Аннотация
Marinating is a traditional technology to improve the shelf - life and quality of products. Enriched Vietnamese tamarind fish sauce was prepared and used to marinate green mussels. Subjects of this research were: green mussels (C1), marinated green mussels (C2), and marinated green mussels packaged in modified atmosphere (M1, M2, and M3). A percentage of O2:CO2:N2 was 5:50:45 for M1, 5:70:25 for M2, and 5:90:5 for M3. Microbiological, hemical, and sensory qualities of the samples were analyzed during storage at 4°C for 30 days. The results indicate that glycogen, iron, and zinc contents as well as pH values decreased in all the cases during storage. In addition, TVB-N and TBARS values as well as mesophilic and lactic acid bacteria increased with time in all the samples. The number of psychrophilic bacteria in marinated samples was small or not detected at all. Escherichia coli, Staphylococcus aureus, Salmonella spp. and Vibrio cholera were below standard values. Based on sensory acceptability, the shelf-life of C1, C2, M1, M2, and M3 samples was 12, 18, 24, 24, and 27 days, respectively.ВВЕДЕНИЕ
Green mussel (Perna viridis L.) is a common marine animal in the Asia-Pacific region [1]. This is a large bivalve with smooth, elongated shell typical of several mytilids. Flesh of the green mussel is considered an excellent source of selenium, calcium, iron, magnesium, phosphorous and vitamins (A, B1, B2, B6, B12 and C) [2, 3]. In addition, mussel fat is rich in polyunsaturated fatty acids (PUFA, 37–48% of total fatty acids) mainly ω–3 PUFA [4], which is biologically important and can decrease the risk of cardiovascular disease [5]. However, this filter feeder animal has a high risk of microbiological contamination that lead to a short shelf-life, poor quality, and economic losses. In addition, only well–cooked green mussels are microbiologically safe.
Marinating is a seasoning process mainly using organic acid containing liquids [6]. Liquid marinades generally include sugar, spices, oil, and acid such as vinegar, fruit juice or wine. Marinating improves tenderness, juiciness, taste and aroma, as well as improves the shelflife of red meat, poultry, seafood, and vegetables [7–10].
Vietnamese tamarind fish sauce is popular in its country of origin. The main ingredients are tamarind pulp, fish sauce and sugar, while some fresh garlic and chili are used to enhance taste. The Vietnamese consume this sauce with various foods, such as seafood, fried food, dried food. It is even used as a condiment in various kinds of food products. Ready-to-cook products are expected to gain popularity in South-East Asia as well. Mineral deficiencies, including iron and zinc, remain problematic in developing countries. Thus, the enrichment of food products with minerals can be an effective way to solve this problem. However, according to a market survey, green mussels marinated with enriched tamarind fish sauce are not commercially available, especially when packed in a modified atmosphere. Modified atmosphere packaging (MAP) is a technology that prolongs the shelf-life of food products by retarding microbial growth and biochemical reactions, often with an elevated carbon dioxide level [11]. Moreover, in MAP oxygen may inhibit the growth of anaerobic bacteria and accumulate toxins from Clostridium botulinum, type E [12]. However, botulinum toxins can form at below 2% oxygen [13, 14].
Therefore, the aim of this study was to determine the shelf-life of green mussels marinated with tamarind fish sauce enriched with iron and zinc. The mussels were packed in modified atmosphere and stored at 4°C.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
Materials. Tamarind pulp without seeds, fish sauce, white sugar, finger chili, garlic, and the fresh green mussels (40–50 individuals/kg) were purchased from a supermarket located in Songkhla province. Experiments were carried out at the Food Technology Department, the Agro-Industry Faculty of the Prince of Songkla University, Thailand. Iron sodium ethylenediaminotetraacetate (FeNaEDTA·3(H2O)) and zinc sulfate (ZnSO4·7(H2O)) were taken from Sigma–Aldrich Company (Steinheim, Germany).
Marinated green mussel preparation. The green mussels were washed during 2 hours with tap water, then blanched in boiling water at 100°C for 30 s to facilitate meat removal. A ratio of sample to water was 1:2 w/w. Afterwards, the meat of green mussels was separated from the shells. We prepared tamarind fish sauce contained 42% of tamarind slurry, 37% of sugar, and 21% of fish sauce. 236 mg of FeNaEDTA·3(H2O) and 88.5 mg of ZnSO4·7(H2O) were added into 30 g of the sauce, which was 36 mg of Fe and 20 mg of Zn, respectively. The green mussel meat was soaked in the enriched sauce at the ratio of 1:1. The marinated meat was packaged in 15 х 30 cm Havel Vacuum bags, b.v. (Europac Co., Ltd) with O2 transmission rate 46.6 cm3m2 day-1 at 38°C, 1 atm. We tested five samples of green mussels: meat without marinade solution packed under normal air (C1), marinated meat without gases (C2), and meat packed in the modified atmosphere (M1, M2, and M3). A percentage of O2:CO2:N2 was 5:50:45 for M1, 5:70:25 for M2, and 5:90:5 for M3. All samples were stored at 4°C for 30 days. Microbiological, chemical and sensory analyses were performed every 3 days.
pH. The marinated green mussels were blended with sterilized distilled water at the ratio of 1:5 and allowed to stand for 2 min. Then pH was measured by using a pH meter (Mettler 350, Singapore).
Thiobarbituric acid reactive substances (TBARS). The thiobarbituric acid reactive substances (TBARS) were determined by the method described in [15]. 10 g of chopped samples were homogenized with 50 ml of distilled water for 2 min and then transferred to distillation tubes. 47.5 ml of distilled water was additionally infused into the distillation tube to avoid losing the sample. 2.5 ml of 4 N HCl and a few drops of an antifoam agent were added. 5 ml from 50 ml of the distillate were mixed with 5 ml of 0.02 M 2-thiobarbituric acid in 90% glacial acetic acid (TBA reagent). The mixture was placed in a vial that was capped and heated in a water bath for 35 min. Afterwards, the vial was cooled down and the sample was measured at 532 nm using a spectrophotometer (UV/VIS T180, PG Instrument Ltd., UK). The results are expressed in mg malondialdehyde/kg sample.
Glycogen content. Glycogen content was determined based on the method of [16]. 50 mg of chopped marinated green mussels were boiled for 20 min with 400 μl of 33% KOH, cooled down, and 700 μl of 96% ethanol was added. The mixture was placed in an ice bath for 2 hours to reach complete precipitation. Thereafter, the mixture was centrifuged at 7,500 g for 20 min using a microlitre centrifuge (Z-233-M2, Hermle–Germany) and the supernatant was collected. The supernatant was mixed with 1 ml of distilled water and sonicated for 5 min using Crest Ultrasonic (575 DAE, Malaysia). Then 100 μl of the solution and 2 ml of anthrone reagent were kept at 90°C for 20 min in darkness. The reagent consisted of 38 ml of concentrated sulphuric acid with 15 ml of distilled water and 0.075 g of anthrone. The solution was measured at 620 nm using a spectrophotometer. Glycogen concentration was calculated from a standard curve for glycogen from Sigma–Aldrich Company (Steinheim, Germany).
Total volatile base nitrogen (TVB – N). Total volatile base (TVB – N) contents in the green mussels were determined using Conway micro-diffusion method described in [17]. 2 g of a sample were mixed with 8 ml of 4% tricloroacetic acid (TCA), then homogenized with a high-speed homogenizer for 1 min. The supernatant was filtered by Whatman No. 41 filter paper (Whatman International, Ltd., Maidstone, UK). 1 ml of the sample was placed in the outer ring, while 1% boric acid containing the Conway indicator was pipetted into the inner ring. 1 ml of saturated K2CO3 was added to the sample to initiate the reaction after mixing. Thereafter, the mixture was incubated at room temperature for 3 hours. Afterwards, the inner ring solution was titrated using 0.02N HCl until the green color turned to pink similar to a blank sample. TVB-N content was calculated as follows, mg/100 g:
![]()
where: N is normality of HCl in titrant; A is titration volume for actual sample; B is titration volume for blank; V is total initial volume of sample with TCA.
Iron and zinc content. Samples of 0.5 g in mass were digested in 5 ml of concentrated nitric acid with 2 ml of 30% hydrogen peroxide under pressure in a closed vessel before heating in a microwave oven until the samples were digested. Afterwards, the samples were cooled down and filtered by Whatman filter paper No. 1 with a pore size of 110 nm (Whatman International, Ltd., Maidstone, UK)). Each sample solution was then transferred into a 25 ml volumetric flask and the volume was made up with distilled water [18]. The extract samples were used to determine iron and zinc by Inductively Couple Plasma Mass Spectrometry (ICP-MS) (Perkin-Elmer SCIEX, Shelton, CT, USA). The RF power was 1,350 W, the plasma gas flow rate was 15 l/min, the carrier gas flow was 0.94 l/min, and make-up gas flow was 0.15 l/min. The sample take rate was 100 μl/min and the sample depth was 6.0 mm.
Microbiological quality. 25 g of the marinated green mussels were blended with 225 ml of 0.1% sterilized peptone water. From the 10–1 dilution, dilutions to 10–2 and 10–3 were prepared with 0.1% sterilized peptone water. Mesophilic and psychrophilic bacteria, Escherichia coli, Salmonella spp, Vibrio cholera, and Staphylococcus aureus were determined using the method in [19]. Lactic acid bacteria were determined using de Man Rogosa and Shape agar by pour plate method, with incubation in anaerobic conditions at 35°C for 48 hours before colony count [20].
Sensory evaluation. Marinated samples were baked in a casserole pan (House worth HW-1707S, China) at 180°C for 10 min until the core temperature of the meat sample reached 80 ± 2°C. Then the samples were served to fifty untrained panelists of post-graduate students and technicians from the Department of Food Technology, Prince of Songkla University. The panelists were asked to evaluate the likability scores for appearance, aroma, texture, taste and overall on a nine-point hedonic scale.
Statistical analyses. All experiments were run in triplicates. The data were subjected to Analysis of Variance (ANOVA) and the differences between means were assessed for significance by Duncan’s Multiple Range Test [21]. Data analysis was performed with the SPSS package (SPSS 6.0 for Windows, SPSS Inc., Chicago, IL, USA).
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
We found that the sauce had pH 3.31, total titratable acidity 32.13%, and water activity 0.87. The viscosity of this enriched sauce was 840 cP, with non-Newtonian behavior. The colour coordinates L*, a*, and b* were 26.44, 15.8 and 33.7, respectively.
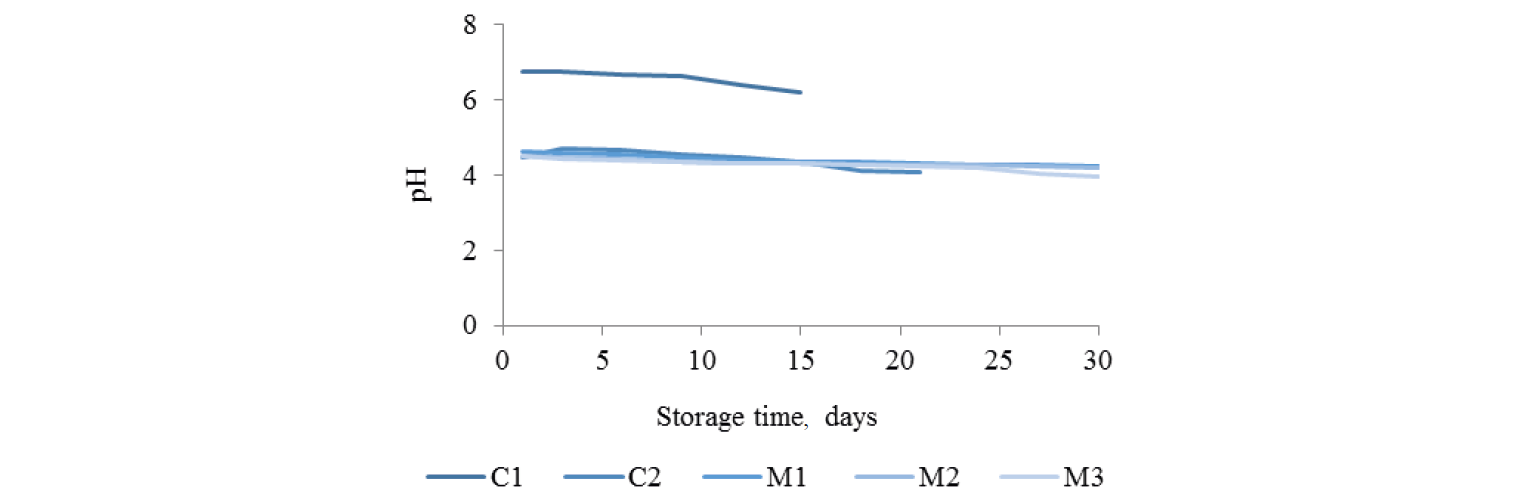
pH. pH levels of un-treated and treated green mussels are shown in Fig. 1. In general, pH with all treatments decreased with storage time. The initial pH of un-treated green mussels (C1) was 6.77 and decreased to 6.21 at 15 days of storage (p < 0.05). The marinated samples had significantly lower pH than the control group due to acidity of the sauce (pH 3.31) (p < 0.05). The pH of marinated green mussels without gases (C2) was 4.48 on day 1 and declined to 4.07 by day 21 of storage (p < 0.05). The initial pH of marinated green mussel (M1, M2, and M3) was about 4.51–4.64 and dropped to 4.24, 4.2 and 3.95, respectively, at 30 days of storage (p < 0.05). The decrease in pH of meat during chilled storage is normally caused by conversion of glycogen to lactic acid and other volatile acids, as a result of muscle degradation [22]. Cao et al. also reported that the spoilage of mollusk shellfish was partly by fermentation indicated by decreasing pH [23]. Moreover, pH was lower with MAP using gases than with other treatments, due to the dissolution of CO2 that produced carbonic acid [24] and reduced production of alkaline compounds in the marinated samples [24, 25]. Higher concentrations of CO2 (M3 > M2 > M1) gave lower pH levels in our experiments, although without significant differences. The pH value in our experiments was mainly due to the acidic marinade sauce with tamarind pulp.
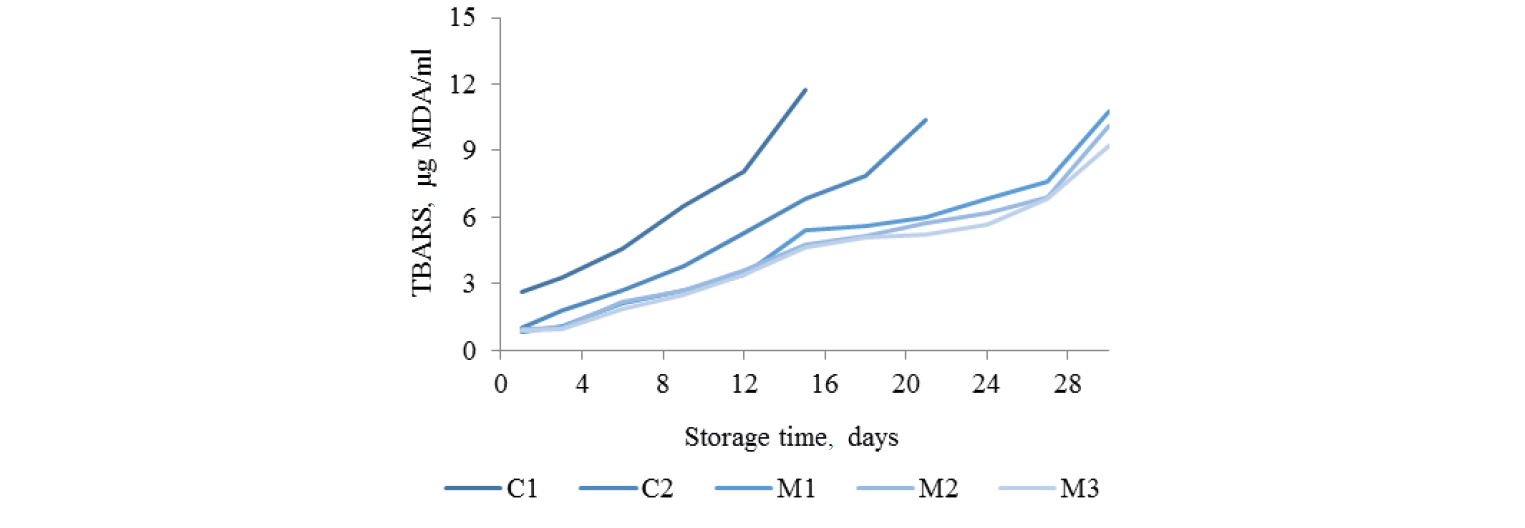
TBARS. Fig. 2 demonstrates the TBARS levels in green mussels during storage for the various treatments. TBARS is indicator of the malondialdehyde content, which is a secondary lipid oxidation product from polyunsaturated fatty acids [26] causing off-flavors in meat products [27]. The initial TBARS of the control (C1) was 2.62 mg MDA/kg, while the marinated samples (C2, M1, M2, M3) had 0.99, 0.81, 0.92, 0.88 mg MDA/kg, respectively. It should be noted that the marinated green mussels had lower TBARS values than the control samples, apparently due to osmosis during marinating conveying TBARS out from the mussels. Also the bacterial count was higher for C1 than for the other samples, which could also play a role in producing TBARS [28]. The lower TBARS values of the marinated green mussels were consistent throughout the storage, and sulfur compounds from garlic in the marinade may have played a role as antioxidants [29]. However, TBARS in all cases increased during storage because of high content of polyunsaturated fatty acids in mussel fat, as mentioned in the introduction. Moreover, MAP packaging reduced TBARS may be due to O2-elimination by enriched CO2 proportion [22]. Masniyom et al. reported that seabass flesh in MAP did not suffer from increased lipid oxidation [30]. The TBARS level of 5–8 mg/kg malonaldehyde is a standard criterion of the poor quality of oily food [31, 32]. TBARS in the control (C1) reached 8.03 mg MDA/kg on day 12 of storage, while C2 had 7.85 mg MDA/kg on day 18. M1, M2, and M3 had TBARS levels below the standard on day 27, namely 7.59, 6.89, and 6.82 mg MDA/kg, respectively. The marinated green mussels packed at the percentage of O2:CO2:N2 equaled 5:90:5 gave the least TBARS level at the end of storage (p > 0.05).
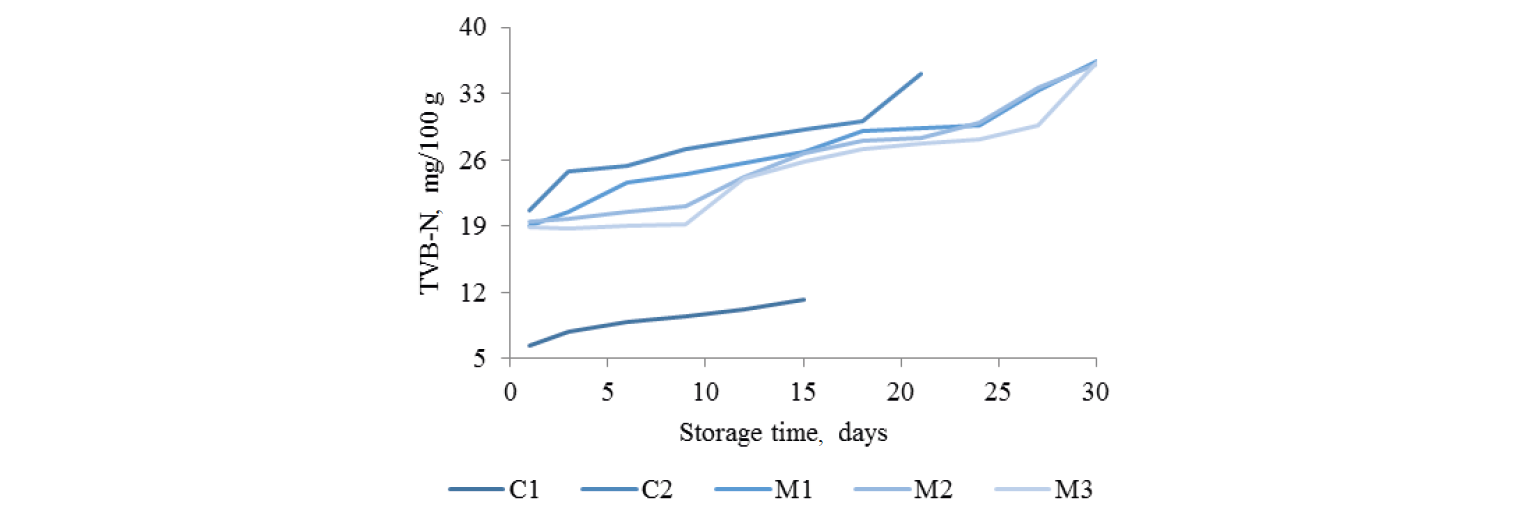
Total volatile base nitrogen (TVB-N). Changes in the TVB-N value of the treated and un-treated green mussels during storage are shown in Fig. 3. The TVB-N increased with storage time. The initial TVB-N of green mussels (C1) was 6.44 mg/100 g and increased gradually to 10.22 mg/100 g during storage. The treated samples C2, M1, M2, and M3 had initial TVB-N values 20.6, 18.97, 19.53 and 18.94 mg/100 g, respectively. By the end of the storage they reached 30.1, 33.33, 33.67, and 29.69 mg/100 g, respectively (p < 0.05). TVB-N is composed of volatile amines, typically dimethylamine, trimethylamine and ammonia [22], that commonly determine the spoilage of seafood [33]. The marinated samples had higher TVB-N values than the control even on day 1, due to volatile amines in the fish sauce [34]. In addition, the marinade contained garlic with sulfur compounds that could also react with boric acid, contributing to TVB-N content [29]. The increasing trend of TVB-N with time in this study agrees with the findings of [35]. Fishery products are acceptable for human consumption when TVB-N does not exceed 30 mg/100 g of sample [34]. Based on this, the shelf-lives of M3, M2, and M1 samples were 27, 24, and 25 days, respectively. In addition, [36] noticed that for seafood TVB-N indicated the freshness of raw materials and acceptability for human consumption. They pointed out that TVB-N increased with storage time as a result of quality deterioration. However, the initial TVB-N of the marinated products may not indicate the quality, considering that fish sauce and garlic used as ingredients cause erroneous results.
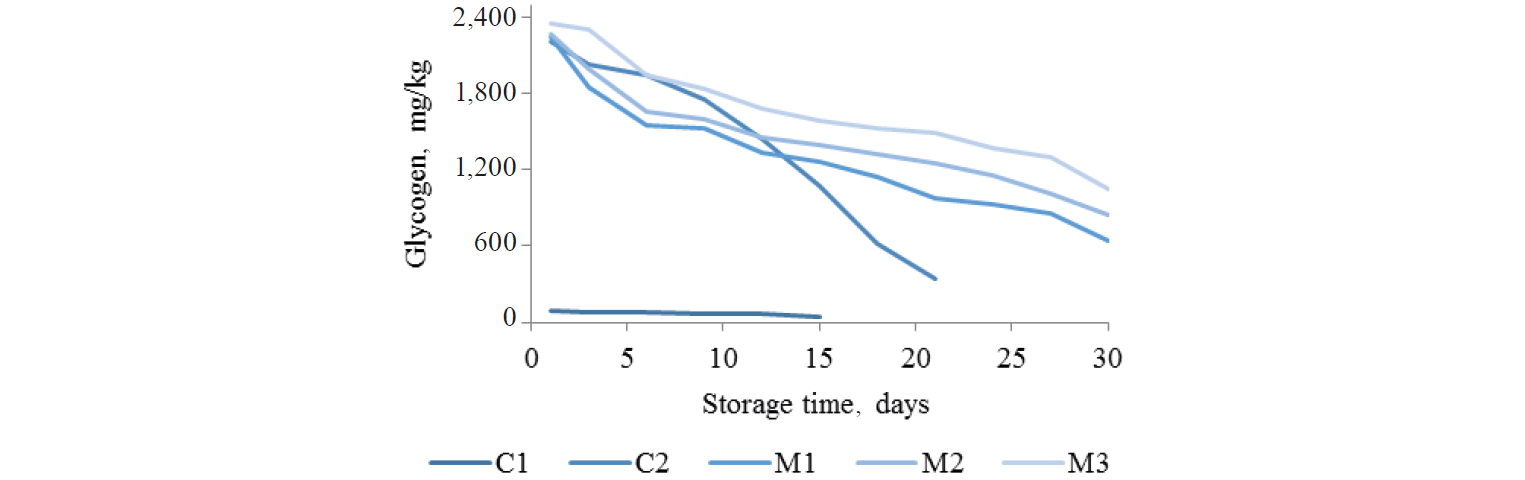
Glycogen content. During storage glycogen content decreased gradually in all samples, but declined rapidly in C2 samples (p < 0.05), as shown in Fig. 4. The initial glycogen content in the control sample (C1) was significantly lower than that in the marinated samples (C2, M1, M2, and M3). Glycogen is a polysaccharide including glucose units in a branched structure. Extraction and hydrolysis were used to obtain simple sugars in the glycogen content determination. As mentioned earlier, the marinade solution had 37% of sugar, so it positively contributed to the glycogen content. Jermyn [37] mentioned that in the determination of carbohydrates by anthrone method, other simple sugars in the system interfered. He pointed out that using glycogen content for glycolysis determination, which should decrease with storage time, might not give good results for high sugar products. The glycogen content decreased with storage time due to metabolism by glycolysis pathway in some bacteria [22]. The glycogen content of the control sample (C1) dropped to 61.42 mg/kg by day 12 of storage, while the marinated without MAP sample (C2) had 614.01 mg/kg after 18 days (p < 0.05). In the marinated samples packed with gases (M1, M2, and M3) the glycogen contents were 857.33; 1,005.68; and 1,293.17 mg/kg. The marinade with sugar strongly increased glycogen levels in marinated products. However, in C1the glycogen content decreased from 94.75 to 61.42 mg/kg during 12 days. The rapid decrease in glycogen levels of C2 may due to faster bacterial growth than with the treatments that included MAP, and the garlic from marinade solution seems to accelerate glycogen loss relative to the control (C1) perhaps by sulfur compounds and prebiotics in garlic that support lactic acid bacterial growth [22].
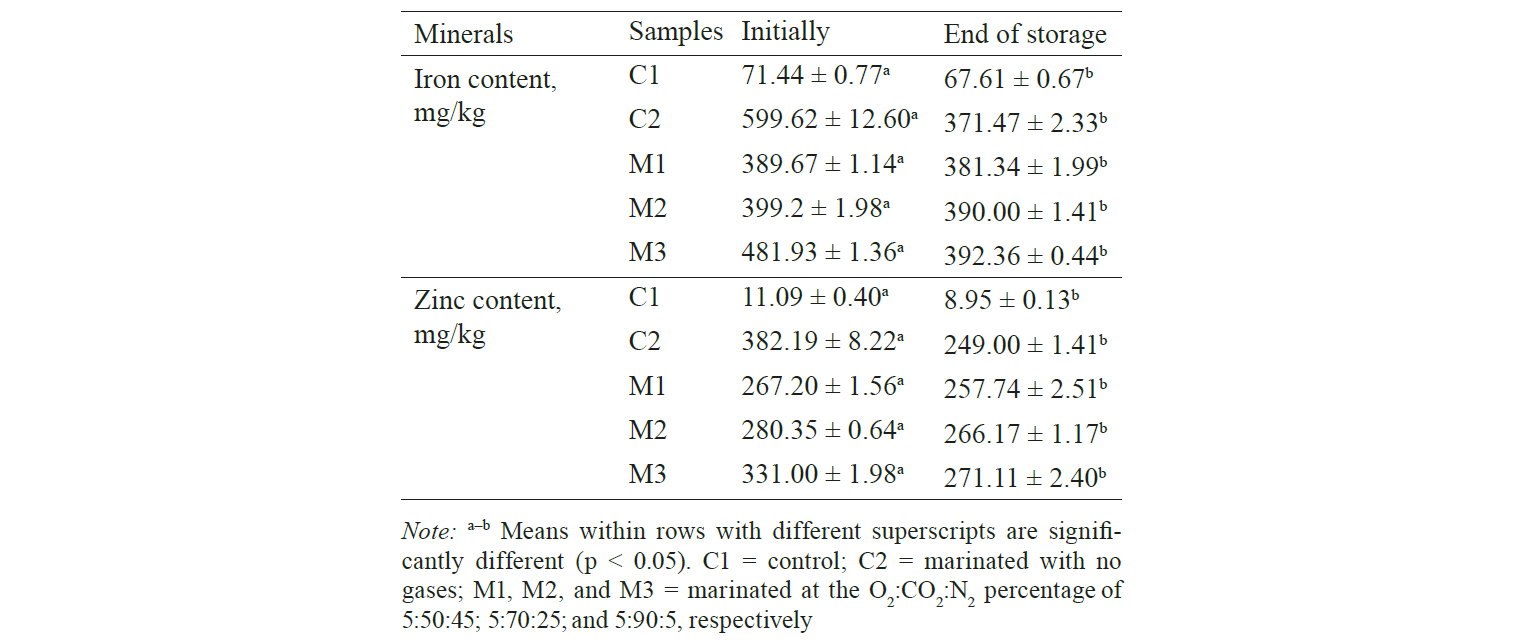
Iron and zinc content. Iron and zinc contents of green mussels with various treatments are presented in Table 1. On day 1, the iron content in C1 was 71.44 mg/kg, while that in the marinated samples C2, M1, M2, and M3 was 599.62; 389.67; 399.2; and 481.93 mg/kg, respectively. The initial zinc contents in C1, C2, M1, M2, and M3 were 11.09; 382.19; 267.2; 280.35; and 331 mg/kg, respectively. The iron and zinc contents may have fluctuated due to uncontrolled factors, such as uneven marinade absorption, the amount of marinade drip from the surface of green mussels during sample preparation for testing, or the small weight (1.0 g) of samples tested. If the samples are not uniform, then consistent results may not be obtained. The iron and zinc content in the marinated samples was higher than that in the control sample due to the absorption of iron and zinc from the marinade. Generally, the iron and zinc contents decreased by the end of the shelf-life to 67.61; 371.47; 381.34; 390.00; and 392.36 mg/kg for C1, C2, M1, M2, and M3, respectively. The zinc contents similarly were 8.95; 249.00; 257.74; 266.17; and 271.11 mg/kg for the same samples. The decrease in iron and zinc contents during storage may be due to the degradation of proteins by browning reaction, which may easily interact with iron and zinc.
El-Din et al. [38] mentioned that when iron and zinc minerals were added into dairy products, such as milk, the iron was bound with amino acids into casein micelles. Zinc associated with colloidal calcium phosphate in the casein micelles. Therefore, the contents of available iron and zinc may be reduced. In addition, it was noted that the drip increased with storage time due to high salt content (~ 5.5% NaCl) and acidity of the marinade, leaching iron and zinc out from the mussels. Baygar et al. [39] observed that water content decreased during marinating.
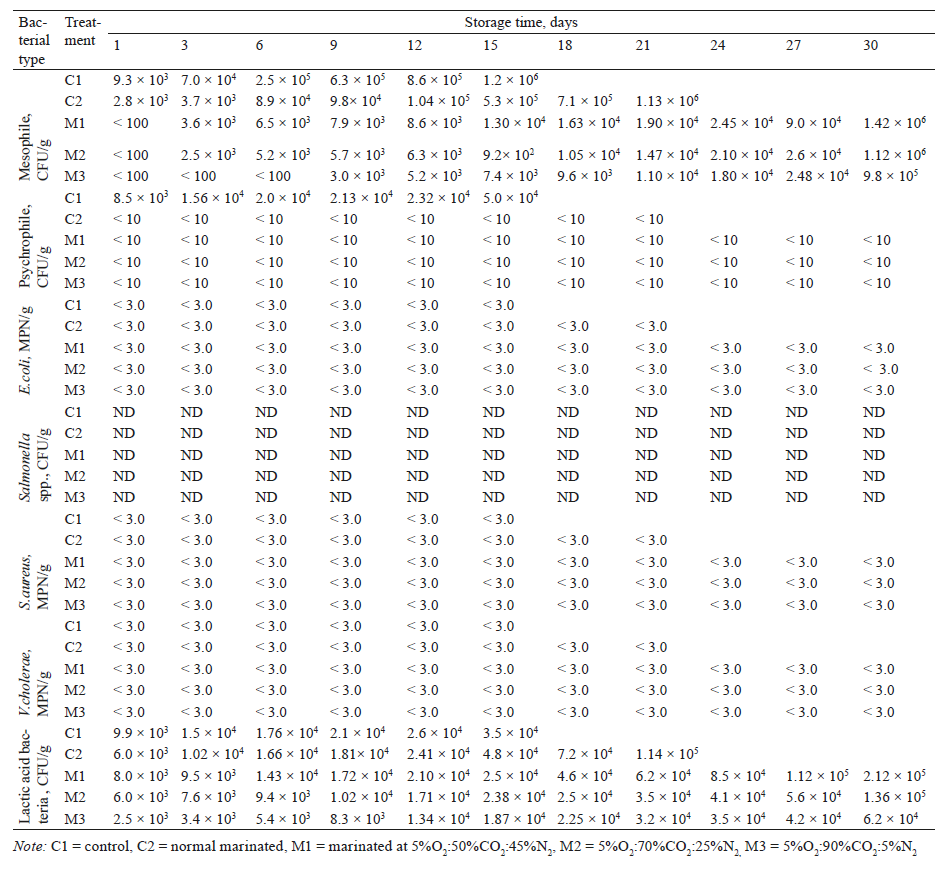
Microbiological quality. Mesophilic and lactic acid bacteria were found in all treated samples throughout the experiment. However, psychrophilic bacteria were found only in the control sample (C1), while Escherichia coli, Staphylococcus aureus, Salmonella spp., and Vibrio cholera were very low or not detected, as shown in Table 2. Psychrophilic bacteria in the marinated samples (C2, M1, M2, and M3) were present in the amount of no more than 10 CFU/g. This indicates that the hurdle effect of the sauce (aw 0.87, pH 3.31, TA 32.13%, and salt 5.26%) strongly controlled psychrophilic bacterial growth. During storage, mesophilic and lactic acid bacteria increased with time (p < 0.05). Not surprisingly that the bacterial count in the control sample (C1) was the highest, followed by the marinated samples (C2), (M1), (M2) and (M3) in this order. ICMSF [40] recommended 106CFU/g as the standard limit of total viable count in freshwater fish and seafood. By this threshold the shelf-life of the control was 12 days, while the marinated green mussel (C2) had 18 days. With higher CO2 contents lower bacterial counts were found. This suggests that MAP can prolong the shelf-life when CO2 inhibits microbial growth. Farber [25] also stated that CO2 becomed more antibacterial as its concentration increased. CO2 can decarboxylate enzymes and inhibit the metabolic activity of microflora, thereby extending the lag phase and reducing logarithimic phase growth of spoilage bacteria [25, 41, 12]. Goulas et al. [42] reported that when mussels (Mytilus galloprovincialis) were kept in 80%CO2:20%N2 atmosphere, the total viable count of bacteria was retarded compared to other tested conditions. Masniyom et al. [43] also reported that MAP with 80–100% of CO2 effectively extended the shelf-life of green mussels (Perna viridis). Our current study indicates that 90% CO2 atmosphere is the most effective to prolong the shelf-life of marinated green mussels. Therefore, combined acidity, salt and atmospheric CO2 were effective in retarding microbial growth.
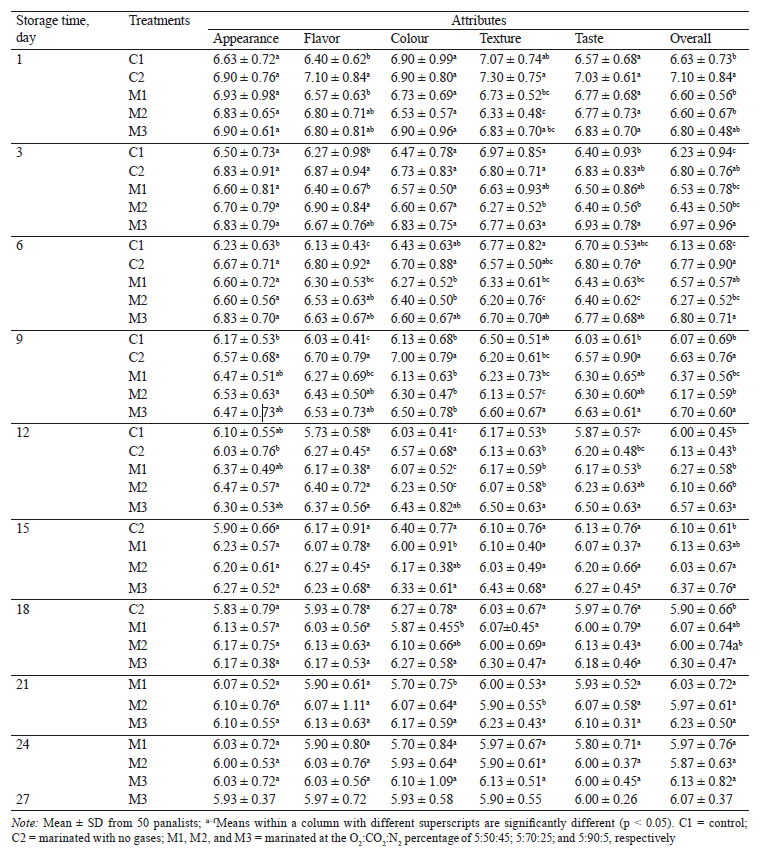
Sensory evaluation. Generally, sensory evaluation is applied in the estimation of seafood quality and correlates well with the microbiological and chemical analyses [44]. The sensorial scores from fifty non-trained panelists for green mussels with the various treatments during storage are summarized in Table 3. The scores from panelists became worse with storage time (p < 0.05). The threshold score for rejection was chosen to be 6, and only aroma showed significant differences between un-treated and treated green mussels during storage (Table 3). While all the sensory attributes with any treatment degraded towards the end of shelf-life, the scores remained on an acceptable level. Unexpectedly, the marinated mussels did not score better than the control group. This may be due to too soft texture, and unusual aroma and taste from the sauce that was highly acidic and contained iron and zinc. This altered the good natural aroma and texture of the mussels. This type of sauces is served as dips in Vietnam, not as marinades.
ВЫВОДЫ
TVB-N, TBARS, TVC, and lactic acid bacteria increased in all samples during storage, while pH, glycogen, iron, and zinc contents decreased. The shelf-life of marinated green mussel was extended from 12 to 18, 24, or 27 days based on microbiological quality, TBARS and TVB-N value, by use of CO2 rich MAP packaging. For marinated green mussels, high concentration of CO2 (90%) appears to be both feasible and beneficial to shelf-life.
КОНФЛИКТ ИНТЕРЕСОВ
The authors declare no conflict of interest.
БЛАГОДАРНОСТИ
This work was supported by the Higher Education Research Promotion and the Thailand’s Education Hub for Southern Region of ASEAN Countries Project Office of the Higher Education Commission.
We would like to thank the team of the Research and Development Office (RDO) of the Prince of Songkla University, Thailand for the assistance with English.
СПИСОК ЛИТЕРАТУРЫ
- Srisunont C. and Babel S. Uptake, release, and absorption of nutrients into the marine environment by green mussel (Perna viridis). Marine Pollution Bulletin, 2015, vol. 97, no. 1–2, pp. 285–293. DOI: https://doi.org/10.1016/j.mar- polbul.2015.06.004.
- Karakoltsidis P.A., Zotos A., and Constantinides S.M. Composition of the Commercially Important Mediterranean Finfish, Crustacteristics and Molluscs. Journal of Food Composition and Analysis, 1995, vol. 8, no. 3, pp. 258–273. DOI: https://doi.org/10.1006/jfca.1995.1019.
- Vareltzis K. Mussel as Food. Fishing News, 1996, no.11, pp. 38–47.
- Orban E., Di Lena G., Nevigato T., et al. Seasonal changes in meat content, condition index and chemical composi- tion of mussels (Mytilus galloprovincialis) culture in two different Italian sites. Food Chemistry, 2002, vol. 77, no. 1, pp. 57–65. DOI: https://doi.org/10.1016/S0308-8146(01)00322-3.
- Kronhout D., Bosschieter E.B., and Lezenne C.C. The inverse relationship between fish consumption and 20-yearmortality from coronary heart disease. New England Journal of Medicine, 1985, vol. 312, no. 19, pp. 1205–1209.
- Aktas N.E., Aksu M.I., and Kaya M. The effect of organic acid marination on tenderness cooking loss and bound water content of beef. Journal of Muscle Foods, 2003, vol. 14, no. 3, pp. 181–194. DOI: https://doi. org/10.1111/j.1745-4573.2003.tb00699.x.
- Zheng M., Huang Y.W., Nelson S.O., Bartley P.O., and Gates K.W. Dielectric Properties and Thermal Conductivity of Marinated Shrimp and Channel Fish. Journal of Food Science, 1998, vol. 63, no. 4, pp. 668–672. DOI: https://doi. org/10.1111/j.1365-2621.1998.tb15809.x.
- Carlos A.M.A. and Harrison M.A. Inhibition of Selected Microorganisms in Marinated Chicken by Pimento Leaf Oil and Clove Oleoresin. The Journal of Applied Poultry Research, 1999, vol. 8, no. 1, pp. 100–109. DOI: https://doi. org/10.1093/japr/8.1.100.
- Hull R. Marination overview. Meat & Poultry Marination Short Course. Athens, Georgia, 2001.
- Sheard P.R., Nute G.R., Richardson R.I., and Wood J.D. Effects of breed and marination on the sensory attributes of pork from Large White and Hampshire-sired pigs. Meat Science, 2005, vol. 70, no. 4, pp. 699–707. DOI: https://doi. org/10.1016/j.meatsci.2005.03.006.
- Stiles M.E. Scientific Principles of Controlled/Modified Atmosphere Packaging. In: Ooraikul B. and Stiles M.E. (eds) Modified Atmosphere Packaging of Food. London: Ellis Horwood Publ., 1991, pp. 18–25. DOI: https://doi. org/10.1007/978-1-4615-2117-4_2.
- Pantazi D., Papavergou A., Pourins N., Kontominas M.G., and Savvaidis I.N. Shelf-life of chilled fresh Mediterranean swordfish (Xiphias gladius) stored under various packaging conditions: microbiological, biochemical and sensory attributes. Food Microbiology, 2008, vol. 25, no. 1, pp. 136–143. DOI: https://doi.org/10.1016/j.fm.2007.06.006.
- Sperber W.H. Requirements of Clostridium botulinum for growth and toxin production. Food Technology, 1982, vol. 36, no. 12, pp. 89–94.
- Whiting R.C. and Naftulin K.A. Effect of Headspace Oxygen Concentration on Growth and Toxin Production by Pro- teolytic Strains of Clostridium botulinum. Journal of Food Protection, 1992, vol. 55, no. 1, pp. 23–27. DOI: https:// doi.org/10.4315/0362-028X-55.1.23.
- Buege J.A. and Aust S.D. Microsomal lipid peroxidation. Methods in Enzymology, 1978, vol. 52, pp. 302–310. DOI: https://doi.org/10.1016/S0076-6879(78)52032-6.
- Rosa R. and Nunes M.L. Tissue biochemical composition in relation to the reproductive cycle of deep-sea decapod Aristeus antennatus in the Portuguese south coast. Journal of the Marine Biological Association of the United King- dom, 2003, vol. 83, no. 5, pp. 963–970. DOI: https://doi.org/10.1017/S0025315403008154h.
- Conway E.J. and Byrne A. An absorption apparatus for the micro-determination of certain volatile substances: The micro-determination of ammonia. Biochemical Journal, 1933, vol. 27, no. 2, pp. 419–429.
- AOAC. 2012. Official methods of analysis of AOAC international. 19th ed. The association of official analyticalchemists, Inc.
- BAM. Bacteriological Analytical Manual. U.S. Department of Health and Human Services. U. S. Food and Drug Administration Center of Food Safety and Applied Nutrition, 2001, n.p.
- De Man J.C., Rogosa D., and Sharpe M.E. A medium for the cultivation of lactobacilli. Journal of applied Bacteriol- ogy, 1960, vol. 23, no. 1, pp. 130–135. DOI: https://doi.org/10.1111/j.1365-2672.1960.tb00188.x.
- Steel R.G.D. and Torrie J.H. Principles and procedures of statistics: A Biometrical Approach. 2nd ed. New York: McGraw-Hill Publ., 1980. 633 p.
- Bunruk B. Effect of Sa-Tay marinade on quality changes and safety of oyster (Crassostrea belcheri) meat during chilled storage. Doctoral dissertation, Thailand, 2013.
- Cao C., Love G.D., Hays L.E., et al. Biogeochemical evidence for euxinic oceans and ecological disturbance presag- ing the end-Permian mass extinction event. Earth and Planetary Science Letters, 2009, vol. 281, no. 3–4, pp. 188–201. DOI: https://doi.org/10.1016/j.epsl.2009.02.012.
- Sivertsvik M., Rosnes J.T., and Jeksrud W.K. Solubility and absorption rate of carbon dioxide into non-respiring foods. Part 2: Raw fish fillets. Journal of Food Engineering, 2004, vol. 63, no. 4, pp. 451–458. DOI: https://doi. org/10.1016/j.jfoodeng.2003.09.004.
- Farber J.M. Microbiological Aspects of Modified Atmosphere Packaging Technology – A review. Journal of Food Protection, 1991, vol. 54, no. 1, pp. 58–70. DOI: https://doi.org/10.4315/0362-028X-54.1.58.
- Goulas A.E. and Kontominas M.G. Combined effect of light salting, modified packaging and oregano essential oil on the shelf-life of sea bream (Sparus aurata): Biochemical and sensory attributes. Food Chemistry, 2007, vol. 100, no. 1, pp. 287–296. DOI: https://doi.org/10.1016/j.foodchem.2005.09.045.
- Juntachote T., Berghofer E., Siebenhandl S., and Bauer F. The antioxidative properties of Holy basil and Galangal in cooked ground pork. Meat Science, 2006, vol. 72, no. 3, pp. 446–456. DOI: https://doi.org/10.1016/j.meats- ci.2005.08.009.
- Ruiz-Capillas C. and Moral A. Residual effect of CO on hake (Merluccius merluccius L.) stored in modified and controlled atmospheres. European Food Research and Technology, 2001, vol. 212, no. 4, pp. 413–420. DOI: https:// doi.org/10.1007/s002170000270.
- Pakawatchai C., Siripongvutikorn S., and Usawakesmanee W. Effect of Herb and Spice Pastes on the Quality Changes in Minced Salmon Flesh Waste during Chilled Storage. Asian Journal of Food and Agro-Industry, 2009, vol 2, no 4, pp. 481–492.
- Masniyom P., Benjakul S., and Visessanguan W. Combination effect of pyrophosphate and modified atmosphere on quality and shelf-life extension of refrigerated seabass slices. LWT – Food Science and Technology, 2005, vol. 38, no. 7, pp. 745–756. DOI: https://doi.org/10.1016/j.lwt.2004.09.006.
- Shamberger R.J., Shamberger B.A., and Willis C.E. Malonaldehyde Content of Food. The Journal of Nutrition, 1977, vol. 107, no. 8, pp. 1404–1409. DOI: https://doi.org/10.1093/jn/107.8.1404.
- Bergamo P., Fedele E., Balestrieri M., Abrescia P., and Ferrara L. Measurement of Malondialdehyde Levels in Food by High-Performance Liquid Chromatography with Fluorometric Detection. Journal of Agriculture and Food Chemistry, 1988, vol. 46, no. 6, pp. 2171–2176. DOI: https://doi.org/10.1021/jf9709919.
- Smaoui S., Ben Hlima H., and Ghorbel R. The effect of sodium lactate and lactic acid combinations on the microbial, sensory, and chemical attributes of marinated chicken thigh. Poultry Science, 2012, vol. 91, no. 6, pp. 1473–1481. DOI: https://doi.org/10.3382/ps.2011-01641.
- Park J.N., Fukumoto Y., Fujita E., et al. Chemical composition of fish sauces produced in Southeast and East Asian countries. Journal of Food Composition and Analysis, 2001, vol. 14, no. 2, pp. 113–125. DOI: https://doi.org/10.1006/ jfca.2000.0963.
- Bono G., Okpala C.O.R., Alberio G.R., et al. Toward shrimp consumption without chemicals: combined effects of freezing and modified atmosphere packaging (MAP) on some quality characteristics of giant red shrimp (Aristae- omorpha foliacea) during storage. Food Chemistry, 2016, vol. 197, pp. 581–588. DOI: https://doi.org/10.1016/j. foodchem.2015.10.146.
- Büyükcan M., Bozoglu F., and Alpas H. Preservation and shelf-life extension of shrimps and clams by high hydrostat- ic pressure. International Journal of Food Science & Technology, 2009, vol. 44, no. 8, pp. 1495–1502. DOI: https:// doi.org/10.1111/j.1365-2621.2007.01628.x.
- Jermyn M.A. Increasing the sensitivity of the anthrone method for carbohydrate. Analytical Biochemistry, 1975, vol. 68, no. 1, pp. 332– 335. DOI: https://doi.org/10.1016/0003-2697(75)90713-7.
- El-Din A.M.G., Hassan A.S.H., El-Behairy S.A., and Mohamed E.A. Impact of zinc and iron salts fortification of buf- falo’s milk on the dairy product. World Journal of Dairy & Food Sciences, 2012, vol. 7, no. 1, pp. 21–27. DOI: https:// doi.org/10.5829/idosi.wjdfs.2012.7.1.1102.
- Baygar T., Alparslan Y., Guler M., and Okumus M. Effect of Pickling Solution on Maturing and Storage Time of Marinated Sea Bass Fillets. Asian Journal of Animal and Veterinary Advances, 2010, vol. 5, no. 8, pp. 575–583. DOI: https://doi.org/10.3923/ajava.2010.575.583.
- ICMSF. Sampling plans for fish and shellfish. In ICMSF, Microorganisms in Foods. Sampling for Microbiological Analysis: Principles and Scientific Applications Vol. 2. 2nd edition, International Commission on Microbiological Specifications for Foods, Toronto, Canada: University of Toronto Press, 19986, n.p.
- Lalitha K.V., Sonaji E.S., Manji S., et al. Microbiological and biochemical changes in pearl spot (Etroplus suratensis Bloch) stored under modified atmospheres. Journal of Applied and Microbiology, 2005, vol. 99, no. 5, pp. 1222–1228. DOI: https://doi.org/10.1111/j.1365-2672.2005.02694.x.
- Goulas A.E., Chouliara I., Nessi E., Kontominas M.G., and Savvaidis I.N. Microbiological, biochemical and sensory assessment of mussels (Mytilus galloprovin-cialis) storage under modified atmosphere packaging. Journal of Applied and Microbiology, 2005, vol. 98, no. 3, pp. 752–760. DOI: https://doi/org/10.1111/j.1365-2672.2004.02512.x.
- Masniyom P., Benjama O., and Maneesri J. Extending the shelf-life of refrigerated green mussel (Perna viridis) under modified atmosphere packaging. Songklanakarin Journal of Science and Technology, 2011, vol. 33, no. 2, pp. 171–179.
- Karungi C., Byaruhanga Y.B., and Muyonga J.H. Effect of pre-icing duration on quality deterioration of iced Nile perch (Lates niloticus). Food chemistry, 2004, vol. 85, no. 1, pp. 13–17. DOI: https://doi.org/10.1016/S0308- 8146(03)00291-7.