Аннотация
Water is the most crucial thing to mankind and so its contamination by various agencies is posing a threat to the natural balance. So, in the present work, the efficiency of various adsorbents derived from plant waste, to remove different dyes from aqueous solution was evaluated. Parameters for study were contact time, concentration and pH. Various combinations of plant ashes were used for the study. It was found that adsorbent prepared from the combination of orange peels, pomegranate and banana peels ashes, exhibited good adsorption capacity for methylene blue, congo red and crystal violet. All these dyes were completely removed from the aqueous solution while methyl orange was not removed. Congo red was removed completely within 40 min of contact with the adsorbent while methyl orange took 3 hrs to be removed to the extent of 48% only. The adsorption coefficient of congo red was found to be 2.33 while value for methylene blue and crystal violet was 1 and 1.66 respectively. The characterization of adsorbent was done by Scanning Electron Microscopy and IR spectroscopy. SEM image revealed the surface of adsorbent to be made of differential pores. From the results it became evident that the low-cost adsorbent could be used as a replacement for costly traditional methods of removing colorants from water.Ключевые слова
Textile water, orange peels, pomegranate peels, adsorption, congo red, SEMВВЕДЕНИЕ
Water is one of the most imperative substances on the Earth. About 75% of our body consists of water. Water is used for such a wide variety of purposes like drinking, washing, bathing, as well as in agriculture and many others industries. According to World Health Organization (WHO) data, about 85% of rural population lacks potable drinking water. Currently, the water contamination is serious problem. About 80% of diseases in First world countries are associated with stained drinking water. In Second world countries, 15 million infants die annually due to poor hygiene, polluted drinking water, and malnutrition. Chemical impurities such as heavy synthetic fertilisers, industrial metals, dyes of textile industry, and poisonous minerals can cause hazardous effect on human and animal life. Since these particles are very small in size, they can penetrate into the ground water [1].
Purification of water is a tedious process that requires a number of stages [2]. Textile goods are the necessary need of individuals, while textile industry is of immense economic importance. There are 2324 textile industries that require using a number of dyes, additional chemicals, and sizing materials [3]. Different stages of technological processes of textile dyeing industry produce huge volumes of waste water. The waste water discharged from textile mill includes a large amount of concentrated industrial dyes.
Generally, dye stuffs are complex aromatic substances that are difficult to be removed. Methods used for dye removal include flocculation, chemical coagulation, chemical oxidation, photochemical degradation, membrane filtration, adsorption, as well as aerobic and anaerobic biological degradation. However, waste after removing dyes reduces light diffusion, affecting thus aquatic plants. In turn, it may be toxic to some aquatic animals [4]. Moreover, these methods are not cost effective and environmentally friendly. None of them is effective in complete removal of dye from wastewater [4]. Dyed water not only poses aesthetic problem, but also causes serious ecological problems, for example, it significantly impacts photosynthetic.
Modern studies show that adsorption with the help of activated carbon is a very efficient method to remove various organic compounds from the waste water [5]. Numerous researchers have searched alternative adsorbents deriving them from farming waste or natural materials to remove dyes from wastewater. Some of these alternatives are palm ash, orange peel ash, shale oil ash, pomelo (Citrus grandis L.) peel, fat-free soya, bottom ash, sunflower seed shells, mandarin peel, wheat husk guava leaf powder, as well as steel and fertiliser industries waste [6].
Enormous amounts of fruit peels are disposed, while they might be used in the interest of the environment. Agricultural wastes can be employed as a low-cost adsorbent for removal of dyes, such as methylene blue, crystal violet, methyl orange, and congo red, from aqueous solution [6]. Orange peel consists of a large amount of cellulose, hemi-cellulose, pectin, lignin, and other low molecular weight compounds, together with limestone. It can be used as an efficient and costeffective bio-adsorbent for removing dyes metals and organic pollutants from industrial wastewater [7–12]. Apart from the traditional methods, there are a number of recent studies on bioremediation [16–22].
Consequently, the aim of the study was to determine the effectiveness of the combination of plant ash in removing congo red, crystal violet, methylene blue, and methyl orange dyes from aqueous solution. The parameters studied were contact time, dye concentration and pH variation.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
Materials. Glassware and apparatus used: conical flasks, a round bottom flask, a volumetric flask, funnel measuring cylinders, beakers, pipettes, a condenser, a soxhlet apparatus, an electronic weighing balance, an oven, a muffle furnace, a magnetic stirrer pH meter, and a UV-visible spectrometer.
Chemical used: AgNO3, ethanol, double distilled water, methylene blue, congo red, crystal violet, and methyl orange.
Plants used: orange peels, pomegranate peels, banana peels, drumsticks, and pea pods.
Methods. To prepare peel extracts, peels of pomegranate, orange, banana, and drumstick tree obtained from local market or fruit stalls were cleaned with distilled water twice to remove dust and watersoluble impurities. After that, these were cut into small pieces, and kept for 2 days for proper drying. The dried material was powdered, and extraction was carried out in a Soxhlet apparatus using methanol as solvent.
Activated charcoal was obtained by putting the dried plant peels in the muffle furnace at 450–500°C and keeping the samples to constant weight.
The stock solution with a concentration of 0.1 g/L was prepared for different dyes. The different concentrations of the dye solutions were obtained from the stock solution by dilution method. Methylene blue, congo red, crystal violet, and methyl orange were used as adsorbates.
Kinetics study was performed as follows. 0.6 g of adsorbent was added into 250 mL conical flasks filled with 100 mL of diluted solutions (25–200 mg/L). The solutions were stirred constantly, and the concentration of dye at maximum wavelength was measured using a double beam UV-visible spectrometer. The capacity of dye adsorbed at time t, Qt (mg/g), was calculated by the given formula:
where At is concentration at time t, A0 is the initial concentration, v is volume of solution, and W is the weight of adsorbent used [13].
To study the dependence of initial concentration of dyes and contact time on the degree of removing dyes, 0.6 g of each sample (orange, banana, and pomegranate ash) was added to each 100 mL flask with various dyes having different concentrations. The solution was stirred on the magnetic stirrer at room temperature. The time required for complete adsorption was determined.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
Different dyes, namely, methylene blue, congo red, crystal violet, and methyl orange were taken to evaluate the adsorption capacity of the adsorbent.
According to Figs. 1–4, the effectiveness of dye removal increased with an increase in time. This might be due to the better interaction between dye molecules and those of activated charcoal. It was observed that the initially dye removal occurred faster and followed first order kinetics. This was proportional to the availability of active sites, and an equilibrium between adsorption and desorption was than established.
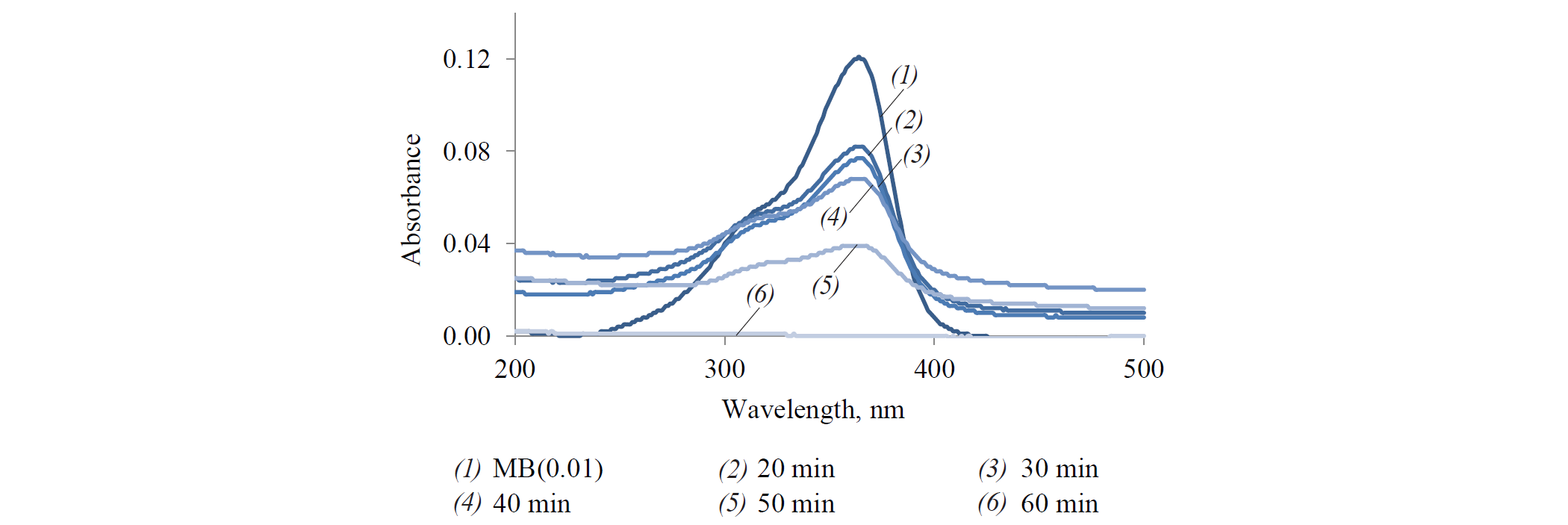
The absorbance of methylene blue at λmax (about 390 nm) decreased with increasing contact time (Fig. 1). The complete absorbance of methylene blue with the adsorbent took 60 min.
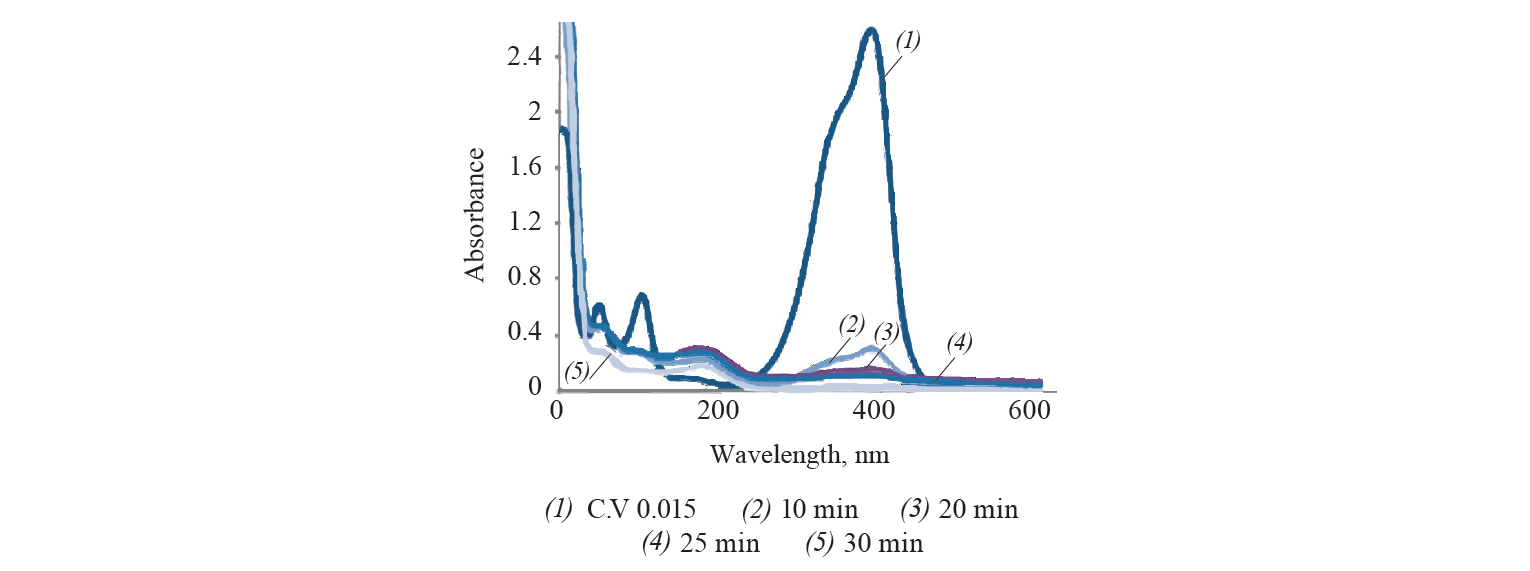
The variation of absorbance of crystal violet with time was studied by a UV-visible spectroscopy (Fig. 2). Crystal violet exhibited λmax at 390 nm. It was found that the dye was completely removed after 30 min of contact with adsorbent.
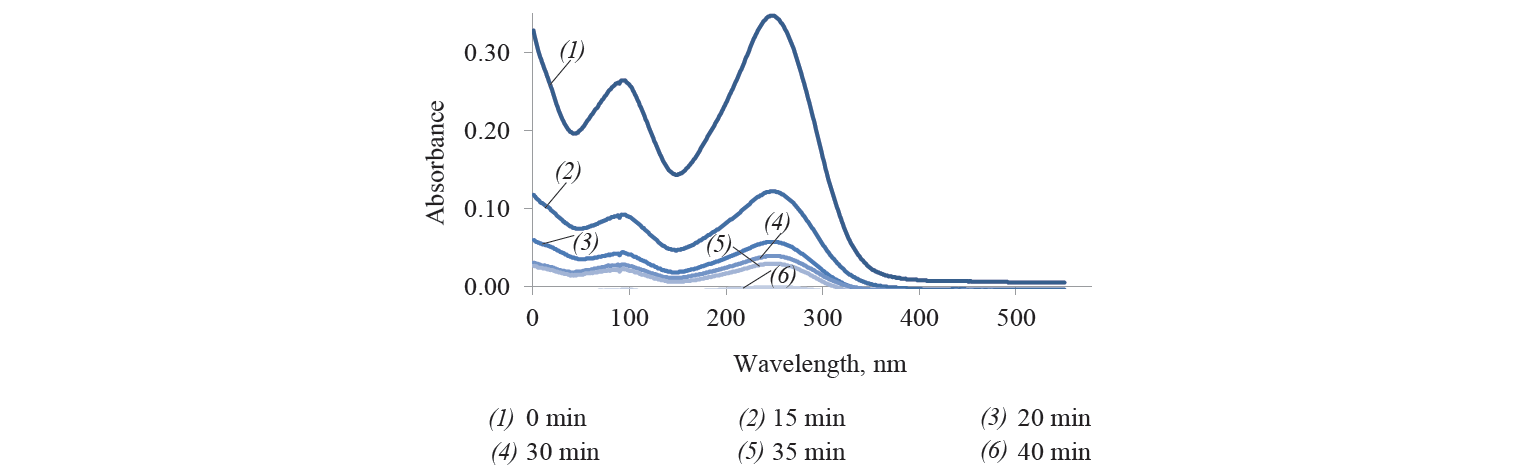
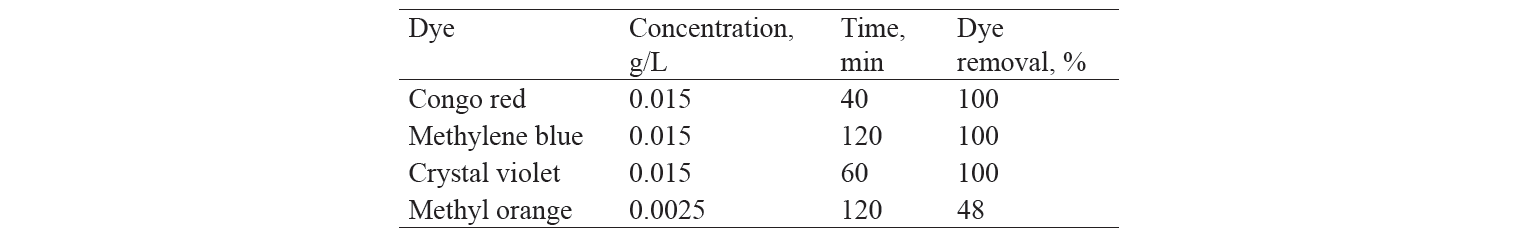
Similarly, congo red was completely removed in 40 min of contact with the adsorbent (Fig. 3).
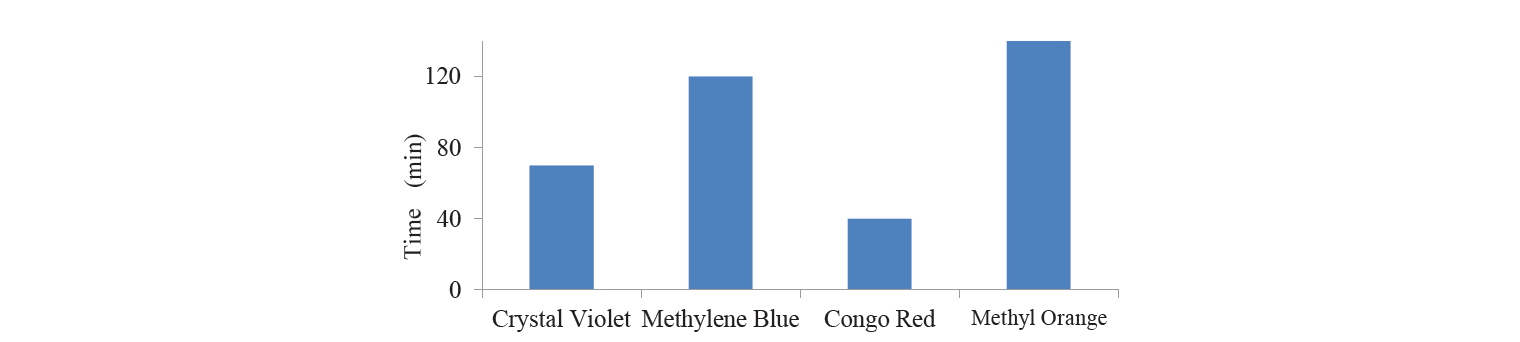
On the contrary, the adsorbent was not effective for methyl orange removal (Fig. 4). Even two hours of contact time was not enough to adsorb the dye. This could be due to the fact that methyl orange does not have any functionality that could make the Vander Waals’ interaction with the adsorbent.
The successful removal of various dyes by the combination of plant ashes proved the efficacy of the combination for bioremediation of textile water. As seen from Figure 5, complete dye removal took 5 h. The textile effluent water contained a large amount of heavy metals and different kinds of dyes, so it took longer for adsorbent to absorb the colourant.
Adsorption coefficient of activated charcoal for different dyes at time t. Adsorption coefficient was calculated as the amount of dye adsorbed with one gram of the adsorbent (mg/g). Adsorption coefficient was found to be different for each dye (Table 1) because adsorption depended upon the compatibility of the dye structure with the surface and the porosity of the adsorbent. It was found that absorption capacity for methyl orange was significantly lower, whereas that for congo red had maximum value at contact time of 30 min (Fig. 6).
Percentage of dyes adsorbed with adsorbent. The percentage of dye elimination indicated the efficiency of adsorbent (Table 2). The results made it possible to conclude that 100% of congo red was removed in 40 min, whereas the removal of only 48% of methyl orange took 120 min. In spite of the fact that both methyl orange and congo red dyes have similar structure, the percentage of their removal from the solution differs. One of the causes for that could be the presence of two primary amine groups in congo red, which could contribute to binding of dye with adsorbent. Thus, the structure of adsorbate played a crucial role in adsorption efficacy. The efficiency of adsorption depended upon the pore size of the adsorbent. The results of the study confirmed that the structure of congo red dye was wellmatched with the pore size of the adsorbent, which allowed it to exhibit fairly efficient adsorption (Table 2).
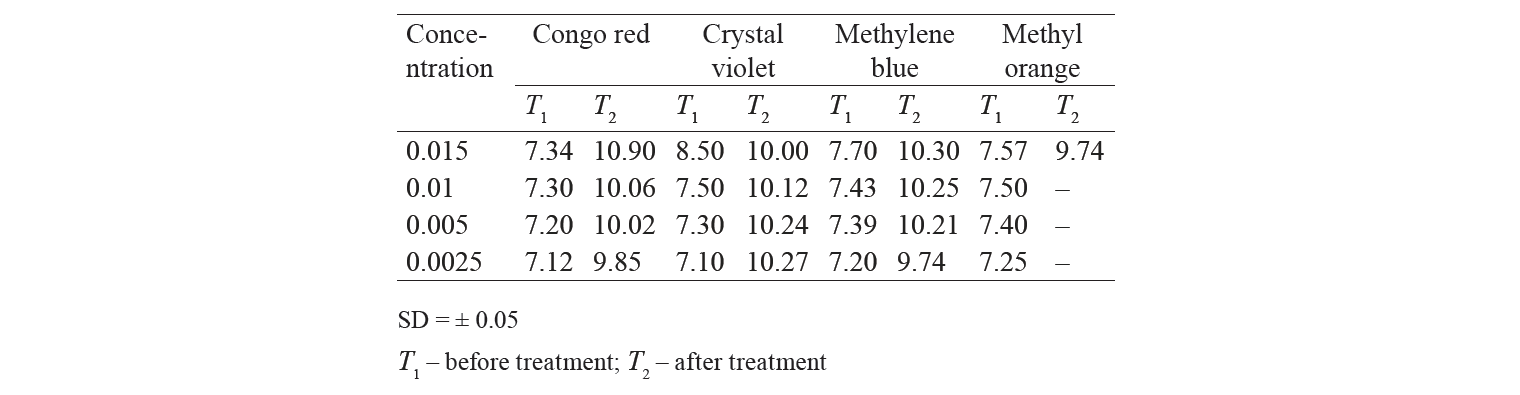
pH of dyes after treatment. The change in pH of different dye solutions was studied before and after the treatment with adsorbent. As one can see from Table 3, pH of the solution increased after the treatment. It could be due to introduction of basic component from the activated charcoal to the dye solution. Additional work could be done to find out the reason for the same.
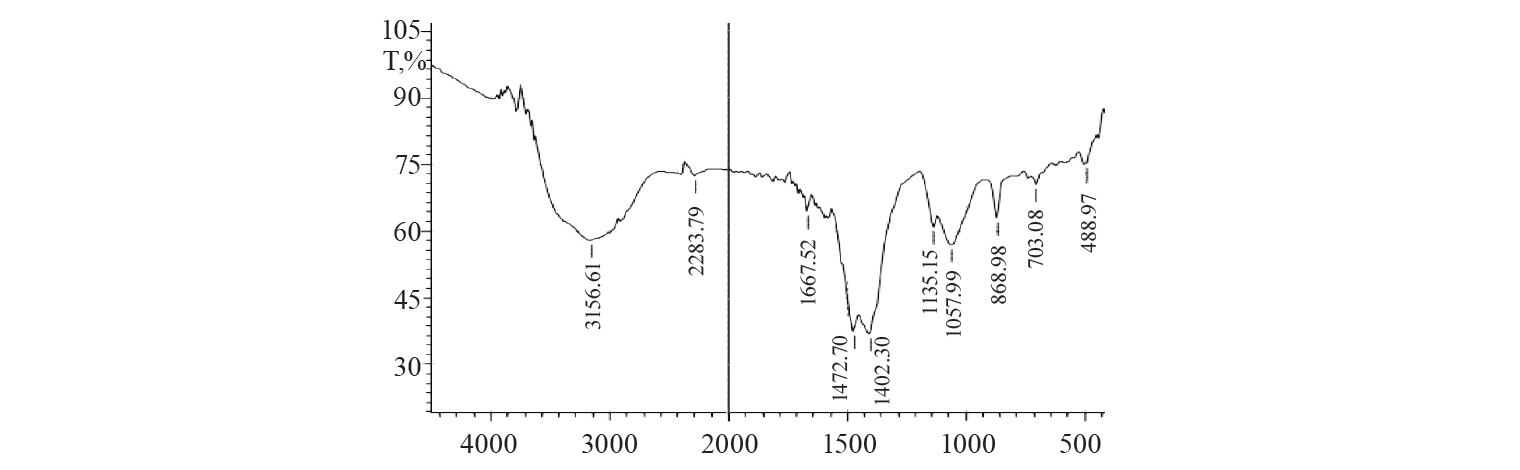
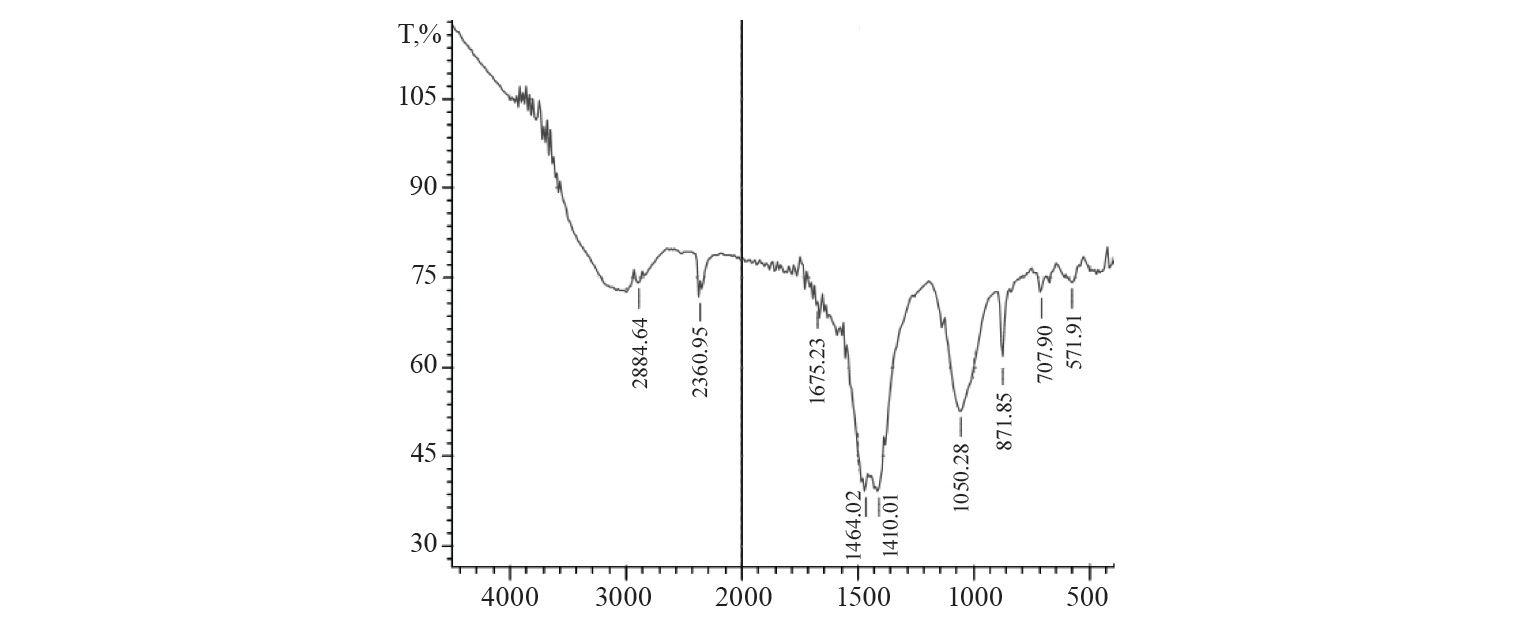
Characteristics of the adsorbent. The adsorbent was analysed by Fourier transform infrared spectroscopy (FTIR) and scanning electron microscopy (SEM). The FTIR spectrum of the activated charcoal is shown in Figure 7. The various peaks were observed due to different functional groups. The peak at about 2200 cm–1 could be due to the presence of sp hybridised carbon. The peak at 1660 cm–1 corresponded to aromatic C=C stretching. The peak value at 3166 cm–1 indicated the presence of C-H group.
Surface morphology revealed the adsorbent had porous structure. This could be due to the evaporation of the chemical reagent throughout the carbonisation process, leaving the vacant spaces on the surface of the adsorbent. It is obvious from the SEM image (Fig. 8) that the adsorbent is a mixture of activated charcoal prepared from dissimilar plant material. The presence of dissimilar plant materials in the adsorbent could be accountable for elimination of broad range of dyes both cationic and anionic.
Characteristics of the adsorbent after adsorption. The FTIR analysis of adsorbent after reaction with dye showed a number of additional peaks, perhaps due to the functional groups present in the dye that was adsorbed onto the adsorbent.
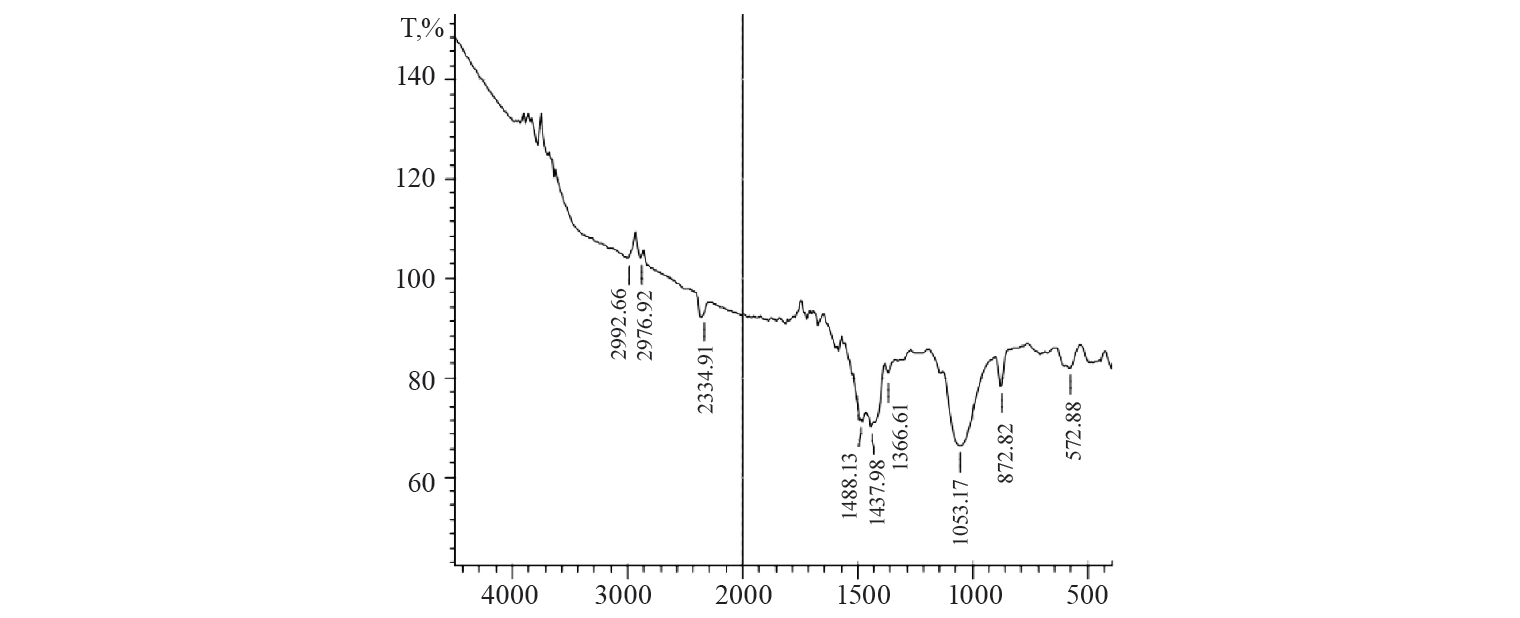
FTIR spectrometry demonstrated one additional vibrational peak at 1386.61 cm–1, which can be due to C-N stretching. The stretching vibration was observed at 872.88 cm–1 due to the presence of C-Cl bond. The C-S stretching band was observed at 572 cm–1. Every new peak definited that methylene blue was adsorbed on the activated charcoal by assembling altered kinds of bonds.
The FTIR analysis of activated charcoal after adsorption of crystal violet revealed no additional peaks (Fig. 10). One of the causes can be crystal violet inserted in pores.
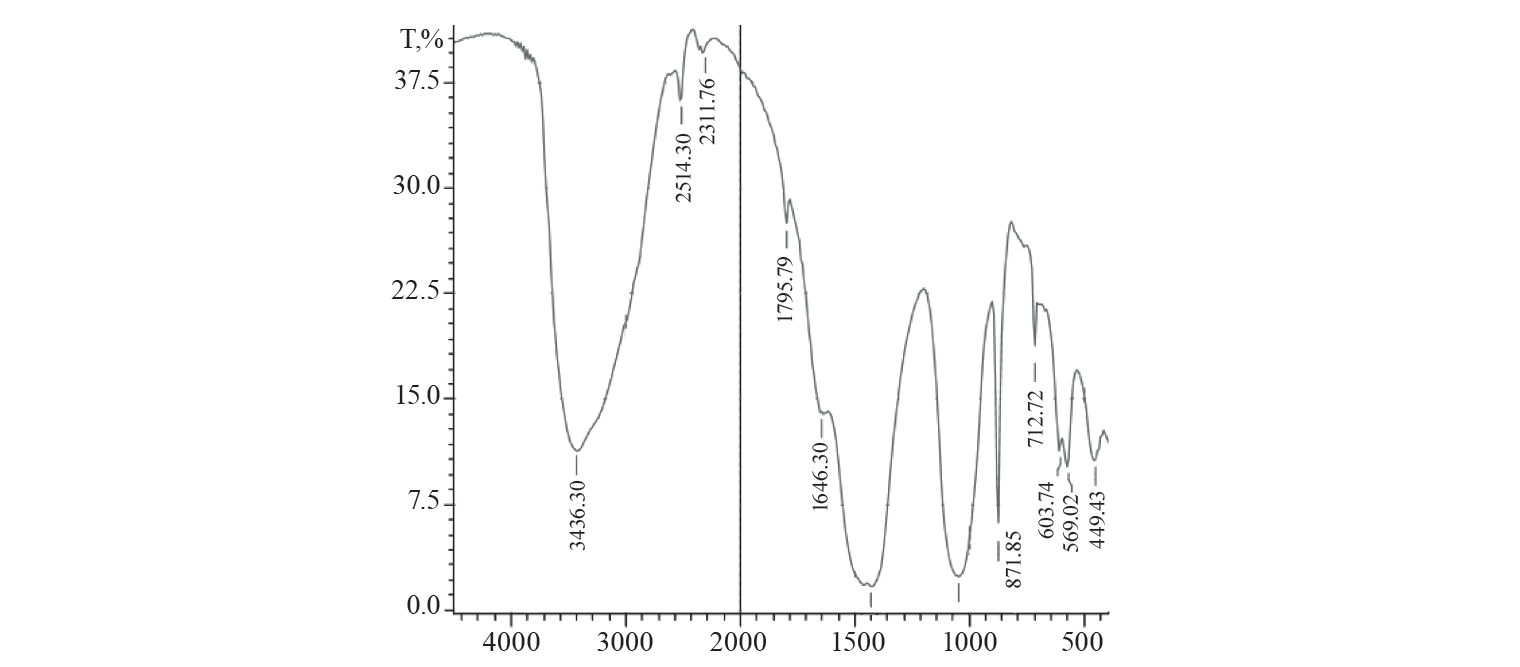
As for the FTIR spectra (Fig. 11) of activated charcoal with adsorbed congo red dye, three additional peaks were observed. Those were recorded at 3463, 2514, 1795, and 603 cm–1 that were due to N-H stretching, O-H stretching of carboxylic acid, C=O stretching, and C-C bending due to alkane, respectively. The presence of these functional groups confirms the adsorption of congo red dye on the activated charcoal.
ВЫВОДЫ
The results of this study made it possible to conclude that activated charcoal prepared from mixture of orange, banana, and pomegranate peels by carbonisation method had a great potential for removal of dyes from textile wastewater. In the present work, this adsorbent was tested on congo red, methylene blue, crystal violet, and methyl orange dyes. Studies showed that this adsorbent was effective in removing congo red, methylene blue, and crystal violet dyes from aqueous solutions, while it was not quite capable of removing methyl orange. Surface chemistry of activated carbon played an important role in dye adsorption. The type of the dye adsorbed on the adsorbent also depended on its textural properties, such as porosity and surface area. The adsorbent under study gave the best result for congo red dye. Thus, the present research developed a low-coat and environmentally friendly technology to remove dyes, as an alternative to known expensive and damaging methods.
СПИСОК ЛИТЕРАТУРЫ
- Robert S. Africa south of the Sahara-A geographical interpretation. Second Edition. New York: Guilford Press; 2004. 477 p.
- Farhaoui M, Derraz M. Review on Optimization of Drinking Water Treatment Process. Journal of Water Resource and Protection. 2016;8(8):777–786. DOI: https://doi.org/10.4236/jwarp.2016.88063.
- Romero V, Vila V, de la Calle I, Lavilla I, Bendicho C. Turn–on fluorescent sensor for the detection of periodate anion following photochemical synthesis of nitrogen and sulphur co–doped carbon dots from vegetables. Sensors and Actuators, B: Chemical. 2019;280:290–297. DOI: https://doi.org/10.1016/j.snb.2018.10.064.
- Kale RD, Kane PB. Colour removal using nanoparticles. Textile and clothing sustainability. 2016;2(4). DOI: https://doi.org/10.1186/s40689-016-0015-4.
- Dixit A, Dixit S, Goswami CS. Process and plants for wastewater remediation: A review. Scientific Reviews and Chemical Communications. 2011;1(1):71–77.
- Tan KA, Morad N, Teng TT, Norli I, Pannerselvam P. Removal of Cationic Dye by Magnetic Nanoparticle (Fe3O4) Impregnated onto Activated Maize Cob Powder and Kinetic Study of Dye Waste Adsorption. APCBEE Procedia. 2012;1:83–89. DOI: https://doi.org/10.1016/j.apcbee.2012.03.015.
- Ranganathan K, Karunagaran K, Sharma DC. Recycling of wastewaters of textile dyeing industries using advanced treatment technology and cost analysis–Case studies. Resources, Conservation and Recycling. 2007;50(3):306–318. DOI: https://doi.org/10.1016/j.resconrec.2006.06.004.
- Zhang L, Zhang Y, Tang Y, Li X, Zhang X, Li C. et al. Magnetic solid-phase extraction based on Fe3O4/graphene oxide nanoparticles for the determination of malachite green and crystal violet in environmental water samples by HPLC. International Journal of Environmental Analytical Chemistry. 2018;98(3):215–228. DOI: https://doi.org/10.1080/03067319.2018.1441837.
- Dalavi DK, Suryawanshi SB, Kolekar GB, Patil SR. AIEE active SDS stabilized 2-naphthol nanoparticles as a novel fluorescent sensor for the selective recognition of crystal violet: application to environmental analysis. Analytical Methods. 2018;10(20):2360–2367. DOI: http://doi.org/10.1039/C8AY00328A.
- Gautam S, Khan SH. Removal of methylene blue from waste water using banana peel as adsorbent. International journal of Science, Environment and Technology. 2016;5(5):3230–3236.
- Bello OS, Bello LA, Adegoke KA. Adsorption of dyes using different types of sand: A review. South African Journal of Chemistry. 2013;66:117–129.
- Akinola LK, Umar AM. Adsorption of crystal violet onto adsorbents derived from agricultural wastes: kinetic and equilibrium studies. Journal Applied Science Environment Management. 2015;19(2)279–288. DOI: https://doi.org/10.4314/jasem.v19i2.15.
- Rafatullah M, Sulaiman O, Hashim R, Ahmad A. Adsorption of methylene blue on low-cost adsorbents: A review. Journal of Hazardous Material. 2010;117(1–3):70–80. DOI: https://doi.org/10.1016/j.jhazmat.2009.12.047.
- Fba R, Akter M. Removal of dyes form textile wastewater by Adsorption using Shrimp shell. International Journal of Water Resources. 2016;6(3). DOI: https://doi.org/10.4172/2252-5211.1000244.
- Ibrahim MB, Sulaiman MS, Sani S. Assessment of Adsorption Properties Of Neem Leaves Waste for the Removal of Congo Red and Methyl Orange. 3rd International Conference on Biological, Chemical & Environmental Sciences; 2015; Kuala Lumpur. Kuala Lumpur, 2015. pp. 85–89. DOI: http://doi.org/10.15242/IICBE.C0915067.
- Beldean-Galea MS, Copaciu F-M, Coman M-V. Chromatographic Analysis of Textile Dyes. Journal of AOAC International. 2018;101(5):1353–1370. DOI: https://doi.org/10.5740/jaoacint.18-0066.
- Pradel JS, Tong WG. Determination of malachite green, crystal violet, brilliant green and methylene blue by microcloud-point extraction and nonlinear laser wave-mixing detection interfaced to micellar capillary electrophoresis. Analytical Methods. 2017;9(45):6411–6419. DOI: https://doi.org/10.1039/c7ay01706e.
- Foguel MV, Pedro NTB, Wong A, Khan S, Zanoni MVB, Sotomayor MDPT. Synthesis and evaluation of a molecularly imprinted polymer for selective adsorption and quantification of Acid Green 16 textile dye in water samples. Talanta. 2017;170:244–251. DOI: https://doi.org/10.1016/j.talanta.2017.04.013.
- Tan L, Chen K, He R, Peng R, Huang C. Temperature sensitive molecularly imprinted microspheres for solidphase dispersion extraction of malachite green, crystal violet and their leuko metabolites. Microchimica Acta. 2016;183(11):2991–2999. DOI: https://doi.org/10.1007/s00604-016-1947-8.
- De B, Karak N. A green and facile approach for the synthesis of water-soluble fluorescent carbon dots from banana juice. RSC Advances. 2013;3(22):8286–8290. DOI: https://doi.org/10.1039/c3ra00088e.
- Shen C, Shen Y, Wen Y, Wang H, Liu W. Fast and highly efficient removal of dyes under alkaline conditions using magnetic chitosan-Fe (III) hydrogel. Water Research. 2011;45(16):5200–5210. DOI: https://doi.org/10.1016/j.watres.2011.07.018.
- Pedraza A, Sicilia MD, Rubio S, Pérez-Bendito D. Assessment of the surfactant-dye binding degree method as an alternative to the methylene blue method for the determination of anionic surfactants in aqueous environmental samples. Analytica Chimica Acta. 2007;588(2):252–260. DOI: https://doi.org/10.1016/j.aca.2007.02.011.