Аннотация
The use of ultraviolet radiation in the treatment of milk and other liquid foods is a very promising field of study since it reduces their bacterial load. It is rarely used to increase the vitamin D3 content and modify the protein and fatty acid composition of milk. The paper describes how different parameters of ultraviolet radiation influence such characteristics of raw and pasteurized milk as the mass fraction of total protein, nonprotein nitrogen content, active and titratable acidity, general bacterial load (QMA&OAMO), fatty acid composition, and vitamin D3 content. Low-pressure gas-discharge lamps were used to treat a 400 µm moving layer of milk with ultraviolet radiation. The radiation time, its doses, and the milk flow rate changed in the ranges of 5–25 min, 5.1–102 mJ/cm2, and 0.04453- 0.13359 m3/s, respectively. We identified optimal radiation ranges that lead to both a lower microorganism content and a higher vitamin D3 content. Our study also determined specific correlations in the mutual changes of the given parameters. The treatment ranges did not produce any significant changes in other physico-chemical properties of milk. We also found that vitamin D3 was synthesized in raw and pasteurized milk in a similar way. Moreover, there was an insignificant decrease in the vitamin D3 content in milk treated with ultraviolet radiation during storage for up to 48 hours. On the whole, the results indicate that the treatment of milk with ultraviolet radiation in the dosage range from 5.1 to 102 mJ/cm2 has a complex effect on the total bacterial load (QMA&OAMO) and vitamin D3 content, whereas it has almost no effect on the protein and fatty acid composition.Ключевые слова
Ultraviolet radiation, milk, protein, fatty acids, vitamin D3, (QMA&OAMO) CFU/cm3ВВЕДЕНИЕ
The assessment of the ultraviolet radiation (UVR) effect on the physico-chemical and vitamin composition, as well as the bacterial load of dairy and other food products, is a promising field of research as it permits directional regulation of their properties.
Numerous studies are currently conducted in this area. However, the data are still insufficient to accurately assess all the aspects and mechanisms of the ultraviolet radiation effect on food and dairy products during their processing. There is therefore a need for further research in this area.
Milk is an important source of nutrition. It contains proteins, milk fat, minerals, and different vitamins. The main components of milk vary according to the breed of cows, feeding, and livestock management. These changes mostly affect the content of fat-soluble vitamins, in particular vitamin D3. In European countries, such as Denmark, the consumption of milk, cheese, and other dairy products accounts for about 12% of the total intake of vitamin D3 [1, 2]. Dairy products with a low content of vitamin D3 cannot serve as its natural source. Insufficient intake of vitamin D3 increases the risk of developing hypertension, autoimmune diseases, diabetes, rickets, and cancer [1].
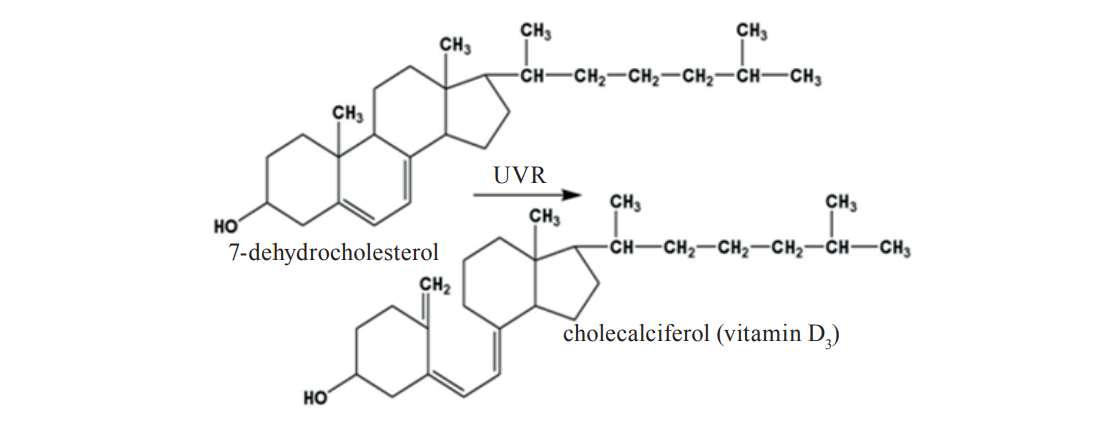
Lactating cows have two primary sources of increased vitamin D3 content in milk. This vitamin can come with food, including vitamin-enriched supplements, or be produced endogenously under the impact of ultraviolet radiation on the cow’s skin [3, 4]. Under ultraviolet light radiation, 7-dehydrocholesterol turns into precalciferols as a result of the prototropic rearrangement. Precalciferols serve to form the D vitamins (Fig. 1).
The vitamin D3 content in milk and dairy products can be increased by its introduction at a particular stage of the technological process. This can also be achieved by ultraviolet treatment due to its directional effect on 7-dehydrocholesterol.
Various gas-discharge radiators are currently the most common sources of ultraviolet radiation. Mercury lamps are used because mercury in a gaseous state is activated at relatively low temperatures. Furthermore, the discharge in mercury vapor provides the largest number of intense lines in the ultraviolet spectrum. High- and low-pressure mercury lamps are used to ensure effective ultraviolet treatment of the material. They differ in their intensity depending on mercury vapor pressure. The advantage of low-pressure mercury lamps is that the largest share of radiation falls on the wavelength of λ = 253.7 nm, which has a maximum bactericidal effect. Therefore, lamps of this type are mostly used to reduce the bacterial load of the product. In high-pressure mercury lamps, the spectral area of impact has a higher wavelength range, which makes them less suitable for the bacterial treatment.
Flash xenon or argon high-pressure lamps became quite widespread, along with mercury gas-discharge lamps. Compared to argon lamps, flash xenon lamps have better bactericidal activity, shorter exposure time, and higher safety. Their disadvantages include a shorter period of guaranteed action, as well as increased operating costs.
We should note a shortage of comparative studies into the use of ultraviolet radiators in the field of food biotechnology.
Depending on the modes, the UV treatment can have a different effect on the composition and properties of milk. The most important components of milk – proteins, fats, and vitamins – can absorb UVR throughout its range. The energy absorbed by them can change the physico-chemical properties of these organic molecules. Furthermore, UVR produces active forms of oxygen that can change the chemical composition and properties of the main components of milk as a result of free radical reactions. The active forms of oxygen cause the DNA damage in microorganisms and oxidation of specific protein groups. Therefore, UVR is successfully used to reduce the bacterial load of dairy raw materials [5, 6].
The changes occurring in bacteria under UVR are multistage, with DNA molecules being the final stage of their action [7].
In addition to the direct influence on DNA, increased UVR intensity can cause cell mutations. This phenomenon is accompanied by the formation of free radicals and peroxides with mutagenic properties. For example, [8] reports an increased number of mutational changes in microorganisms cultivated in nutrient media.
UVR has the greatest oppressive influence on microorganisms in the wavelength range of 205–315 nm [9].
The first practical application of UVR as bactericidal treatment was for disinfection of water [10, 11]. Positive results were also achieved in the treatment of other highly transparent liquid foods, such as sugar solutions, their derivatives, and juices [12–17].
Numerous studies of UVR influence on milk and dairy products showed that the treatment is complicated by their low transparency due to the screening ability of protein and fat. Furthermore, dairy products have a complex composition, and their components are closely related to each other [18–23].
Nevertheless, those studies served as a basis for industrial facilities designed to reduce the quantitative content of bacteria in milk [6, 10, 24, 25].
Ultraviolet treatment has no direct effect on milk proteins in a certain range of parameters [26–28]. At the same time, we can use ultraviolet light to change the structure of proteins and give them new properties by changing treatment modes. This is confirmed by Cho et al. which showed that UV treatment could affect the molecular structure of β-lactoglobulin, the main allergen in milk [29]. Similar results were obtained in another study that recorded shifts in the molecular structure of β-lactoglobulin. These results also indicate a possibility of regulating the peptide profile of milk proteins and using UVR to reduce milk allergenicity [8].
Biotechnological methods used in the dairy industry can affect the secondary and tertiary structure of milk proteins [2, 9]. At the same time, the influence of physico-chemical factors can lead to the unfolding of the protein globule and increased proteolysis of denatured proteins.
This was confirmed by studying the proteolysis of milk proteins with pepsin and trypsin after UV treatment. The analysis of pepsin and trypsin hydrolysates showed that the number of cleaved protein substrates and peptide fractions was similar for all the milk samples subjected to ultraviolet radiation [13, 30]. Thus, the UV treatment of milk usually has no influence on the proteolysis of milk proteins with pepsin and trypsin and on its digestibility.
The UV treatment of milk can intensify the formation of vitamin D3 [13, 24, 25]. However, this technology is not in wide industrial use yet due to a more common method of directly introducing vitamin supplements in dairy products. At the same time, a combination of the latter method with the benefits of UR remains highly relevant. Thus, the UV treatment of milk can be used to reduce its bacterial load, increase the vitamin D3 content, and change certain components of milk.
Despite extensive studies in this area, the results are quite controversial. Moreover, most authors are mainly interested in studying the effectiveness of ultraviolet treatment in reducing the bacterial load or increasing the vitamin D3 content.
This paper attempts to fully investigate the effect of certain ultraviolet radiation parameters on the above indicators and, at the same time, evaluate the changes in the protein and fatty acid composition of milk.
Our main objective was to find an optimal range of UVR which could reduce the bacterial load of milk and increase the vitamin D3 content without having any significant effect on the protein and fatty acid composition of the treated product. Furthermore, our hypothesis was that the changes in the bacterial load and vitamin D3 growth might be interrelated.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
Our objects of study were raw milk with a 3.8% mass fraction of fat and pasteurized milk with a 3.2% mass fraction of fat. The temperature of the product was 4 ± 2°C.
To conduct the experiment, we used a unit for treating milk in a 400 µm circular layer. Three symmetrically arranged Philips gas-discharge lamps (TUV 55W PL-L) were a source of UVR, with a wavelength of 253.7 nm. A thin layer of milk was passed through a gap between two cylinders. The outer cylinder was made of stainless steel, and the inner cylinder was made of quartz glass with gas-discharge lamps placed inside it. The outer diameter and the height of the cylinder were 120 and 600 mm, respectively.
The lamps were cooled with an electric fan that pumped air through the internal quartz glass cylinder.
Milk was supplied to the unit by a pump with adjustable capacity. Sampling for the study was carried out under aseptic conditions.
The study aimed to assess the UVR effect on the content of proteins, nonprotein nitrogen, fatty acids, and vitamin D3, as well as the bacterial background of processed milk. The unit parameters included productivity (Q) 100–420 l/h; treatment time (τ) 5–25 minutes; the volumetric milk flow rate in the irradiated layer (V) 0.04453–0.13359 m3/s; the surface bactericidal irradiation dose, i.e. the relation between the bactericidal irradiation energy and the irradiated surface area, (Нe) 5.1–102 mJ/cm2.
Some samples were used to assess their protein and fatty acid composition, as well as vitamin D3 content, both directly after the UV treatment and during storage, namely after 24, 36, and 48 hours, respectively. The assessment was carried out under standard conditions. We used the following State Standards to determine specific parameters, namely: State Standard 38892-2014 for active acidity (pH); State Standard R 54669-2011 for titratable acidity; State Standard 32901-2014 for the total number of microorganisms (QMA&OAMO); State Standard 23327-98 for the mass fraction of total protein; State Standard R 55246-2012 for nonprotein nitrogen content; State Standard 32915-2014 for the fatty acid composition; and State Standard R 54637-2011 for vitamin D3 content. The analysis was conducted in triplicate. State Standard 26754-85 was used to regulate the milk temperature after the ultraviolet treatment.
The following equipment was used to measure the mass fraction of protein and nonprotein nitrogen:
– a SH220F digester (Hanon, China) with the maximum heating temperature of 450°C;
– a WD03 sulfuric acid vapour suction system; and
– a K9840 distillation system (Hanon, China) with an automatic supply of alkali, receiving solution, and distillate.
A 4000M Crystallux gas chromatograph (Russia) was used to separate and identify fatty acids in the samples. It was equipped with a Supelco-SP2560 capillary column (100 m×0.25 mm, df = 0.20 µm, Sigma-Aldrich, USA) and a flame ionization detector.
The following equipment was used to measure the vitamin D3 content:
– a liquid chromatograph equipped with a spectrophotometer (Gilson, France);
– a Luna C18(2) column (5 µm, 250×4.6 mm, Phenomenex, USA); and
– a vacuum unit for Strata C18-E SPE with replaceable cartridges (Phenomenex, USA).
The analysis was performed in the following conditions:
– gradient mode of separation;
– mobile phase: acetonitrile (eluent A) – dichloromethane (eluent B);
– gradient elution programme: A/B = 100/0 at the beginning of the analysis; gradient A/B = 90/10 in 8 min; gradient A/B = 70/30 in 2 min; isocratic elution A/B = 70/30 in 10 min; gradient A/B = 100/0 in 3 min; isocratic elution A/B to 100/0;
– flow rate: 1.0 cm3/min;
– loop dispenser volume: 20 mcl;
– room temperature; and
– spectrophotometric detection with changing the wavelength of the light source during analysis: 0 min with а wavelength of 436 nm, 10 min with а wavelength of 280 nm, and 27 min with а wavelength of 436 nm.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
The experiments involved the evaluation of active (pH) and titratable (Т°) acidity. We found that these indicators did not undergo any significant changes in the following range of treatment parameters: Нe = 5.1–102 mJ/сm2, V = 0.04453–0.13359 m3/s.
As can be seen in Table 1, the mass fraction of total protein remained unchanged, regardless of the irradiation time or type of milk.
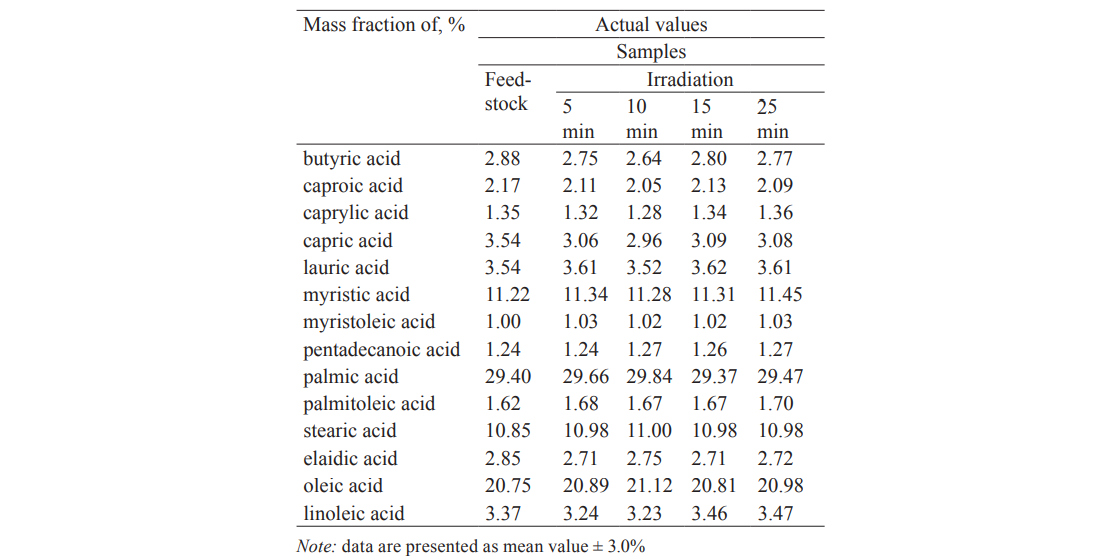
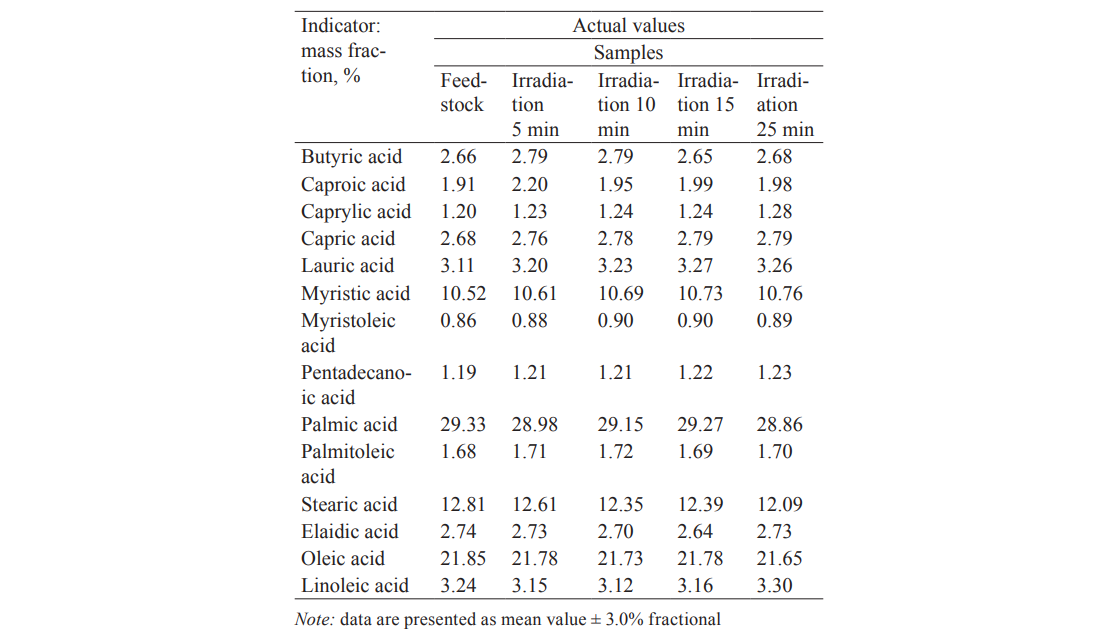
Fatty acids, especially unsaturated, are an important component of the fat phase of milk. Tables 2 and 3 show the fatty acid composition of raw and pasteurized milk after different periods of the UV treatment.
The main fatty acids amounted to 95.78% in the control. Their composition and content hardly changed under the influence of UVR. Furthermore, the fat phase contained 23 minor fatty acids (4.22%). The UV treatment caused a slight increase in some of them and a slight decrease in others, with their total content remaining 4.02%. Similar results were obtained for pasteurized milk (Table 3). We should note that the analysis covered the whole spectrum of fatty acids (37 fatty acids). Those acids which are not included in Tables 2 and 3 showed no significant changes either.
Thus, the ultraviolet treatment of milk in the given range of exposure did not affect the physico-chemical properties of fatty acids and their composition.
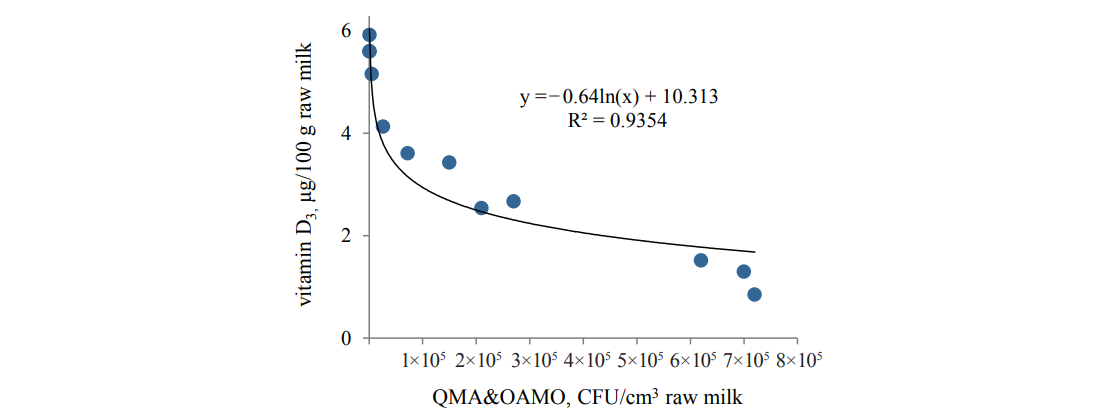
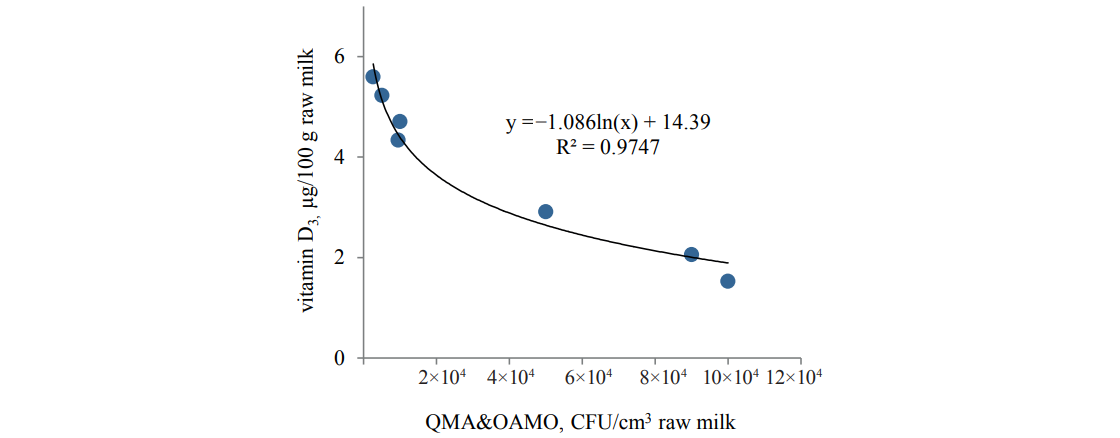
The study revealed a correlation between the changes in the vitamin D3 content and the microbial load (CFU/cm3) of milk (Figs. 2 and 3).
The data for both raw and pasteurized milk confirmed the interrelation between the vitamin D3 content and QMA&OAMO and also indicated that an increase in the initial bacterial load negatively affected the vitamin D3 growth.
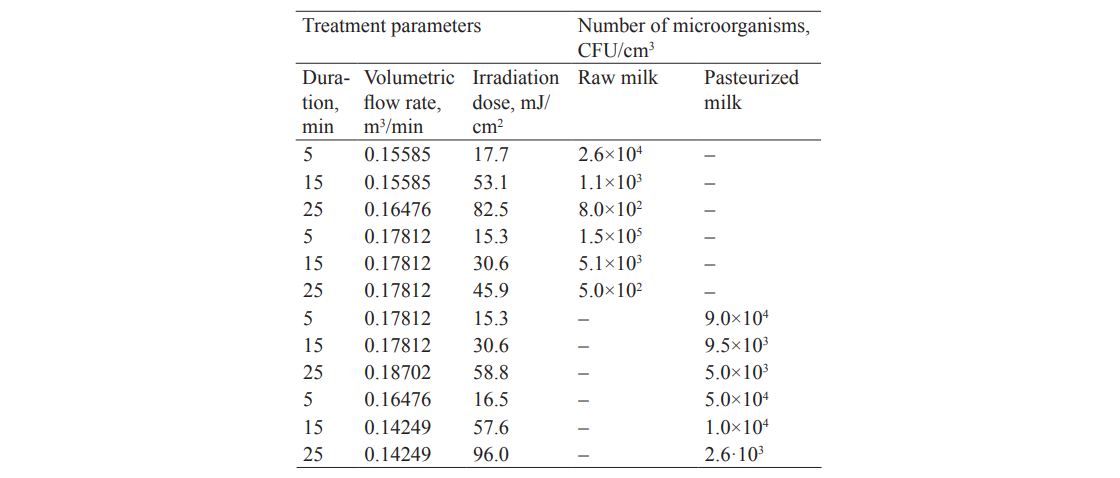
Studying the influence of the volumetric milk flow rate (V = 0.04453–0.13359 m3/s) and treatment duration (τ = 5–25 min) at different irradiation doses revealed a significant effect of UVR on the bacterial load in the given ranges (Table 4).
The feedstock in the experiments included raw milk with a bacterial load of 2.1×105 CFU/cm3 and pasteurized milk with a bacterial load of 1×105 CFU/cm3.
We found that the irradiation doses of over 30 mJ/cm2 and the treatment duration of over 15 min allowed for a more intensive reduction of the bacterial load in raw milk, compared to pasteurized milk.
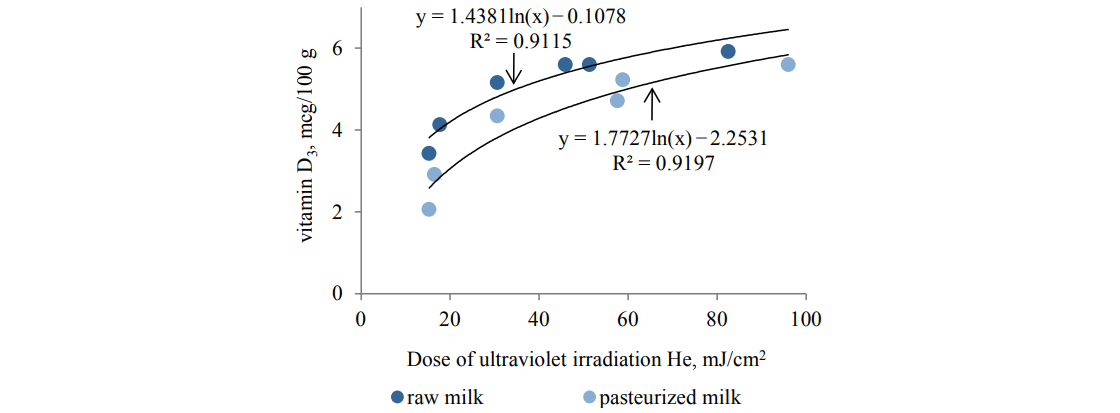
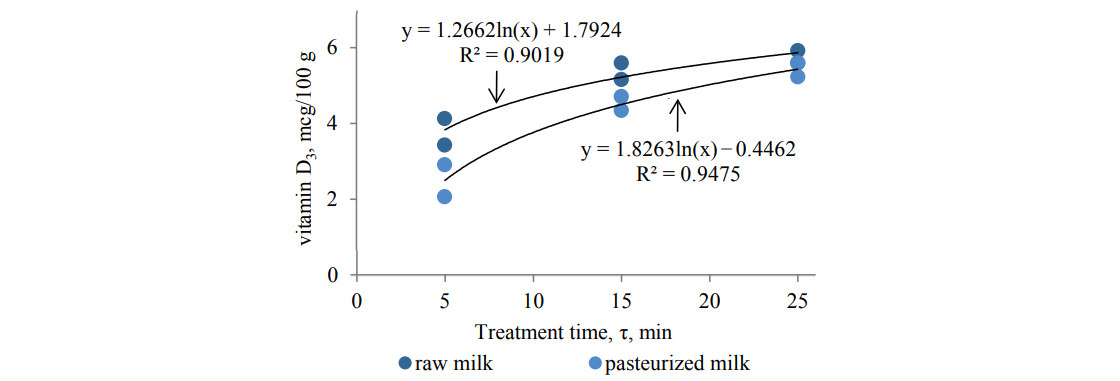
The experiments also showed an increase in the vitamin D3 content within the UV treatment parameters specified in Table 4. This was confirmed by the results shown in Figs. 4 and 5.
The analysis showed that the irradiation doses below 5 mJ/cm2 had an insignificant effect on the microbiological load and the vitamin D3 content (Table 4; Figs. 4 and 5). Higher doses of irradiation, however, led to a rather intensive growth in vitamin D3 and a decrease in the bacterial load.
On the whole, we found low-pressure gas-discharge irradiation sources effective in producing a considerable simultaneous effect on the vitamin D3 content and the microbiological load of both raw and pasteurized milk within the treatment modes. At the same time, low irradiation doses up to He = 102 mJ/cm2 did not have a significant effect on the protein and fatty acid compositionof milk.
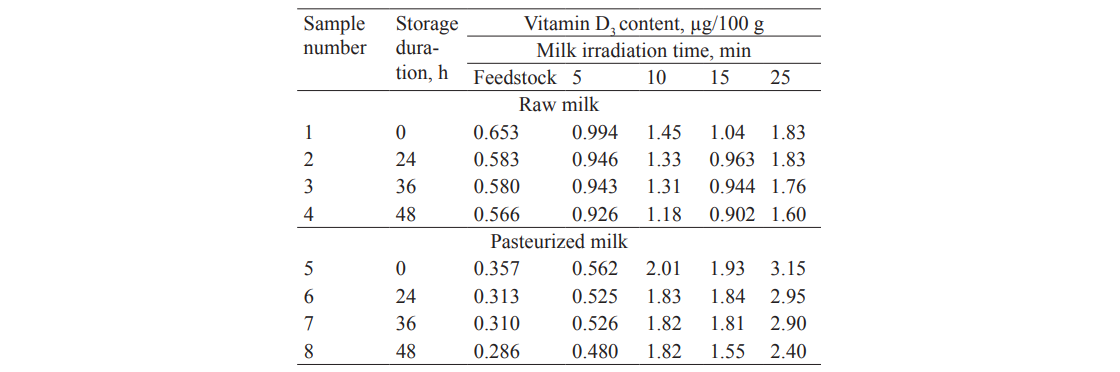
To assess changes in the vitamin D3 content during storage, we treated raw and pasteurized milk with UVR at different durations in the range of 20–102 mJ/cm2. After the treatment, the milk samples were stored at 4 ± 2°C. We found that the vitamin D3 content in both raw and pasteurized milk remained almost unchanged during storage under standard conditions for two days (Table 5).
ВЫВОДЫ
Thus, we found that the ultraviolet treatment of a 400 µm layer of milk with low-pressure gas-discharge lamps at a wavelength of 253.7 nm in the dose range from 5 to 102 mJ/cm2 makes it possible to simultaneously reduce the bacterial load and increase the vitamin D3 content. The study proved that these processes are interrelated; furthermore, they do not cause any significant changes in the protein and fatty acid composition of milk, both after production and during storage. The patterns established are identical for both raw and pasteurized milk with slight changes during storage for 48 hours.
СПИСОК ЛИТЕРАТУРЫ
- DeLuca H.F. Overview of general physiologic features and functionsof vitamin D. The American Journal of Clinical Nutrition, 2004, vol. 80, no. 6, pp. 1689S–1696S. DOI: https://doi.org/10.1093/ajcn/80.6.1689S.
- Pedersen A.N., Fagt S., Groth M., et al. Dietary Habits in Denmark 2003–2008. Main results (in Danish). Copenha- gen, Søborg: Technical University of Denmark Publ., 2010.
- Jakobsen J., Jensen S.K., Hymøller L., et al. Short communication: Artificial ultraviolet B light exposure increases vitamin D levels in cow plasma and milk. Journal of Dairy Science, 2015, vol. 98, no. 9, pp. 6492–6498. DOI: https:// doi.org/10.3168/jds.2014-9277.
- Weir R.R., Strain J.J., Johnston M., et al. Environmental and genetic factors influence the vitamin D content of cows’ milk. Proceedings of the Nutrition Society, 2017, vol. 76, no. 1, pp. 76–82. DOI: https://doi.org/10.1017/ S0029665116000811.
- Kharitonov V.D. and Yurova E.A. Vliyaniye ultrafioleta na sostav i svoystva moloka [The effect of ultraviolet radiation on the composition and properties of milk]. Dairy industry, 2006, no. 7, pp. 32. (In Russ.).
- Gavryushenko B.S., Kharitonov V.D., Dimitriyeva S.E., et al. Promyshlennaya tekhnologiya, oborudovaniye dlya ochistki moloka ot mikroorganizmov i obogashcheniya vitaminom D [Industrial technology and equipment for milk decontamination and enrichment with vitamin D]. Materialy mezhdunarodnoy nauchno-prakticheskoy konferentsii “Molochnaya industriya” [Proceedings from the international scientific and practical conference “Dairy Industry”]. Moscow, 2009, pp. 88–89. (In Russ.).
- Anugu A.K. Microbial inactivation and allergen mitigation of food matrix by pulsed ultraviolet light. Available at:http://ufdc.ufl.edu/UFE0045406/00001. (accessed 15 October 2014).
- Elmnasser N., Dalgalarrondo M., Orange N., et al. Effect of pulsed-light treatment on milk proteins and lipids. Journal of Agricultural and Food Chemistry, 2008, vol. 56, no. 6, pp. 1984–1991. DOI: https://doi.org/10.1021/jf 0729964.
- Letayev S.A. Obosnovaniye parametrov ustanovki obezzarazhivaniya moloka na fermakh ultrafioletovym i in- frakrasnym izlucheniyem. Diss. dokt. tekhn. nauk [Substantiation of the parameters for milk disinfection under ultraviolet and infrared radiation on farms. Cand. eng. sci. diss.]. Moscow, 2012, 24 p.
- Masschelein W.J. and Rice R.G. Ultraviolet light in water and wastewater sanitation. Boca Raton: CRC Press Publ., 2002. 192 p.
- Chevrefils G., Caron E., Wright H., et al. UV dose required to achieve incremental log inactivation of bacteria, proto- zoa and viruses. IUVA News, 2006, vol. 8, no. 1, pp. 38–45.
- Choi L.H. and Nelsen S. The effects of thermal and nonthermal processing methods on apple cider quality and consumer acceptability. Journal of Food Quality, 2005, vol. 28, no. 1, pp. 13–29. DOI: https://doi.org/10.1111/j.1745- 4557.2005.00002.x.
- Guerrero-Beltrán J.A. and Barbosa-Ganovas G.V. Reduction of Saccharomyces cerevisiae, Escherichia coli and Liste- ria innocua in apple juice by ultraviolet light. Journal of Food Process Engineering, 2005, vol. 28, no. 5, pp. 437–452. DOI: https://doi.org/10.1111/j.1745-4530.2005.00040.x
- Koutchma T., Parisi B., and Patazka E. Validation of UV coiled tube reactor for fresh juices. Journal of Environmen- tal Engineering and Science, 2007, vol. 6, no. 3, pp. 319–328. DOI: https://doi.org/10.1139/s06-058.
- Keyser M., Muller I.A., Cilliers F.P., Wihann N., and Gouws P.A. Ultraviolet radiation as a nonthermal treatment for the inactivation of microorganisms in fruit juice. Innovative Food Science and Emerging Technologies, 2008, vol. 9, no. 3, pp. 348–354. DOI: https://doi.org/10.1016/j.ifset.2007.09.002.
- Oteiza J.M., Giannuzzi L., and Zaritzky N. Ultraviolet treatment of orange juice to inactivate E. coli O157:H7 as affected by native microflora. Food and Bioprocess Technology, 2010 vol. 3, no. 4, pp. 603–614. DOI: https://doi. org/10.1007/s11947-009-0194-y.
- Falguera V., Pagan J., and Ibarz A. Effect of UV irradiation on enzymatic activities and physicochemical properties of apple juices from different varieties. LWT – Food Science and Technology, 2011, vol. 44, no. 1, pp. 115–119. DOI: https://doi.org/10.1016/j.lwt 2010.05.028.
- Chumachenko V.A. and Sukhinin V.N. Primeneniye UF-izlucheniya dlya obezzarazhivaniya moloka [The use ofUV radiation to disinfect milk]. Zhivotnovodstvo [Livestock breeding], 1976, no. 1, pp. 75–76. (In Russ.).
- Chernykh E.A. Vliyaniye ultrafioletovoy obrabotki moloka korov na ego biokhimicheskiye, tekhnologicheskiye i gigi-yenicheskiye svoystva. Diss. kand. biolog. nauk [The effect of ultraviolet treatment of cow’s milk on its biochemical, technological, and hygienic properties. Cand. sci. biol. dicc.]. Dubrovitsy, 2006, 111 p.
- Chernykh E.A. and Yurova E.A. Vliyaniye ultrafioletovoy obrabotki na sostav i svoystva korovyego moloka [The effect of ultraviolet treatment on the composition and properties of cow’s milk]. Milk processing, 2006, no. 4, p. 31. (In Russ.).
- Altic L.C., Rowe M.T., and Grant I.R. UV light inactivation of Mycobacterium avium subsp. paratuberculosis in milk as assessed by FASTPlaqueTB phage assay and culture. Applied and Environmental Microbiology, 2007, vol. 73, no. 11, pp. 3728–3733. DOI: https://doi.org/10.1128/AEM.0057-07.
- Kehoe J.J., Remondetto G.E., Subirade M., Morris E.R., and Brodkorb A. Tryptophan-mediated denaturation of be- ta-lactoglobulin A by UV irradiation. Journal of Agricultural Food Chemistry, 2008, vol. 56, no. 12, pp. 4720–4725. DOI: https://doi.org/10.1021/jf0733158.
- Kharitonov V.D. and Sherstneva N.E. Vliyaniye ultrafioletovogo izlucheniya na osnovnyye komponenty i mikro- biologicheskiye zhidkikh pishchevykh produktov [The effect of ultraviolet radiation on the main components and microbiological indicators of liquid foods]. Scientific works of the Belarusian State University, 2014, vol. 9, no. 1, pp. 9–22. (In Russ.).
- Capozzo J.C., Koutchma T., and Barnes G. Chemical characterization of milk after treatment with thermal (HTST and UTH) and nonthermal (turbulent flow ultraviolet) processing technologies. Journal of Dairy Science, 2015, vol. 98, no. 8, pp. 5068–5079. DOI: https://doi.org/10.3168/jds.2014-9190.
- Kharitonov D.V. and Budrik V.G. Innovatsionnoye oborudovaniye dlya pererabotki moloka [Innovative milk process-ing equipment]. Moscow: Tipografiya RAN Publ., 2015. 480 p. (In Russ.).
- Filipov Zh. Changes in the total protein and protein fractions in cow’s milk irradiated with ultraviolet rays: Veterinar- no-meditsinski nauki, 1976, vol. 13, no. 4. pp. 45–49.
- Matak K.E., Summer S.S., Duncan S.E., et al. Effects of ultraviolet irradiation on chemical and sensory properties of goat milk. Journal of Dairy Science, 2007, vol. 90, no. 7, pp. 3178–3186. DOI: https://doi.org/10.3168/jds.2006-642.
- Simonenko S.V., Dimitrieva S.Ye., Agarkova Ye.Yu., Chervyakovskiy Ye.M., and Kurchenko V.P. Influence of ther- mal processing on biological and technological properties of goat milk proteins. Storage and Processing of Farm Products, 2010, no. 3, pp. 26–30. (In Russ.).
- Cho Y.S., Song K.B., and Yamda K. Effect of Ultraviolet Irradiation on Molecular Properties and Immunoglobulin Production-regulating Activity of β-Lactoglobulin. Food Science and Biotechnology, 2010, vol. 19, no. 3, pp. 595–602. DOI: https://doi.org/10.1007/s10068-010-0084-0.
- Simonenko S.V., Dimitrieva S.E., Agarkova E.Yu., et al. Issledovaniye sokhrannosti belkovogo komponenta kozyego moloka pod vozdeystviyem ultrafioletovogo izlucheniya [Study into the preservation of goat milk proteins under ultraviolet radiation]. Scientific works of the Belarusian State University, 2009, vol. 4, no. 2. pp. 277–282. (In Russ.).