Аннотация
Introduction. Extract of goji berries (Lycium Barbarum L.) and buckwheat flour (Fagopýrum esculéntum L.) possess antioxidant and antimicrobial properties. As a result, they can be used to improve traditional Kazakh horse-meat formulations to obtain functional cooked and smoked meat products. These natural biologically active substances can improve the oxidative stability of pigments, lipids, and proteins of finished products. The research objective was to assess the potential of goji extract and buckwheat flour as additives that can improve the oxidative stability and general quality of Kanagat, a national Kazakh cooked and smoked horse-meat product. Goji extract and buckwheat flour were used in two concentrations – 0.5% and 1.0%.Study objects and methods. The research featured sensory evaluation of taste, smell, color, determination of color parameters (L*, a*, b*), pH, free amine nitrogen, total carbonyl proteins, acid value, peroxide value and thiobarbituric acid reactive substances (TBARS), as well as a histological analysis.
Results and discussion. When 1.0% of goji extract and 1.0% of buckwheat flour were added to the traditional formulation, it improved the oxidative stability and quality of the modified horse-meat product while preserving its sensory properties and colour parameters. A set of microstructural studies showed that the processing of meat products with 1.0% of goji extract and 1.0% of buckwheat flour had a destructive effect on most fibers. The affected fibers showed multiple decays of myofibrillar substance, which turned into a finegrained protein mass. The abovementioned concentration caused effective inhibition of hydrolytic changes, as well as oxidation of proteins and lipids.
Conclusion. The new technology made it possible to produce a new national horse-meat product fortified with 1.0% of goji extract and 1.0% of buckwheat flour. The specified amount of biologically active additives improved the oxidative stability and quality of the product, while maintaining its sensory and color characteristics.
Ключевые слова
Meat industry, meat products, hydrolysis, oxidation, goji, buckwheat flourВВЕДЕНИЕ
The socio-technological development of meat industry pursues two main goals. First, meat industry enterprises and research institutions should satisfy consumer demands. Second, they should develop and produce high-quality functional products that are environmentally safe and beneficial for human health in biomedical terms.
To function properly, human body needs a healthy, nutritious, and well-balanced diet. It is becoming more and more difficult to provide food that would satisfy this requirement because of constantly decreasing resources, modern lifestyle, environmental pollution, and overall degradation of food quality [1–3].
Horse meat has a high nutritional value. Its protein content is 18–25%, which is quite high. In addition, the proteins of horse muscle tissue are rich in essential amino acids, which are represented in the optimal ratio. Horse meat provides vitamins B. It is a source of such major mineral elements as magnesium and chlorine. These minerals are known to improve blood buffering and regulation of blood pressure. Almost all vitamins and minerals found in meat are more easily digestible than those found in products of plant origin. Horse meat is rich in vital vitamins and mineral elements that help to improve metabolism in patients with obesity, atherosclerosis, and hypertension. Horse meat is alsogood for people suffering from cardiovascular, hepatic, and pancreatic diseases [1].
In addition, horse-meat fat has a unique fatty acid composition [1]. Adding functional ingredients, e.g. natural antioxidants, is one of the strategies of developing functional national meat products [4]. This strategy has already provided a number of functional meat foods [5]. Improved horse-meat products injected with multicomponent curing solution have already been in the focus of some studies [6]. However, the process of obtaining national meat products from horse meat remains largely understudied.
There are many natural extracts and flours that can be used in functional food production. Goji berries (Lycium barbarum L.) have recently become one of the most popular plants with such properties [7]. Goji berries contain free amino acids, e.g. proline, taurine, and betaine with its anti-aging effect, as well as gamma-aminobutyric acid, phenylpropanoids, flavonoids, and polyphenolic compounds. They are also rich in vitamins, primarily thiamine, riboflavin, and ascorbic acid (vitamin C). Unfortunately, dry berries contain much less ascorbic acid than fresh ones. In addition, goji berries contain zinc, iron, copper in trace amounts, and some oil. Goji juice is known to contain seven different flavonols. Most of them have isohamnetin 3-O-glycosides, but they are poor radical absorbents. Quercetin, 3-O-glycosides, catechins, and hydroxybenzoic acids with catechin structure are strong antioxidants. Unfortunately, their concentration in the juice proved insignificant. It is ascorbic acid that proved to be the main antioxidant in goji berry juice [7–10].
Goji berries demonstrate antioxidant and antibacterial activities against Bacillus cereus, Bacillus coagulans, Bacillus subtilis, Listeria monocytogenes , and Yersinia enterocolitica. Goji extract also exhibits immunomodulating properties and can inhibit chromium-induced production of free radicals, apoptosis, and DNA fragmentation. In addition, goji extract has pronounced cytoprotective properties and can restore the antioxidant status of cells [11, 12].
Goji berries are a powerful hepaprotector. They contain cerebrosides, i.e. natural organic compounds from the group of complex lipids that protect liver cells from toxic chemicals. They are even more beneficial for human liver than such well-known hepatoprotector as milk thistle (Silybum marianum L.). Pyrrole is another hepatoprotective compound found in goji berries. Its rather unusual molecules contain a nitrogen atom in their central ring. Pyrrole proved superior to goji cerebrosides in hepaprotection [13].
The list of the most famous antioxidants involves tocopherols (vitamin E), carotenoids (vitamin A), and ascorbic acid (vitamin C). Vitamin C is believed to be the most important of them. As it was mentioned above, goji berries are rich in these vitamins. Some studies showed that goji antioxidants are five times stronger than those found in prunes and more than 25 times stronger than antioxidants found in broccoli. Surprisingly, broccoli was considered the undisputed record holder among antioxidant plants until very recently. Broccoli is still on the list of the so-called superfoods.
European scientists have compiled a table of the ORAC index, i.e. Oxygen Radical Absorbance Capacity. This is an indicator of the ability of antioxidants to absorb free radicals. According to this table, goji berries are the most powerful antioxidant in the world. The daily human need is about 5000 ORAC units, whereas 100 g of goji berries contains 25300 ORAC units [14].
Buckwheat flour (Fagopýrum esculéntum L.) is another interesting component that can be used in formulations of functional national meat products. Its popularity in food science is associated with flavonoids. Buckwheat flour flavonoids prevent the development of malignant tumors, protect human body from aging and disease, and boost immune system. Buckwheat grains, and hence buckwheat flour, do not contain gluten, which means that buckwheat products can be consumed by patients with celiac disease. Bakery from buckwheat flour helps to make their diet diverse [15].
The chemical composition of buckwheat flour also contains rutin, which is a very useful flavonoid. It gives buckwheat useful properties for the cardiovascular system. This fragrant flour lowers blood pressure by expanding blood vessels. Ground buckwheat prevents excessive platelet formation, lowers cholesterol, and saturates blood with oxygen. Buckwheat flour is good for blood circulation, as it decreases the permeability of blood vessels. In addition, buckwheat flour is rich in rutin, which makes it useful for people with varicosis and gout, as well as for those who have undergone radiation treatment [15].
Buckwheat prevents development of gallstones and regulates bile acid secretion. This product is known for its ability to strengthen and cleanse intestines; it also helps against chronic diarrhea and dysentery. Buckwheat flour improves the absorption of calcium, thus strengthening bone tissue and preventing osteoporosis. It is very good for nervous system and improves brain function. In addition, it boosts immune system and metabolism. Buckwheat flour is rich in vitamins, which makes is good for hair, nails, and skin. Finally, this product improves food absorption and has a beneficial effect on the pancreas [15].
As it was already mentioned, buckwheat is rich in rutin, which cannot be produced by human body. Rutin enters the body with food products and improves the elasticity and strength of blood vessels, thereby reducing the risk of hypertension. Regular consumption of buckwheat flour products can significantly lower blood sugar levels. Buckwheat flour is also rich in high-grade proteins and complex carbohydrates that provide body with energy [16].
Buckwheat flour is rich in vitamins, minerals, and plant proteins. It contains vital amino acids, natural antioxidants, and dietary fiber. Buckwheat flour contains neither harmful carbohydrates nor gluten. Other beneficial effects of buckwheat flour manifest themselves in that it removes wastes, toxins, and other harmful substances, produces a powerful general tonic effect on human health, activates metabolism, improves cardiovascular system, and lowers blood sugar [16].
Our research objective was to establish the potential of goji extract and buckwheat flour for improving oxidative stability and general quality of meat products. The substances were used as additives in the amounts of 0.5% and 1.0% to produce a functional Kazakh horsemeat product called Kanagat.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
Kanagat is a national Kazakh horse-meat product of new generation. It was produced in the processing department of the limited liability partnership AF Kaynar (Almaty, Kazakhstan) from first-category chilled horse meat. The upper layer of muscle tissue was trimmed from the hip part of the carcass together with the superficial fat layer. The first-category chilled horse meat was cut into pieces of ≤ 0.4 g and about 10 cm thick.
15% of curing solution was injected into the meat pieces by weight of the raw material with a special injector intended for pickle pumping. The amount of curing ingredients in the curing solution corresponded to the addition of 2.5 kg of salt and 150 g of sugar per 100 kg of raw meat. 2.5–5.0 kg of goji extract or buckwheat flour was added to the curing solution meant for test samples. The cured meat was massaged in a TUZ-KZ tenderizer of ETDU brand for 40 min at 0–4°C. After massaging, the meat was cut into 0.1 kg pieces with a thickness of ≤ 5 cm and coated with a waterproof material. After that, the meat underwent heat treatment in a multi-purpose heat chamber. The product was then boiled at 74–75°C for 2–2.5 h until the temperature in the center of each piece reached 72°C. The cooked product was cooled and then smoked for 30 min at 40°C. The finished Kanagat was cooled to 10–12°C, vacuumpackaged, and stored for 21 days at 0–4°C.
The research featured five samples. For the control sample, 15% of curing solution was introduced into pieces of horse meat, as described above. The test samples were injected with 15% of curing solution that contained 2.5 kg of goji extract per 100 kg (which was equivalent to 0.5%-concentration in the finished product), 5.0 kg of goji extract (1.0%), 2.5 kg of buckwheat flour (0.5%), and 5.0 kg of buckwheat flour (1.0%).
The goji extract (Lycium barbarum L.) was supplied by Dannie Chen Shaanxi Jintai Biological Engineering Co., Ltd. (Xi’an, Shaanxi, China). The buckwheat flour (Fagopýrum esculéntum L.) was produced by the Scientific Developpment and Production Center “Kudesnitsa” of the company “Aladushkin Grupp” (St. Petersburg, Russia).
The sensory properties of the samples were determined by five panelists with certified tasting abilities. The panelists passed a triangular test to differentiate the aroma, smell, and color of fresh and rancid sausage. The samples were evaluated using a 1-to-5 scale [17].
A Konica Minolta CR-410 colorimeter (Konica Minolta Holding, Inc., Ewing, NJ, USA) was used to estimate lightness (L*), redness (a*), and yellowness (b*) [17].
Free amine nitrogen was determined using a modified Serensen titration method [18].
Protein oxidation was measured by evaluating the formed carbonyl groups [19].
As a standard for fat hydrolysis rate, the acid value of extracted lipids was measured as specified in ENISO 660:2001 [19].
The standard IDF method was used to determine the peroxide values of the meat. The test used all lipids extracted from the samples [18].
As for the substances of 2-thiobarbituric acid reagent, TBARS were determined by the method described by Botsoglou et al. [17]. The research employed a UV-VIS Camspec M550 dual-beam spectrophotometer (Camspec Ltd, Cambridge, UK). The pH of the samples was determined using a Microsyst MS 2004 pH-meter (Mikrosist, Plovdiv, Bulgaria). The pH-meter was equipped with a combined pH electrode and a Sensorex S450CD combined recorder (Sensorex pH electrode station, Garden Grove, California, USA) [20].
High performance liquid chromatography (HPLC) with a coulometric electrochemical detector was used to analyze oil-soluble antioxidants extracted from the goji berries and the buckwheat flour and their concentrations in the horse meat [21, 22].
The method of ISO 4833:2003 was used to prepare the samples for microbiological analysis and total microscopic count of facultative anaerobic mesophilic microorganisms [23].
The data obtained from different samples were independently analyzed using SAS software [17]. Multiple Student-Newman-Keuls tests were used to compare the differences between means. Mean values and standard mean errors were calculated. The significance of differences was determined at P ≤ 0.05.
The histological studies of the Kanagat were performed in accordance with the classical microstructural analysis and standard methods. Histological sections were made using a MICROM HM-525 cryostat microtome (CarlZeiss, Germany) [24, 26].
The following method of short-term additional fixation was used for the sections mounted on the slide. An 8% formalin solution was applied to the histological section for 30 min. After that, the section was thoroughly washed with water for 3 min, dried at room temperature, and stained with hematoxylin and eosin. The histological preparations were studied and photographed using an AxioImaigerA1 light microscope (CarlZeiss, Germany) and an AxioCamMRc5 video camera. Image processing and morphometric studies were performed using the AxioVision 4.7.1.0 computeraided image analysis system adapted for histological studies. To obtain reliable results, the experiments were performed in triplicates with 3–5 replications of the analyses of each sample for all parameters.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
The samples were stored at 0–4°C for 21 days. On day 21, the concentrations of antioxidants extracted from the goji extract and buckwheat flour of Kanagat were determined as follows: the samples with 0.5 and 1.0% of goji extract – 4.78 ± 0.21 and 9.81 ± 0.26 mg/g and the samples with 0.5 and 1.0% of buckwheat flour – 4.73 ± 0.19 and 9.75 ± 0.20 mg/g, respectively.
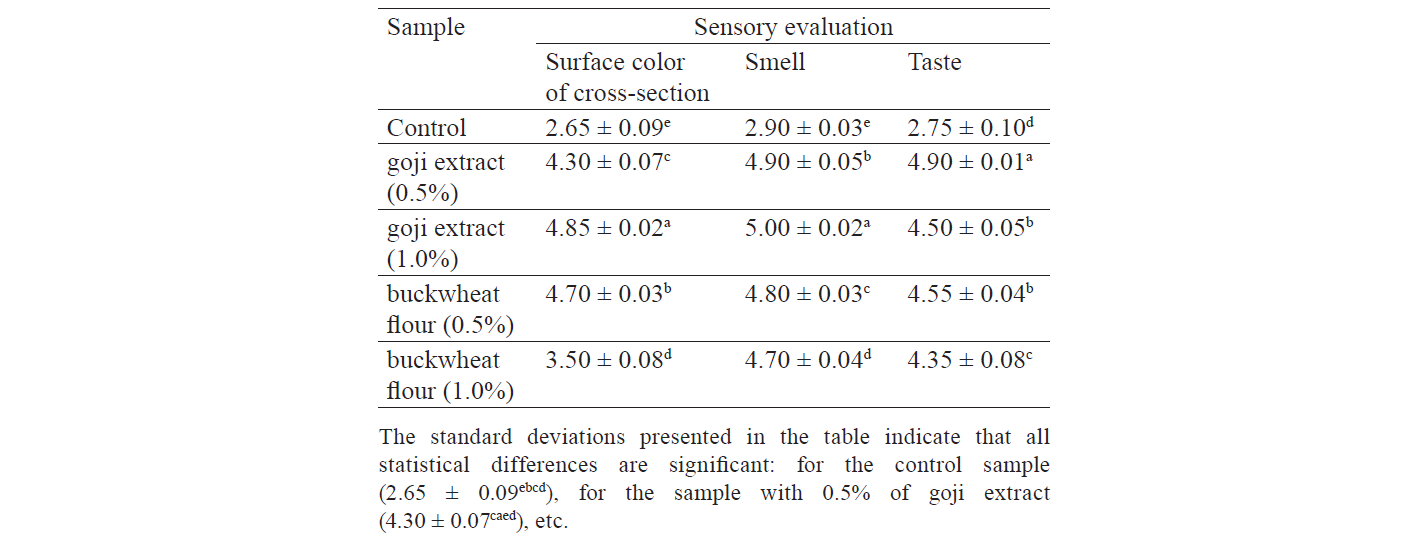
Sensory evaluation. The samples with 1.0% of goji extract received the highest sensory indices for taste, smell, and color after 21 days of storage at 0–4°C (Table 1). The samples with 0.5% of goji extract and 0.5% of buckwheat flour gotalmost the same results (Table 1). The control sample showed the worst sensory properties. It scored significantly lower (P ≤ 0.05) than the other samples. Therefore, 2.5% of goji extract added to the curing solution preserved the fresh color and especially aroma of the vacuum-packaged horse-meat product after 21 days of storage. A similar research also reported the positive effect of a mix of dried goji berries and pumpkin powder on the quality and storage stability of cooked and smoked beef tenderloin [8].
Color characteristics. Table 2 demonstrates the changes in lightness (L*), redness (a*), and yellowness (b*). The samples with 0.5% and 1.0% of goji extract again showed the most significant changes. The obtained results were consistent with sensory evaluation. They proved that goji extract produced a better effect on the color characteristics of the restructured horse meat than buckwheat flour.
Oxidative stability and quality. After 21 days of storage, the modified horse-meat samples revealed the following changes. The content of free amine nitrogen in all test samples was significantly lower (P ≤ 0.05) than in the control samples. The samples with 0.5% and 1.0% of goji extract had the lowest content of free amine nitrogen. The content of carbonyl proteins increased in all samples after 21 days of storage at 0–4°C. This process was significantly slower (P ≤ 0.05) in the samples with 1.0% of goji extract and 1.0% of buckwheat flour, where the total content of protein carbonyls decreased by 51 and 36% (Table 3).
Acidity values increased significantly (P ≤ 0.05) in all samples during 21 days of refrigerated storage. The lipolytic changes were lower by 38% in the samples with 1.0% of goji extract and 1.0% of buckwheat flour and by 28% in the samples with 0.5% of goji extract and 0.5% of buckwheat flour, if compared with the control sample. Similar changes were registered in peroxide value and TBARS. Primary products of lipid oxidation (lipid hydroperoxides) showed a significant decrease (P ≤ 0.05) by 24% in the samples with 1.0% of goji extract and 1.0% of buckwheat flour and by 17% in the samples with 0.5% of goji extract and 0.5% of buckwheat flour. Secondary products of lipid oxidation (TBARS) decreased by 53% in the samples with 1.0% of goji extract and 1.0% of bucruheat flour and by 44% in the samples with 0.5% of goji extract and 0.5% of buckwheat flour.
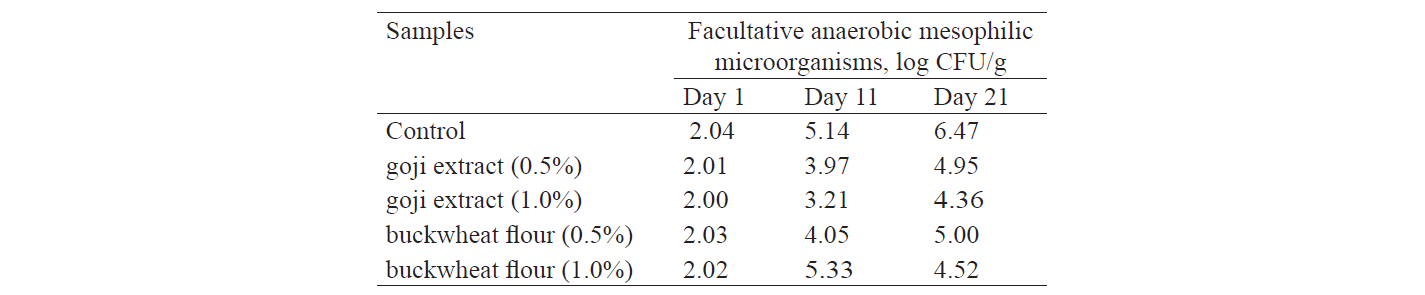
As for the comparison of pH value, samples with 0.1% and 0.5% of goji extract and buckwheat flour s howed a small (1.3–2.6%) but significant (P ≤ 0.05) increase after 21 days of storage. Unlike the control samples, the test samples demonstrated a statistically significant decrease in pH by 11.8%. The conclusions were confirmed by the results obtained for the total count of facultative anaerobic mesophilic microorganisms in the vacuum-packaged samples after 21 days of refrigerated storage (Table 4).
Histological analysis is widely used to determine the condition of raw materials and products, as well as their real composition. The analysis makes it possible to study the structure of the product as a whole together with the changes in its parts and components. It detects the presence of various tissues and cellular structures and their quantity in the product [24, 25].
The method of histological analysis is widely used in biology and medicine. However, in this study it was applied to the national cooked and smoked horse-meat product “Kanagat” after it had undergone thermal treatment and other types of technological impact [26].
In this research, histological analyses were performed in order to determine the effect of goji extract (Lycium barbarum L.) and buckwheat flour (Fagopýrum esculéntum L.) on the muscle and connective tissue of Kanagat. According to the microstructural analysis, the control sample consisted of large fragments of muscle, connective, and adipose tissue of 0.7–1.4 μm (Fig. 1).
The muscle fibers were straight, swollen, tightly adjacent to each other, and quite often fragmented. A fine-grained protein mass that formed as a result of mechanical action on muscle tissue during the grinding was spread between the coarse-grained structural elements. The fine-grained protein mass revealed particles of spices and fat drops of 12–100 μm in size, which were uniformly distributed over the mass of the sample. The surface coagulation layer adhered tightly to the coating. Bundles of muscle fibers that retained their integrity were so tightly adjacent to each other and swollen that the boundaries between them were difficult to detect. The transverse striation was wide and visible in occasional fibers. However, the bulk of muscle fibers had a homogeneous structure, with some disintegration and violation of the direction of myofibrils to one another. The nuclei of the fiber were homogeneous. Destructive changes were spotted in the form of individual microcracks.
As for the experimental samples, the coarse-grained structural components were in a fine-grained protein mass that included fragments of plant components, i.e. buckwheat flour, goji extract, and spices. The layout of the sample was dense, with no large cracks or cavities loosening the mass of the sample. The structural components of meat were closely interconnected. The fine-grained protein mass was penetrated by roundshaped microcapillaries of 250–350 μm in size (Fig. 2).
The fragments of muscle tissue that retained their microstructural features demonstrated swollen muscle fibers. The boundaries between them were hardly discernible. The transverse striation was either poor or not detected in some parts of the sample.
Destructive changes were multiple. The destruction degree of the fibers was greater than in the control samples. The fiber nuclei were homogeneous or shadowlike. Microflora was detected as a fine-grained protein mass in the form of small microcolonies of 0.2–0.3 μm. Microflora was diffuse between the fibers, under the sarcolemma, in the areas of fiber destruction, and in connective tissue layers. The layout of the structural elements was dense. The vacuoles were 70–300 μm in size, had clearly defined boundaries, and occasionally merged with each other.
1.0% of goji extract and 1.0% buckwheat flour accelerated the destructive changes in the main structural elements of meat, and, consequently, boosted its secondary structure formation. The samples with goji extract and buckwheat flour had a greater degree of swelling and destruction of muscle fibers. The destructive changes covered most fibers and were detected as multiple decays of myofibrillar substance, which turned into a fine-grained protein mass.
The intensive formation of the fine-grained protein mass contributed to the development of a compact monolithic mass of meat pieces, which formed a dense space framework after heat treatment. Unlike meat products developed according to traditional technologies, the pieces of meat in the test samples were more compact and less porous. There were fewer vacuoles, and they were smaller.
The microstructural studies showed that 1.0% of goji extract and 1.0% of buckwheat flour caused destructive changes in most fibers. The affected fibers showed multiple decays of myofibrillar substance, which turned into a fine-grained protein mass. This, in turn, contributed to the development of monolithic structure.
ВЫВОДЫ
Injecting 1.0% of buckwheat flour (Fagopýrum esculéntum L.) or 1.0% of goji extract (Lycium barbarum L.) into horse meat resulted in a functional national cooked and smoked horse-meat product with 1% of biologically active substances. This concentration inhibited lipolytic changes and oxidation of proteins and lipids. It also improved the oxidative stability and quality of the new national horse-meat product, while maintaining its sensory properties.
КОНФЛИКТ ИНТЕРЕСОВ
The authors declare that there is no conflict of interests regarding the publication of the present article.
ФИНАНСИРОВАНИЕ
The present study was part of research work No. 0457 “Study of the functional and bio-correcting characteristics of plantanimal complexes and the development on their basis of technology of functional national meat products using local raw materials”. The project was funded by the Ministry of Education and Science of the Republic of Kazakhstan.СПИСОК ЛИТЕРАТУРЫ
- Lorenzo JM, Sarries MV, Tateo A, Polidori P, Franco D, Lanza M. Carcass characteristics, meat quality and nutritional value of horsemeat: A review. Meat Science. 2014;96(4):1478–1488. DOI: https://doi.org/10.1016/j.meatsci.2013.12.006.
- Kozyrev IV, Mittelshtein TM, Pchelkina VA, Kuznetsova TG, Lisitsyn AB. Marbled beef quality grades under various ageing conditions. Foods and Raw Materials. 2018;6(2):429–437. DOI: https://doi.org/10.21603/2308-4057-2018-2-429-437.
- Lisitsyn AB, Chernukha IM, Lunina OI. Fatty acid composition of meat from various animal species and the role of technological factors in transisomerization of fatty acids. Foods and Raw Materials. 2017;5(2):54–61. DOI: https://doi.org/10.21179/2308-4057-2017-2-54-61.
- Comert ED, Gokmen V. Evolution of food antioxidants as a core topic of food science for a century. Food Research International. 2018;105:76–93. DOI: https://doi.org/10.1016/j.foodres.2017.10.056.
- Thangavelu KP, Kerry JP, Tiwari BK, McDonnell CK. Novel processing technologies and ingredient strategies for the reduction of phosphate additives in processed meat. Trends in Food Science & Technology. 2019;94:43–53. DOI: https://doi.org/10.1016/j.tifs.2019.10.001.
- Kaldarbekova M, Uzakov Y, Chernukha I, Kuznetsova O. Research of technology of restructured combined meat products using a multicomponent brine. EurAsian Journal of BioSciences. 2019;13(2):1625–1632.
- Sekinaeva MA, Serebryanaya FK, Denisenko ON, Lyashenko SS. The study of anatomical features of the herb Lycium (Lycium Barbarum L.). Advances in Current Natural Sciences. 2015;(9–2):231–235. (In Russ.).
- Serikkaisai MS, Vlahova-Vangelova DB, Dragoev SG, Uzakov YM, Balev DK. Effect of dry goji berry and pumpkin powder on quality of cooked and smoked beef with reduced nitrite content. Advance Journal of Food Science and Technology. 2014;6(7):877–883.
- Menchetti L, Brecchia G, Branciari R, Barbato O, Fioretti B, Codini M, et al. The effect of Goji berries (Lycium barbarum) dietary supplementation on rabbit meat quality. Meat Science. 2020;161. DOI: https://doi.org/10.1016/j.meatsci.2019.108018.
- Mocan A, Cairone F, Locatelli M, Cacciagrano F, Carradori S, Vodnar DC, et al. Polyphenols from Lycium barbarum (Goji) fruit european cultivars at different maturation steps: extraction, HPLC-DAD analyses, and biological evaluation. Antioxidants. 2019;8(11). DOI: https://doi.org/10.3390/antiox8110562.
- Lu S, Ji H, Wang QL, Li BK, Li KX, Xu CJ, et al. The effects of starter cultures and plant extracts on the biogenic amine accumulation in traditional Chinese smoked horsemeat sausages. Food Control. 2015;50:869–875. DOI: https://doi.org/10.1016/j.foodcont.2014.08.015.
- Hayrapetyan SA, Vardanyan LR, Vardanyan RL. Antioxidant activity of creeping thyme (Thymus Serpyllum L.) in cumene oxidation reaction. Proceedings of the Yerevan State University. Chemistry and Biology. 2013;(2):23–31.
- Vardanyan LR, Shutava HG, Ayrapetyan SA, Vardanyan RL, Agabekov VE, Reshetnikov VN. Quantitative content and activity of antioxidants in herbs of different climatic zones. Doklady of the National Academy of Sciences of Belarus. 2013;57(5):72–76. (In Russ.).
- Skenderidis P, Kerasioti E, Karkanta E, Stagos D, Kouretas D, Petrotos K, et al. Assessment of the antioxidant and antimutagenic activity of extracts from goji berry of Greek cultivation. Toxicology Reports. 2018;5:251–257. DOI: https://doi.org/10.1016/j.toxrep. 2018.02.001.
- Li J, Hossain MS, Ma HC, Yang QH, Gong XW, Yang P, et al. Comparative metabolomics reveals differences in flavonoid metabolites among different coloured buckwheat flowers. Journal of Food Composition and Analysis. 2020;85. DOI: https://doi.org/10.1016/j.jfca.2019.103335.
- Hyun JE, Kim H-Y, Chun J-Y. Effect of jeju’s tartary buckwheat (Fagopyrum tataricum) on antioxidative activity and physicochemical properties of chicken meat emulsion – type sausage. Journal of the Korean Society of Food Science and Nutrition. 2019;48(2):231–236. DOI: https://doi.org/10.3746/jkfn.2019.48.2.231.
- Young OA, West J, Hart AL, van Otterdijk FFH. A method for early determination of meat ultimate pH. Meat Science. 2004;66(2):493–498. DOI: https://doi.org/10.1016/S0309-1740 (03)00140-2.
- Skrinjar M, Kolar MH, Jelsek N, Hras AR, Bezjak M, Knez Z. Application of HPLC with electrochemical detection for the determination of low levels of antioxidants. Journal of Food Composition and Analysis. 2007;20(7):539–545. DOI: https://doi.org/10.1016/j.jfca.2007.04.010.
- Paleari MA, Moretti VM, Beretta G, Mentasti T, Bersani C. Cured products from different animal species. Meat Science. 2003;63(4):485–489. DOI: https://doi.org/10.1016/s0309-1740(02)00108-0.
- Qian D, Zhao YX, Yang G, Huang LQ. Systematic review of chemical constituents in the genus Lycium (Solanaceae). Molecules. 2017;22(6). DOI: https://doi.org/10.3390/molecules22060911.
- Falowo AB, Fayemi PO, Muchenje V. Natural antioxidants against lipid-protein oxidative deterioration in meat and meat products: A review. Food Research International. 2014;64:171–181. DOI: https://doi.org/10.1016/j.foodres.2014.06.022.
- Sekinaeva MA, Lyashenko SS, Yunusova SG, Ivanov SP, Sidorov RA, Denisenko ON, et al. About biologically active substances of Lycium barbarum L. Bulletin of the State Nikitsky Botanical Gardens. 2018;(129):92–95. (In Russ.). DOI: https://doi.org/10.25684/NBG.boolt.129.2018.12.
- Islam T, Yu XM, Badwal TS, Xu BJ. Comparative studies on phenolic profiles, antioxidant capacities and carotenoid contents of red goji berry (Lycium barbarum) and black goji berry (Lycium ruthenicum). Chemistry Central Journal. 2017;11:8. DOI: https://doi.org/10.1186/s13065-017-0287-z.
- Hvilya SI, Pchelkina VA, Burlakova SS. Application of the histological analysis for investigation of meat raw materials and finished products. Food Processing: Techniques and Technology. 2012;26(3):132–138. (In Russ.).
- Lisitsyn AB, Chernukha IM, Lunina OI. Functional food products: innovative methods of counterfeit detection. Production Quality Control. 2018;(4):13–18. (In Russ.).
- Fira A, Joshee N, Cristea V, Simu M, Hârța M, Pamfil D, et al. Optimization of micropropagation protocol for goji berry (Lycium barbarum L.). Bulletin of University of Agricultural Sciences and Veterinary Medicine Cluj-Napoca. Horticulture. 2016;73(2):141–150. DOI: https://doi.org/10.15835/buasvmcn-hort:12177.