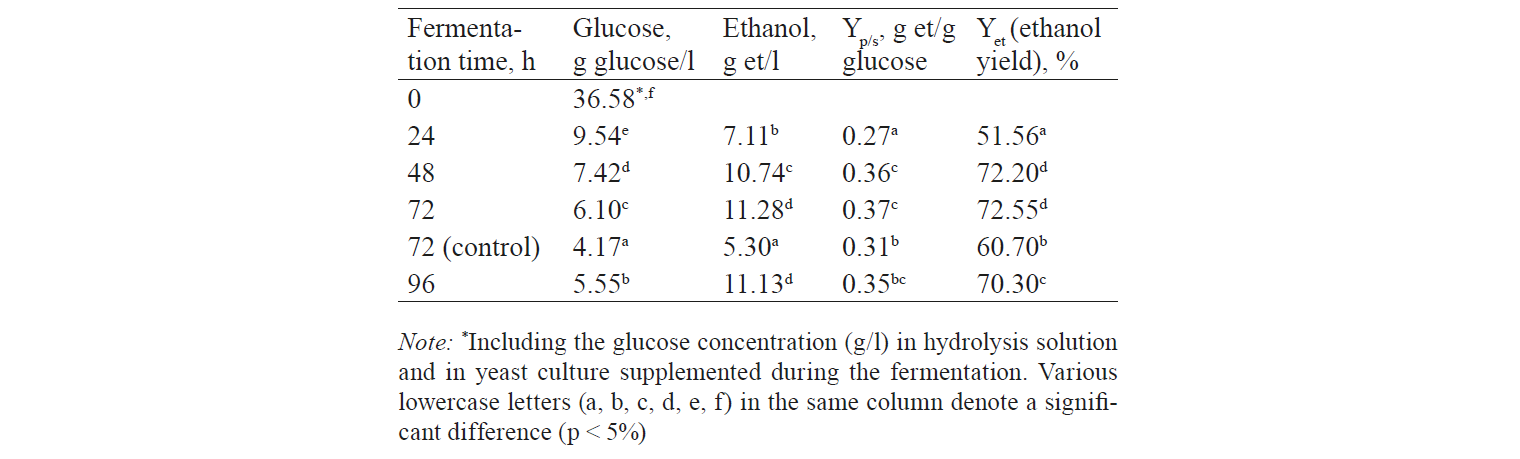
Table 3 shows that YEH (yield of enzymatic hydrolysis) was 76.8% (the result was calculated by formula 2). It means that 76.8% of cellulose was converted to glucose after the hydrolysis process. The productivity was relatively high. According to Menezes et al., hydrolysis of coffee pulp (Arabica) resulted in producing 27.02 g/l of glucose and 60.48% of hydrolysis efficiency [18].
Silverstein et al. pre-treated cotton stalks and stems with a NaOH solution (2% NaOH, 60 min, 121°C) and obtained conversion of cellulose equal to 60.8% after the enzymatic hydrolysis. This productivity was lower than the results obtained in the study [40], which can be explained by the following factors. When the alkaline pretreatment of lignocellulose was combined with the microwave hydrolysis, the hydrolysis yield was higher. The hydrolyzed sugar content also improved, as compared to the case when alkaline pre-treatment was used on its own. In addition, if combined with microwaves, hydrolysis of the alkaline pre-treatment increases the surface area of cellulose from the breakdown of the hemicellulose and lignin layers. As a result, the sensitivity of hydrolysis enzymes increases [41, 42]. On the other hand, Chen et al. [17] pre-treated barley straw with 2% NaOH at 121°C. According to their experiment, 74.03–84.89% of cellulose conversed to glucose during the enzymatic hydrolysis process. The cellulase concentration was 40–60 FPU/g cellulose supplemented with cellobiase (Novozyme 188). Their result was better than that of this study, with an enzyme loading of 19.27 FPU/g of substrate.
The productivity increased considerably when the amount of enzyme was increased (FPU/g), or enzyme cellobiase was supplemented (CBU/g). Chen et al. pre-treated corn straw with a 2% NaOH solution for 1 hour at 80°C with an 8% substrate and 20 FPU cellulase/g substrate (which contained 1.64 CBU/g) [43]. The hydrolysis process produced a RS concentration equal to 52 g/l and a high amount of cellobiose. In addition, the RSs content peaked at 64.1 g/l after 60 hour with an increase in the activity of cellobiase to 10 CBU/g substrate. The cellulase activity is inhibited by cellobiose and glucose (to a lesser extent). These problems can be solved by adding cellobiase. In the hydrolysis of lignocellulosic biomass, cellulases attack the cellulose chain to form glucose and cellobiose, then cellobiose decomposes to glucose by cellobiase. Thus, the presence of cellobiase helps reduce the inhibition of cellulase by cellobiose and results in a higher yield of sugars [31].
Fermentation. The glucose concentration in the hydrolysate was consumed by the yeast. The initial concentration was 36.58 g/l and decreased to 6.1 g/l after 72 hour of fermentation. The final concentration of ethanol was 11.28 g/l with a yield of 0.37 g ethanol/g glucose. The control sample (unprotected by hydrolysis and fermentation) showed the initial glucose concentration of 20.85 g/l and 4.17 g/l at the end of fermentation. The ethanol concentration was only 5.3 g/l (Table 4). Therefore, it was necessary to conduct a pre-treatment process to remove lignin and hemicellulose.
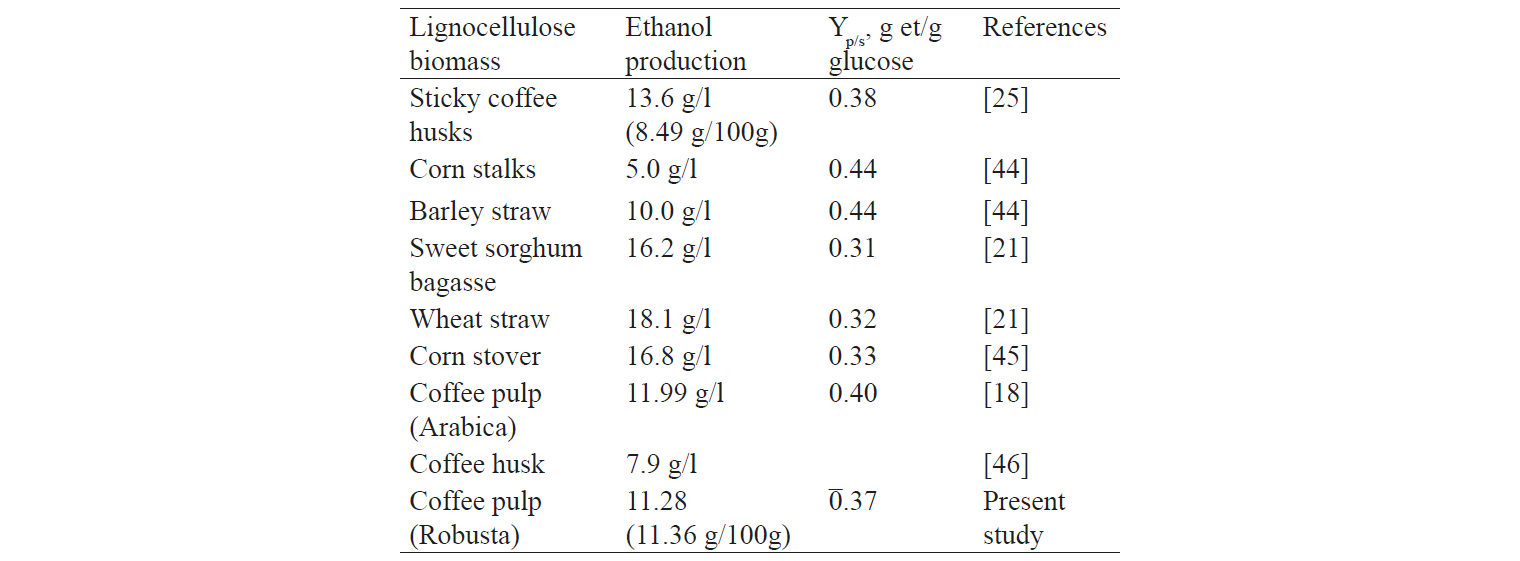
According to Menezes et al., coffee pulp (Arabica) was pre-treated by a 4% NaOH solution (w/v) (equiequivalent to 0.2 g/g biomass) and then was hydrolysed with 13.82 FPU/g of cellulase [18]. The results showed that 27.02 g of glucose/l and 11.99 g of ethanol/l were achieved after hydrolysis and fermentation (Table 5).
In this study, the pre-treatment process was combined with the microwave system. As a result, the hydrolysis efficiency was high (30.36 g of glucose/l was formed) (Table 3). However, the ethanol yield from fermentation was only 11.28 g/l, which was slightly lower than that of the study by Menezes et al. because of the yeast strain or its amount. Besides, they used 3 g/l of dry yeast while the yeast in this study was used as a secondary breed and then added to the hydrolysate with a cell density of 3.108 (cells/ml). Chen et al. pre-treated barley straw with a 2% NaOH solution at 121°C for 1 hour, and the pre-treated material was hydrolysed by Celluclast 1.5 l at a concentration of 40 FPU/g glucose and Novozyme 188 (cellobiase) [17]. The hydrolysate was inoculated by S. cerevisiae (ATCC 24859) and incubated at 30°C for 72 hour. At the end of the fermentation, the ethanol yield (Yp/s) was 0.31 g ethanol/g glucose, which is lower than the result of this study (0.37).
The coffee pulp was hydrolysed by using sulphuric acid concentrations of 1%, 2%, and 4% for 1 hour; the achieved ethanol concentrations were 6.097, 4.395, and 3.323 g/l, respectively [47]. The dilute acid hydrolysis resulted in a low ethanol production compared with the deionised water hydrolysis. The maximum ethanol concentration of 6.315 g/l was obtained from the coffee pulp, which was hydrolysed by deionised water [48]. Sugar cane bagasse was pre-treated by steam explosion and hydrolysed by cellulase (26 g RS/l in hydrolysate). The final concentrations of ethanol were 7.4 g/l (0.28 g ethanol/g RS) and 8.2 g/l (0.31 g ethanol/g RS) when two types of yeast were used, i.e. S. cerevisiae ATCC96581 and S. cerevisiae TMB3001 [39].
150 ml of filtrate yeast (Saccharomyces cerevisiae) was added at a concentration of 5.0 g/l and subjected to fermentation for 48 hour at 30°C in a shaker incubator at 120 rpm. The ethanol yield in the fermented broth was found to be 0.50; 0.46; and 0.46 g/g sugar in squeezed CAP, DCP, and WCP. The theoretical ethanol yields (Ymax%) of squeezed cashew apple pulp, dry coffee pulp, and wet coffee pulp were found to be 46.0; 9.35; and 40.0%, respectively [5].
Other studies showed that after fermentation barley straw produced the final ethanol concentration equalled to 10 g/l [44]. In addition, Gouvea et al. [25] indicated that when coffee husk was fermented in water (13%, w/v) with S. cerevisiae commercial Baker’s yeast, the final ethanol concentration equalled 13.6 g/l (Table 5).
The conversion of glucose to high or low ethanol content is attributed either to the low concentration of glucose in the hydrolysis solution or to the poor pre-treatment process, or to the low cellulose content in the raw material. This is consistent with the study conducted by Belkacemi et al. [44] that showed an ethanol conversion efficiency of 0.44, while the ethanol content was 5 g/l (Table 5). However, Ballesteros et al. obtained quite opposite results [21]: the conversion efficiency reached only 0.32 while the obtained ethanol content was 18.1 g/l (Table 5). Thereby, the conversion efficiency of ethanol indicated that the glucose content in the fermentation broth was high. The high glucose content showed that the hydrolysis process or the fermentation process was very effective.
ВЫВОДЫ
One hundred gram of dry coffee pulp produced 11.36 g of ethanol (the corresponding glucose conversion efficiency of ethanol was 0.37). Comparing with the literature data, it can be seen that ethanol production by fermentation is quite potential. In addition, the present study offers some methods to improve ethanol yields, including the use of a combination of yeast strains for xylose fermentation, hydrolysis coupled with concurrent fermentation, or selection of more potent strains of S. cerevisiae.
БЛАГОДАРНОСТИ
The authors would like to thank the Institute of Biotechnology and Food Technology, the Industrial University of Ho Chi Minh City (Vietnam), and our colleagues for the technical and equipment support.
СПИСОК ЛИТЕРАТУРЫ
- Ribeiro Filho E., Paiva P.C.A., Barcelos A., et al. The effect of coffee hulls on the performance of Holstein-zebu steers during the growing period. Ciência e Agrotecnologia, 2000, vol. 24, pp. 225–232.
- Leifa F., Pandey A., and Soccol C.R. Production of Flammulina velutipes on Coffee Husk and Coffee Spent-ground. Brazilian Archives of Biology and Technology, 2001, vol. 44, no. 2, pp. 205–212. DOI: https://doi.org/10.1590/S1516- 89132001000200015.
- Shankaranand V. and Lonsane B. Coffee husk: an inexpensive substrate for production of citric acid by Aspergillus niger in a solid-state fermentation system. World Journal of Microbiology and Biotechnology, 1994, vol. 10, no. 2, pp. 165–168. DOI: https://doi.org/10.1007/bf00360879.
- Franca A.S. and Oliveira L.S. Coffee processing solid wastes: current uses and future perspectives. Agricultural wastes, 2009, vol. 9, pp. 155–190.
- Shenoy D., Pai A., Vikas R., et al. A study on bioethanol production from cashew apple pulp and coffee pulp waste. Biomass and Bioenergy, 2011, vol. 35, no. 10, pp. 4107–4111. DOI: https://doi.org/10.1016/j.biombioe.2011.05.016.
- Oliveira L.S., Franca A.S., Camargos R.R., and Ferraz V.P. Coffee oil as a potential feedstock for biodiesel production. Bioresource Technology, 2008, vol. 99, no. 8, pp. 3244–3250. DOI: https://doi.org/10.1016/j.biortech.2007.05.074.
- Saha B.C. and Cotta M.A. Fuel ethanol production from agricultural residues: current status and future prospects. Journal of Biotechnology, 2008, vol. 136, pp. S285–S286. DOI: https://doi.org/10.1016/j.jbiotec.2008.07.613.
- Phuong D.V., Tan V.P., and Duy L.N.D. Determination of caffeine in coffee pulp (Coffea robusta) using UV- Visiblespectrophotometer. Vietnam Journal of Chemistry, 2017, vol. 55, pp. 86–91.
- Ayesha S., Premakumari K., and Roukiya S. Antioxidant activity and estimation of total phenolic content of Muntingia calabura by colorimetry. International Journal of ChemTech Research, 2010, vol. 2, no. 1, pp. 205–208.
- Lowry O.H., Rosebrough N.J., Farr A.L., and Randall R.J. Protein measurement with the Folin phenol reagent. Jour- nal of Biological Chemistry, 1951, vol. 193, pp. 265–275.
- Carré M.H. and Haynes D. The estimation of pectin as calcium pectate and the application of this method to the de- termination of the soluble pectin in apples. Biochemical Journal, 1922, vol. 16, pp. 60. DOI: https://doi.org/10.1042/ bj0160060.
- Dubois M., Gilles K.A., Hamilton J.K., Rebers P.T., and Smith F. Colorimetric method for determination of sug- ars and related substances. Analytical Chemistry, 1956, vol. 28, no. 3, pp. 350–356. DOI: https://doi.org/10.1021/ ac60111a017.
- Miller G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Analytical Chemistry, 1959, vol. 31, no. 3, pp. 426–428. DOI: https://doi.org/10.1021/ac60147a030.
- Le P.T.Q. and Pham M.H. The effects of ethephon on the ripening of carambola (Averrhoa carambola L.). Internatio- nal Food Research Journal, 2017, vol. 25, pp. 1497–1501.
- Von Soest P. and Wine R. Use of detergents in the analysis of fibrous feed. Iv. Determination of plant cell-wall consti-tuent’s journal. Association of Analytical Chemistry, 1967, vol. 50, pp. 50–55.
- Sayyad S.A.F., Chaudhari S., and Panda B. Quantitative determination of ethanol in arishta by using UV-visible spec- trophotometer. Pharmaceutical and Biological Evaluations, 2015, vol. 2, no. 5, pp. 204–207.
- Chen Y., Sharma-Shivappa R.R., Keshwani D., and Chen C. Potential of Agricultural Residues and Hay for Bioe- thanol Production. Applied Biochemistry and Biotechnology, 2007, vol. 142, no. 3, pp. 276–290. DOI: https://doi. org/10.1007/s12010-007-0026-3.
- Menezes E.G., do Carmo J.R., Alves J.G.L., et al. Optimization of alkaline pretreatment of coffee pulp for production of bioethanol. Biotechnology Progress, 2014, vol. 30, no. 2, pp. 451–462. DOI: https://doi.org/10.1002/btpr.1856.
- Yang B. and Wyman C.E. Effect of xylan and lignin removal by batch and flowthrough pretreatment on the enzymatic digestibility of corn stover cellulose. Biotechnology and Bioengineering, 2004, vol. 86, no. 1, pp. 88–98. DOI: https:// doi.org/10.1002/bit.20043.
- Thanonkeo P. The batch ethanol fermentation of jerusalem artichoke using Saccharomyces cerevisiae. KMITL Science and Technology Journal, 2007, vol. 7, no. 2, pp. 93–96.
- Ballesteros M., Oliva J., Negro M., Manzanares P., and Ballesteros I. Ethanol from lignocellulosic materials by a simultaneous saccharification and fermentation process (SFS) with Kluyveromyces marxianus CECT 10875. Process Biochemistry, 2004, vol. 39, no. 12, pp. 1843–1848. DOI: https://doi.org/10.1016/j.procbio.2003.09.011.
- Bonilla-Hermosa V.A., Duarte W.F., and Schwan R.F. Utilization of coffee by-products obtained from semi-washed process for production of value-added compounds. Bioresource technology, 2014, vol. 166, pp. 142–150. DOI: https:// doi.org/10.1016/j.biortech.2014.05.031.
- Elias L. Chemical composition of coffee-berry by-products. In: Braham J.E. and Bressani R. (eds) Coffee pulp; com- position, technology, and utilization. Canada, Ottawa: Institute of Nutrition of Central America and Panama Publ., 1979. pp. 11–16.
- Gurram R., Al-Shannag M., Knapp S., et al. Technical possibilities of bioethanol production from coffee pulp: a re- newable feedstock. Clean Technologies and Environmental Policy, 2016, vol. 18, no. 1, pp. 269–278. DOI: https:// doi.org/10.1007/s10098-015-1015-9.
- Gouvea B., Torres C., Franca A., Oliveira L., and Oliveira E. Feasibility of ethanol production from coffee husks. Biotechnology letters, 2009, vol. 31, no. 9, pp. 1315–1319. DOI: https://doi.org/10.1007/s10529-009-0023-4.
- Palonen H. Role of lignin in the enzymatic hydrolysis of lignocellulose. Espoo, Finland: VTT Publ., 2004. 84 p.
- Zhang S., Maréchal F., Gassner M., et al. Process modeling and integration of fuel ethanol production from lignocel- lulosic biomass based on double acid hydrolysis. Energy & fuels, 2009, vol. 23, no. 3, pp. 1759–1765. DOI: https:// doi.org/10.1021/ef801027x.
- Wiselogel A., Tyson S., and Johnson D. 6: Biomass feedstock resources and composition. In: Wyman C.E. (ed) Hand- book on Bioethanol: Production and Utilization. CRC Press Publ., 2018, pp. 105–118.
- Goyal H.B., Saxena R.C., and Seal D. 3: Thermochemical conversion of biomass to liquids and gaseous fuels. In: Pandey A. (ed) Handbook of Plant-Based Biofuels. CRC Press Publ., 2008, pp. 29–43.
- Gnansounou E. 5: Fuel ethanol. Current status and outlook. In: Pandey A. (ed) Handbook of Plant-Based Biofuels.CRC Press Publ., 2008, pp. 57–71.
- Sun Y. and Cheng J. Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresource Technology, 2002, vol. 83, no. 1, pp. 1–11. DOI: https://doi.org/10.1016/s0960-8524(01)00212-7.
- Wang Z. and Cheng J.J. Lime pretreatment of coastal bermudagrass for bioethanol production. Energy & Fuels, 2011, vol. 25, no. 4, pp. 1830–1836. DOI: https://doi.org/10.1021/ef2000932.
- Kim S. and Holtzapple M.T. Lime pretreatment and enzymatic hydrolysis of corn stover. Bioresource Technology, 2005, vol. 96, no. 18, pp. 1994–2006. DOI: https://doi.org/10.1016/j.biortech.2005.01.014.
- Fan L.T., Gharpuray M.M., and Lee Y.H. 2: Nature of cellulosic material. In: Fan L.T. (ed) Cellulose hydrolysis.Springer Publ, 1987, pp. 5–20.
- Tarkow H. and Feist W. A. Mechanism for Improving Digestibility of Lignocellulosic Materials with Dilute Alkali and Liquid Ammonia. Advances in Chemistry Series, 1969, vol. 95, pp. 197–218. DOI: https://doi.org/10.1021/ba- 1969-0095.ch012.
- Zhao X., Zhou Y., Zheng G., and Liu D. Microwave pretreatment of substrates for cellulase production by solid-state fermentation. Applied Biochemistry and Biotechnology, 2010, vol. 160, no. 5, pp. 1557–1571. DOI: https://doi. org/10.1007/s12010-009-8640-x.
- Xu J., Chen H., Kádár Z., et al. Optimization of microwave pretreatment on wheat straw for ethanol production. Biomass and Bioenergy, 2011, vol. 35, no. 9, pp. 3859–3864. DOI: https://doi.org/10.1016/j.biombioe.2011.04.054.
- McIntosh S. and Vancov T. Optimisation of dilute alkaline pretreatment for enzymatic saccharification of wheat straw. Biomass and Bioenergy, 2011, vol. 35, no. 7, pp. 3094–3103. DOI: https://doi.org/10.1016/j.biombioe.2011.04.018.
- Martı́n C., Galbe M., Wahlbom C.F., Hahn-Hägerdal B., and Jönsson L.J. Ethanol production from enzymatic hydro- lysates of sugarcane bagasse using recombinant xylose-utilising Saccharomyces cerevisiae. Enzyme and Microbial Technology, 2002, vol. 31, no. 3, pp. 274–282. DOI: https://doi.org/10.1016/s0141-0229(02)00112-6.
- Silverstein R.A., Chen Y., Sharma-Shivappa R.R., Boyette M.D., and Osborne J. A comparison of chemical pre- treatment methods for improving saccharification of cotton stalks. Bioresource Technology, 2007, vol. 98, no. 16, pp. 3000–3011. DOI: https://doi.org/10.1016/j.biortech.2006.10.022.
- Ooshima H., Aso K., Harano Y., and Yamamoto T. Microwave treatment of cellulosic materials for their enzymatic hydrolysis. Biotechnology Letters, 1984, vol. 6, no. 5, pp. 289–294. DOI: https://doi.org/10.1007/bf00129056.
- Lu X., Xi B., Zhang Y., and Angelidaki I. Microwave pretreatment of rape straw for bioethanol production: focus on energy efficiency. Bioresource Technology, 2011, vol. 102, no. 17, pp. 7937–7940. DOI: https://doi.org/10.1016/j. biortech.2011.06.065.
- Chen M., Zhao J., and Xia L. Enzymatic hydrolysis of maize straw polysaccharides for the production of reducing sug- ars. Carbohydrate Polymers, 2008, vol. 71, no. 3, pp. 411–415. DOI: https://doi.org/10.1016/j.carbpol.2007.06.011.
- Belkacemi K., Turcotte G., and Savoie P. Aqueous/steam-fractionated agricultural residues as substrates for etha- nol production. Industrial & Engineering Chemistry Research, 2002, vol. 41, no. 2, pp. 173–179. DOI: https://doi. org/10.1021/ie0102246.
- Öhgren K., Bura R., Lesnicki G., Saddler J., and Zacchi G. A comparison between simultaneous saccharification and fermentation and separate hydrolysis and fermentation using steam-pretreated corn stover. Process Biochemistry, 2007, vol. 42, no. 5, pp. 834–839. DOI: https://doi.org/10.1016/j.procbio.2007.02.003.
- Sahu O. Bioethanol production by coffee husk for rural area. Advanced Research Journal of Biochemistry and Bio- technology, 2014, vol. 1, pp. 1–5.
- Kefale A., Redib M., and Asfaw A. Bioethanol Production and Optimization test from Agricultural Waste: The case of wet coffee processing waste (pulp). International Journal of Renewable Energy Research, 2012, vol. 2, pp. 446–450.
- Yoswathana N., Phuriphipat P., Treyawutthiwat P., and Eshtiaghi M.N. Bioethanol production from rice straw. Energy Research Journal, 2010, vol. 1, no. 1, pp. 26–31. DOI: https://doi.org/10.3844/erjsp.2010.26.31.