Аннотация
Oyster mushroom (Pleurotus ostreatus L.) is a valuable food product. It possesses an antiatherogenic potential, which has to be preserved during processing. The paper features the production of oyster mushroom sublimates. It focuses on such pre-treatment conditions as grinding, disinfection, and cryostabilisation, and their effect on the antiatherogenic potential of oyster mushrooms. A set of in vitro experiments was performed to measure the levels of lovastatin and antioxidant, catalase, anti-inflammatory, and thrombolytic properties. Various pre-treatment conditions proved to produce different effects on the biological activity of the freeze-dried oyster mushroom product. The best results were obtained after the mushrooms were reduced to pieces of 0.5 cm, underwent UV disinfection, blanched, treated with hot air, and cryostabilised with a 1.5% apple pectin solution. The best conditions for the antioxidant properties included ozonation, UV disinfection, and cryoprotection with pectin. The critical conditions for the antioxidant properties included homogenisation, blanching, and cryostabilisation with 10% solutions of sucrose and lactose. The catalase properties did not depend on the degree of grinding and were most pronounced after ozonation. The optimal conditions for the anti-inflammatory properties included UV disinfection and cryostabilisation with lactose. Ozonation proved to be critical for anti-inflammatory properties. The optimal conditions for thrombolytic properties included ozonation and cryoprotection with a 5% sorbitol solution, while hot air disinfection proved critical. Therefore, the research provided an experimental substantiation for individual pre-treatment conditions or their combinations that turn sublimated oyster mushrooms into a valuable functional product with antiatherogenic properties.Ключевые слова
Oyster mushroom, freeze-drying, functional food, antiatherogenic potential, lovastatin, antioxidant properties, catalase properties, anti-inflammatory properties, thrombolytic effectВВЕДЕНИЕ
According to statistics, atherosclerosis and its complications remain the main cause of death worldwide [1]. The mechanisms of atherogenesis are complex and multiple. Its main causes include hyperlipidemia, oxidative stress, thrombosis, and inflammation [2].
Modern medicine is striving to find a way to curb this trend. Various therapeutic approaches are being introduced to combat atherosclerosis. However, many of them remain expensive and have various contraindications and side effects, which limits their clinical use [3]. As a result, more and more attention is given to functional food products with medicinal properties and minimal side effects. Scientists are looking for biologically active raw materials that could modify human metabolism and prevent the development and progression of atherosclerosis [4, 5].
In this regard, the oyster mushroom (Pleurotus ostreatus L.) is considered advantageous. Its fruit body has a high nutritional value, natural statin, and a whole complex of other biologically active substances (BAS) [6, 7]. Recent researches proved that the oyster mushroom possesses hypolipidemic, antioxidant, antiinflammatory, and thrombolytic properties [8–11], which makes it a valuable raw material. Thus, oyster mushrooms can help to improve the existing antiatherogenic functional foods and develop new ones. However, the concentration and effectiveness of biologically active compounds depend not only on cultivation conditions and age of the mushrooms, but also on the processing methods [12, 13].
Some of the existing food processing technologies make it impossible to preserve the entire complex of biologically active substances [14, 15]. Today, freezedrying is considered the least harsh and the most reliable treatment method of BAS production. It ensures stability of thermolabile and hydrolytically unstable substances, increases shelf life, and optimises storage conditions [16–18]. However, even when all the necessary regulations for freeze-drying have been observed, the properties of the product depend on the pre-processing conditions. An appropriate use of various pre-treatment methods significantly increases the efficiency of drying, improves the quality of the product, and preserves its properties [19–21]. A careless use of pre-treatment methods can lead to a decrease in the content of certain BAS in sublimates [22–24]. Thus, each raw material requires its own freeze-drying technology based on experimental data about the effect that pre-treatment conditions produce on the specific properties of the finished sublimates.
The antiatherogenic effects of freeze-dried oyster mushrooms have already become focus of scientific studies [25]. However, there have been no studies connected with the effect of pre-treatment methods on the preservation of BAS and natural antiatherogenic potential of sublimated oyster mushrooms, which adds to the relevance of the present research.
ОБЪЕКТЫ И МЕТОДЫ ИССЛЕДОВАНИЯ
The present research used the following chemicals: chloroform (CHCl3), hydroxyamine hydrochloride (NH2OH·HCl), reduced iron, perchloric acid (HClO4), ethanol (C2H5OH) (Sigma-Aldrich), microbiologically pure lovastatin (C24H36O5) (TEVA, Hungary), reagent (chromogen containing an ABTS•+ radical) (Institute of Bioorganic Chemistry, National Academy of Sciences of Belarus), trolox 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid, ammonium thiocinate (NH4NCS), ferrous chloride (FeCl2), oleic acid (C18H34O2), hydrogen peroxide (H2O2), monosubstituted potassium phosphate (KH2PO4), disubstituted sodium phosphate (sodium hydrogen phosphate 12-water, Na2HPO4·12H2O) (Sigma-Aldrich), dextrose (C6H12O6·H2O), sucrose (C12H22O11) (Sigma), monohydric citric acid (C6H8O7), sodium citrate (C6H5Na3O5), sodium chloride (NaCl), lactose (C12H22O11·H2O), and sorbitol (C6H14O6) (Sigma-Aldrich). All the substances were purchased from Diaem (Russia).
The study featured oyster mushroom (Pleurotus ostreatus L.), strain NK35 (SYLVAN, Hungary). It was harvested in 2018 and cultivated under the standard mushroom production conditions in the Stavropol Region. The fruit bodies were of the same size and maturity, undamaged. During the experiments, the fresh mushrooms were stored in a refrigerator at 5–7°C. Before the experiments, the fruit bodies were thoroughly washed under running water.
The antiatherogenic potential of the freeze-dried oyster mushroom product was evaluated in vitro based on the concentration of lovastatin, as well as antioxidant, catalase, anti-inflammatory, and thrombolytic properties.
The first stage featured the effect of the degree of preliminary grinding on the antiatherogenic properties of the sublimates. The grinding was conducted by reducing the fruit bodies into pieces with the side sizes of 2.0–2.5 cm and 0.5–1.0 cm. The pieces were homogenised using a laboratory Sterilmixer 12 (PBI, Italy) at No. 9 high-speed mode. Whole mushrooms served as control sample. The oyster mushroom samples were spread in one layer on separate stainless steel trays. The homogenised substance was poured into the trays to form an even layer with a thickness of ≤ 0.8–1 cm. All samples were frozen in a SE-45 refrigerator (TEFCOLD, Denmark) at –40°С for 72 h and subsequently freeze-dried.
The second stage tested the effect of preliminary disinfection methods on the preservation of antiatherogenic properties in the sublimates. The mushrooms were subjected to blanching, UV disinfection, ozonation, and hot air treatment [26, 27].
Blanching is one of the most common pre-treatment methods. It reduces microbial challenge and inactivates the enzymes that reduce the quality of the freeze-dried product. According to Galoburda et al., the optimal blanching temperature regime is 70–80°C, since it provides the best drying performance for mushrooms [28]. Hence, the oyster mushrooms were blanched in water at 70 °C for 3 min, cooled under running water, and drained in a sieve for several minutes.
The UV disinfection of the oyster mushrooms was performed using an Azov portable ultraviolet irradiator, modification OBN-35-01 UHL 4.2 (Russia). The fruit bodies were put on plastic trays in one layer, placed at a distance of 60 cm from the irradiator and treated for 15 min.
The ozonation was performed using a universal ozoniser of air and water Ozone OViV (Ukraine). The ozonation was carried out in a ventilation hood at 22°C in a 10-litre chamber improvised from PVC film. The ozonation mode was based on [29] and the operation manual: ozonator power, 100%; gas flow rate, 2.0 dm3/min; ozone concentration, 8 mg/dm3; exposure time, 20 min.
The hot air treatment was performed using a TS-1/80 SPU dry-air thermostat (Smolensk Special Design-Technological Bureau of Software Management Systems, Russia). The mushrooms were placed on a wire shelf and kept in the thermostat under forced ventilation at 60°C for 60 min.
The third stage assessed the effect of various cryoprotectors on the atherogenic potential of the oyster mushroom sublimates. The experiment involved natural substrates that are widely used in food industry: a 10% sucrose solution, a 10% lactose solution, a 5% sorbitol solution, and a 1.5% pectin solution. In all cases, the whole fruit bodies were soaked in aqueous solutions of the cryoprotectors (volume ratio = 1:20) for 30 min. The untreated oyster mushroom fruit bodies acted as control sample.
After the disinfections and cryostabilisations, the fruit bodies were placed on separate sheets, frozen, and freeze-dried.
All the samples were dried in an LS-500 freeze dryer (Prointech, Russia), which included a freeze dryer and a vacuum station. The glass lid of the drying chamber was covered with an opaque material to prevent degradation of antioxidants by photo-oxidation. The working pressure in the drying chamber was 80–90 Pa; the condenser temperature was 48–49°С. The temperature of the samples did not exceed 29–30°С during the entire drying process. The average drying time was 26–27 h.
The mushrooms were dehydrated until the residual moisture content was 6–8%. The moisture content in the dried oyster mushroom samples was measured using an MB 25 moisture content analyzer (Ohaus, China). The indicators were measured using the following automatic measurement mode: heating temperature = 75°C, measurement time = 5 min. The resulting oyster mushrooms sublimates were placed into a dry, hermetically sealed container and stored in dark at ≤ 25°C for further analysis.
The amount of lovastatin in the sublimates was estimated according to the authentic method using the hydroxam method after lovastatin had been extracted with chloroform and concentrated [30, 31]. The ground dried mushrooms were weighed into portions of 0.1–0.2 g, extracted with 5.0–10.0 cm3 of chloroform, and filtered. The filtrate was evaporated using a RV 10 Basic V rotary vacuum evaporator (IKA, Germany). The remaining filtrate was diluted with 1.0 cm3 of a 0.9 M alcohol alkaline solution of hydroxylamine and 5.0 cm3 of a 5.73 mM solution of ferric (III) chloride. After that, pH was adjusted to 1.2 ± 0.2 with a 2M hydrochloric acid solution. The extinction of the resulting magenta solution was measured using an SF-102 spectrophotometer (Research and Development Centre NPO INTEROFOTOFIKA, Russia) at a wavelength of λ = 513 nm. The calculation was performed according to the calibration curve.
To assess the antioxidant activity of the sublimates, we measured the radical absorption and the degree of inhibition of lipid peroxidation (LPO). To assess the radical absorption, the dry oyster mushrooms were made into powder. Then the powder was extracted with bidistilled water in a shaker at 50–60°C for 3 h. The rotation speed was 190 rpm. After that, the material was filtered as described in [32]. The antioxidant activity of the extract was determined in vitro using the OxiStat test system (Institute of Bioorganic Chemistry, National Academy of Sciences of Belarus). It was a one-stage assessment of reduction value of the resulting ABTS•+ radical by antioxidants. The scheme is described in [33] as follows: ABTS•+ + AO → ABTS + AO•+.
When antioxidants interacted with ABTS•+, the optical density of the solution of the cation radical fell down to 600–800 nm in proportion to the concentration and activity of the antioxidant. The optical density was measured using a spectrophotometer at a wavelength of 675 nm. The optical path length of the cuvettes was 1.0 cm.
To provide a quantitative assessment of the antioxidant activity, we used trolox, i.e. a standard antioxidant, which is a water-soluble analogue of vitamin E:
where:
АА – antioxidant activity;
ΔАо – optical density of the experimental sample;
ΔАc – optical density of the control sample (buffer);
Сst – standard concentration (trolox).
The radical absorption results were expressed in mg of trolox equivalent per gram of dry matter (mg TE/g).
To evaluate the LPO inhibition activity, 0.1 g of powdered dry mushrooms was added to 2.0 cm3 of bidistilled water. After 24 h of maceration at room temperature, the extract was filtered and centrifuged at 1300 rpm for 10 min. The LPO inhibition activity of the obtained extract was measured in an oleic acid emulsion system according to the slightly modified procedure described in [34]. 0.1 cm3 of the extract was added to 4.0 cm3 of phosphate buffer (50 mM, pH 7.0), and 0.1 cm3 of oleic acid was added to 4.0 cm3 of ethanol (95 wt%, aqueous solution). The total volume was brought to 10.0 cm3 with distilled water, mixed in a sealed conical tube with a screw cap, and incubated at 40°C in the dark for 7 days. The oxidation state was evaluated using iron thiocyanate at 24 h intervals. The reaction solution (100 μL) was mixed with 4.7 cm3 of ethanol (75 wt%, aqueous solution), 0.1 cm3 of an ammonium thiocyanate aqueous solution (30% w/v), and 0.1 cm3 of an iron chloride (II) solution (20 mM in 3.5% (v/v) HCl). After 3 min, the absorbance was measured at a wavelength of 500 nm using a UV spectrophotometer. An increase in optical density meant an increase in the level of oleic acid oxidation. Trolox (0.95 mmol/dm3) was used as a reference. The blank sample contained deionised water instead of the extract.
When calculating both the antiradical activity and the activity of LPO inhibition, we took into account the fact that the extracts had their own colour, which absorbed a particular wavelength in the visible spectrum.
The catalase activity of oyster mushroom sublimates was measured using a modified technique based on the Beers and Sizer spectrophotometric method [35]. This fast and accurate analysis presupposes a hydrogen peroxide dehydrogenation and determination of its loss at λ = 240 nm. The powdered oyster mushrooms were weighed into portions of 0.020–0.025 g and extracted with 5.0 cm3 of chilled 50 mM phosphate buffer with pH = 7.0 for 15 min in the cold with periodic stirring. The extract was filtered and centrifuged at 4°C for 20 min and acceleration of 1200 g. The experimental solution contained 1.0 cm3 of 50 mM phosphate buffer with pH=7.0 and 1.0 cm3 of 0.1% hydrogen peroxide. Its optical density (D1) was measured at 240 nm relative to the control solution, which contained 2.0 cm3 of 50 mM phosphate buffer. After filtration, 0.1 cm3 of the sublimate sample extract was introduced into the experimental sample. In addition, 0.1 cm3 of extract was added to the control solution. The optical density of the experimental solution was determined after 30 s (D2).
The enzymatic activity was calculated for 1 mmol of substrate (Н2О2) split in 1 min with 1 g of sublimate sample according to the following formula:
where:
А – enzymatic activity, mmol/g·min;
D1 – optical density of the hydrogen peroxide solution before the extract was introduced;
D2 – optical density of the hydrogen peroxide solution after incubation with the extract;
V1 – total extract volume, cm3;
n – amount of hydrogen peroxide introduced, mmol;
m – weight of oyster mushroom sublimate in the extract, g;
V2 – volume of extract for analysis, cm3;
t – incubation time, min.
The anti-inflammatory activity of the oyster mushroom sublimates was determined in vitro. It employed the method used for assessing the osmotic resistance of erythrocyte membranes [36]. The dry mushroom sublimate was made into powder, suspended in distilled water at a concentration of 5.0 mg/cm3, and incubated at 4°C for 12 h. The suspension was centrifuged at 7000 rpm for 10 min, after which the supernatant was filtered. Blood was obtained from healthy white laboratory Wistar rats and mixed in a 1:1 ratio with Alsever solution. The latter contained equal volumes of aqueous solutions of 2% dextrose, 0.8% sodium citrate, 0.5% citric acid, and 0.42% sodium chloride. The resulting solution was centrifuged at 4000 rpm for 10 min. The precipitated cells were washed with physiological saline and centrifuged three time until the red blood cells were 10% by suspension volume in physiological saline. The extracts of oyster mushroom sublimates were separately mixed with 1.0 cm3 of phosphate buffer, 2.0 cm3 of hypotonic sodium chloride solution (0.42%), and 0.5 cm3 of red blood cell suspension. The control sample contained 2.0 cm3 of distilled water instead of the hypotonic solution. The mixes were incubated at 37°C for 20 min and centrifuged at 3000 rpm.
After that, the supernatant liquid was decanted, and the haemoglobin content was estimated using a spectrophotometer at λ = 560 nm. The percentage of resistance of red cell membranes was assessed based on the fact that the haemolysis obtained in the control sample was 100%. It was calculated by the formula:
To assess the thrombolytic activity of the sublimates, blood obtained from white Wistar rats was distributed into different pre-weighed sterile microcentrifuge tubes (0.5 cm3 in each) and incubated at 37°C for 45 min. After the clot was formed, the serum was completely removed without disturbing the clot, and each tube was again weighed to calculate the weight of the clot. 100 μL of sublimate extract was added into each tube with a preweighed clot. All tubes were incubated at 37°C for 90 min. After incubation, the released liquid was removed, and the tubes were weighed again. The difference in weight before and after clot dissolution was expressed as a percentage [37].
The content of substances and their activity were expressed in terms of absolute dry raw materials. All quantitative parameters were triplicated. The results were recorded as arithmetic mean ± standard error of the arithmetic mean (M ± m) and subjected to statistical processing using the method of one-way ANOVA test and the Biostat software (version 4.03). The significance of the differences was measured at P ≤ 0.05.
РЕЗУЛЬТАТЫ И ИХ ОБСУЖДЕНИЕ
A single-phase ANOVA was conducted to compare the quantitative values of the properties responsible for the antiatherogenic potential of the freeze-dried oyster mushroom product. It also made it possible to check whether there was any significant difference in these properties after various pre-treatment methods.
As a potential antiatherogenic product, the oyster mushrooms were checked for the concentration of lovastatin. This natural statin reduces the production of endogenous cholesterol as it inhibits the activity of hydroxyl-3-methyluracil-coenzyme reductase [38]. The experiment took into account the level of antioxidant and catalase activities that resist the accumulation of excess reactive oxygen. Together with excessive lipids in the blood, reactive oxygen is known to cause atherosclerosis [39]. We tested the abilities of freeze-dried oyster mushrooms to inhibit the inflammation and thrombosis. They are considered the key pathogenetic mechanisms of atherosclerosis as they facilitate the transformation of risk factors into morphological changes [40].
A set of experiments was performed to define the effect of disinfection, cryoprotection, and various degrees of preliminary grinding on the safety and activity of the abovementioned properties.
Thickness, shape, and volume ratio of samples are known to affect the drying rate and quality of the finished product [41].
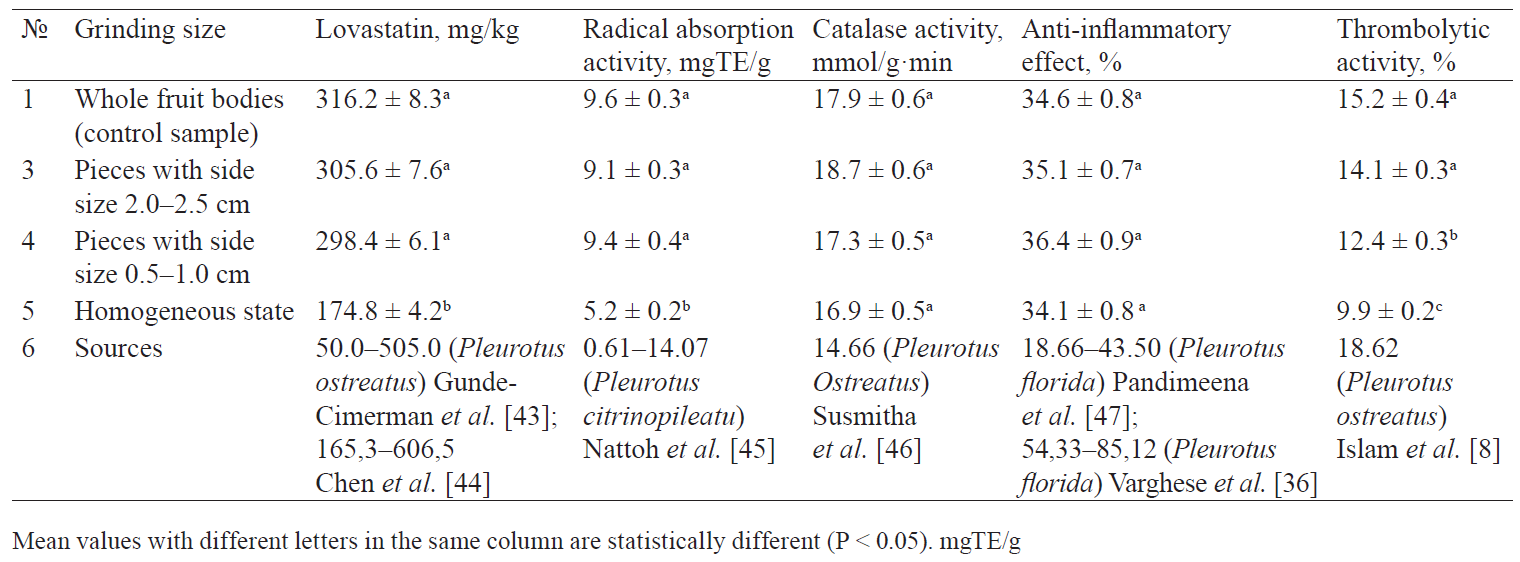
The whole dried oyster mushroom fruit bodies selected as control sample were tested for the abovementioned properties. The obtained quantitative indicators appeared to be comparable with the data for Pleurotus mushrooms presented in previous studies (Table 1). When the fruit bodies were ground to pieces with side sizes of 2.0–2.5 cm and 0.5–1.0 cm at the pretreatment stage, it did not affect the concentration of lovastatin in the freeze-dried product. However, the content of lovastatin in the homogenised sublimates turned out to be 45% less than in the whole-dried samples. The technological process of homogenisation probably reduced the degree of heterogeneity of the distribution of chemicals and phases by volume. It might have changed the sensitivity of lovastatin to the conditions of the subsequent stages of lyophilisation. In addition, homogenisation is known to cause a shift in the pH of raw materials. According to Piecha, when pH of the medium increases, the lactone structures of statins can be partially or completely converted to the corresponding forms of hydroxyacids [42].
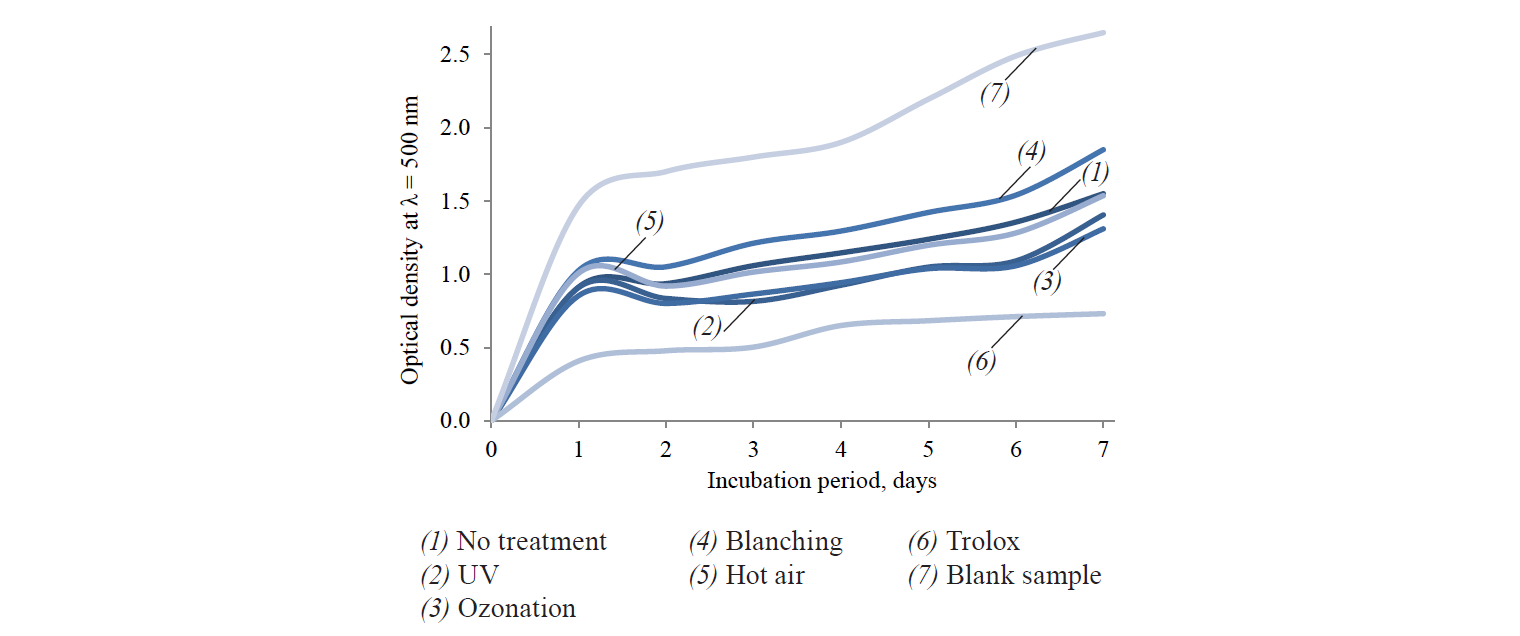
The assessment of the effect of grinding size on the preservation of antioxidant properties had a similar result. Homogenization reduced the activity of radical absorption by almost 46%. The homogenised samples also demonstrated minimal LPO inhibition activity (Fig. 1)
The data were consistent with some studies that featured the effect of homogenisation during freezedrying of berries [19]. According to Paciulli et al., the results may be explained by the fact that large tissue damage caused a loss of antioxidant substances [48].
However, the preliminary grinding of oyster mushroom affected neither catalase nor antiinflammatory properties of its sublimates.
Pleurotus mushrooms owe their thrombolytic properties to the high level of biosynthesis of the protease enzyme complex. Proteases have an affinity for fibrin and cause its lysis [49]. The thrombolytic properties of freeze-dried oyster mushrooms depended on the degree of grinding at the pre-treatment stage: the experiment showed a statistically significant decrease as the fruit bodies were ground into smaller pieces. The samples subjected to preliminary homogenisation demonstrated the lowest thrombolytic activity. Such result might be connected with the fact that cellular disruption facilitates interaction between proteolytic enzymes and extracellular protease inhibitors.
Although pre-treatment grinding may facilitate the drying process, it proved irrational in terms of preservation of lovastatin and other antioxidant and thrombolytic substances [41].
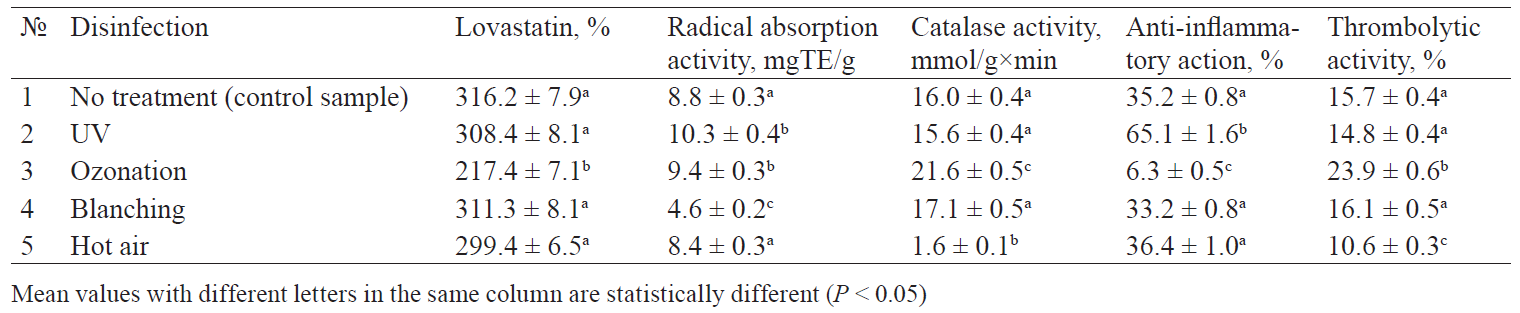
Food security is as important as its nutritional and biological value. Microbiological contamination is an indicator of food security. Therefore, disinfection is a necessary pre-treatment stage. Blanching and hot air treatment had no statistically significant effect on the content of lovastatin in the finished sublimates if compared with the control samples (Table 2). UV disinfection may cause photodegradation of statins. However, it also demonstrated no significant changes in the concentration of lovastatin in the sublimates. The only difference was a slight decrease in the content of lovastain, which is consistent with the results obtained by [42], according to which lovastatin proved to be the most UV resistant statin.
The ozonation resulted in a significant loss of lovastatin. Its concentration in the sublimates decreased by 31.4% compared to the control samples, which confirmed the data published in [50], according to which oxygen makes lovastatin instable.
UV disinfection and ozonation resulted in a higher radical absorption and LPO inhibition (Fig. 2). Such results are consistent with other studies [51–53] that proved a better preservation, and sometimes even an increase, of antioxidant substances in mushrooms and fruits after ozonation and UV disinfection. According to Sudheer et al., ozone can trigger the formation of such secondary metabolites as phenols and flavonoids [54].
Hot air disinfection caused no statistically significant changes in the antiradical activity and LPO inhibition (Fig. 2). These results contradicted with those described in [55]. On the one hand, the effect might be explained by the thermally induced extraction of previously bound or polymerized molecules of antioxidants, in particular, phenols. On the other hand, it may be due to the inactivation of enzymes involved in their catabolism, as demonstrated by recent studies of vegetable drying processes [56]. In addition, the obtained results might be explained by the fact that hot air treatment can trigger the formation of new compounds, e.g. Maillard reaction products, which possess good antioxidant properties [57].
The blanching produced a significant decrease in the antiradical activity of the sublimates. Its value was 47.7% lower than that of the control sample. The results confirmed the data described in [58, 59]. According to Lam et al. and Radzki et al., leaching and a low ability to absorb oxygen radicals resulted in a lower concentration of antioxidant substances after preliminary blanching. In addition, the blanched sublimates showed a minimal LPO inhibition [58, 59].
Various disinfection methods produced different results on the level of catalase activity of the sublimates. Hot-air treatment resulted in the lowest catalase activity. These results confirmed those described in [60], according to which a higher drying temperature reduced the residual activity of the oyster mushroom catalase enzyme.
The ozonation produced the highest catalase activity. These results confirmed those described in [61, 62], which showed an increase in the catalase activity of fruits after ozonation. The increase was explained by the fact that ozone came into contact with the biological tissue of mushrooms and caused oxidative stress, which was accompanied by activation of various antioxidant enzyme systems, e.g. catalase.
Contrary to previous assumptions [63], the blanching caused no changes in the level of catalase activity. According to Egbebi et al., blanching of mushrooms inactivated catalase. In our opinion, it can be explained by the relatively short blanching time [63]. This presumption corresponds with the observations published in [64], which described catalase inactivation only in cases when blanching lasted > 10 min.
Various disinfection methods produced various effects on the anti-inflammatory activity of the product that had undergone a spray-freeze drying. The antiinflammatory properties were determined using the erythrocyte membrane stabilisation test. The UV-treated samples showed the highest anti-inflammatory activity. Mushrooms owe most of their anti-inflammatory properties to polysaccharides, especially glucans [65, 66]. Thus, UV treatment served as an elicitor that increases production of extracellular polysaccharides in mushrooms. In addition, UV treatment might have produced phenolic compounds that produce a protective effect on biological membranes [67].
The ozonation resulted in the lowest antiinflammatory activity. This effect might have been caused by ozone-induced oxidative reactions. According to Mzoughi et al., ozone-induced oxidative reactions lead to the selective depolymerisation of polysaccharides, followed by a possible increase or, conversely, a decrease in their biological activity [68].
The thrombolytic properties also proved to depend on the methods of preliminary disinfection. Thus, the maximum thrombolytic properties were manifested in the ozonised samples. This result might have been caused by the ability of ozone to inactivate protease inhibitors [69]. The minimal thrombolytic properties were detected in the samples that had been treated with hot air. According to Ali et al., protease inhibitors in mushrooms are thermally stable [70]. According to Rai et al. and de Castro et al., proteases demonstrate the maximum activity at 55–60°С and may be wasted on the autohydrolysis of proteins heating [71, 72].
Freezing is an obligatory stage of freeze-drying. Freezing can damage the cell structure with ice crystals. The degree of damage depends on the size of the crystals and the heat transfer rate. It can affect the rheological and textural properties of the product, as well as redox processes in favour of oxidation. As a result, the number and the biological activity of the substances in the product may change. Therefore, a stabilising cryoprotector should be applied before freezing. Cryoprotectors maintain the quality of the frozen product by increasing the reversibility of the process and preserving BAS [73]. In this regard, we assessed various natural cryoprotectants and their effect on the anti-atherogenic properties of the freeze-dried oyster mushrooms (Table 3).
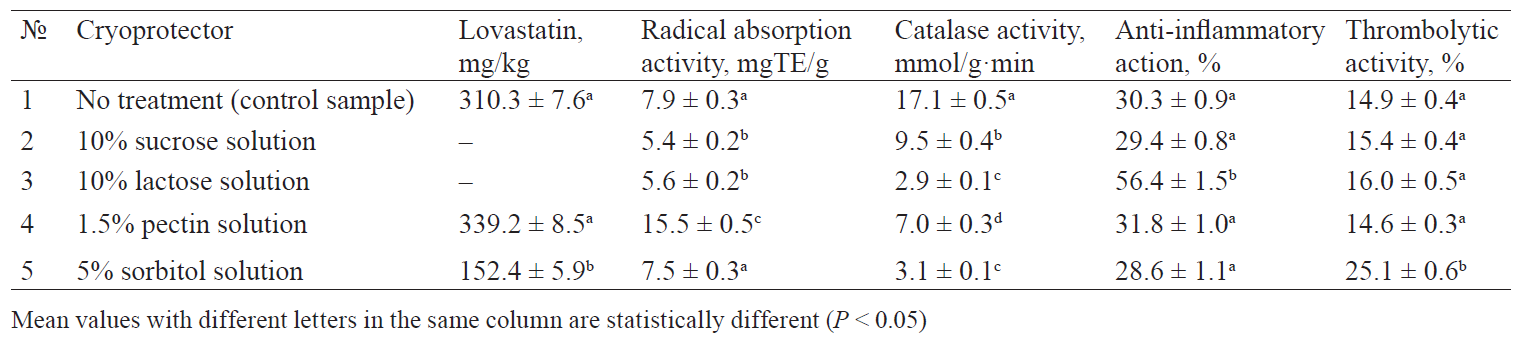
The chemical analysis demonstrated that the cryoprotectors had a different effect on the amount of lovastatin in the mushroom sublimates. The samples pretreated with a 1.5% solution of pectin showed no statistical changes in the concentration of oxidation-sensitive natural statin. However, there was a clear tendency to its increase. It confirmed the results published in [74], which proved that insoluble polysaccharides effectively inhibit oxidation processes in frozen semi-finished products.
The samples pretreated with a 5% sorbitol solution demonstrated a 47.6% decrease in lovastatin. The samples pretreated with a 10% sucrose solution and a 10% lactose solution appeared to contain no lovastatine. It was probably due to the hydrolysis of lovastatine in the aqueous medium of the cryoprotectors. Thus, freezing did not prove to be a limiting factor with respect to lovastatin concentration. Hence, cryoprotectors are not obligatory in this aspect.
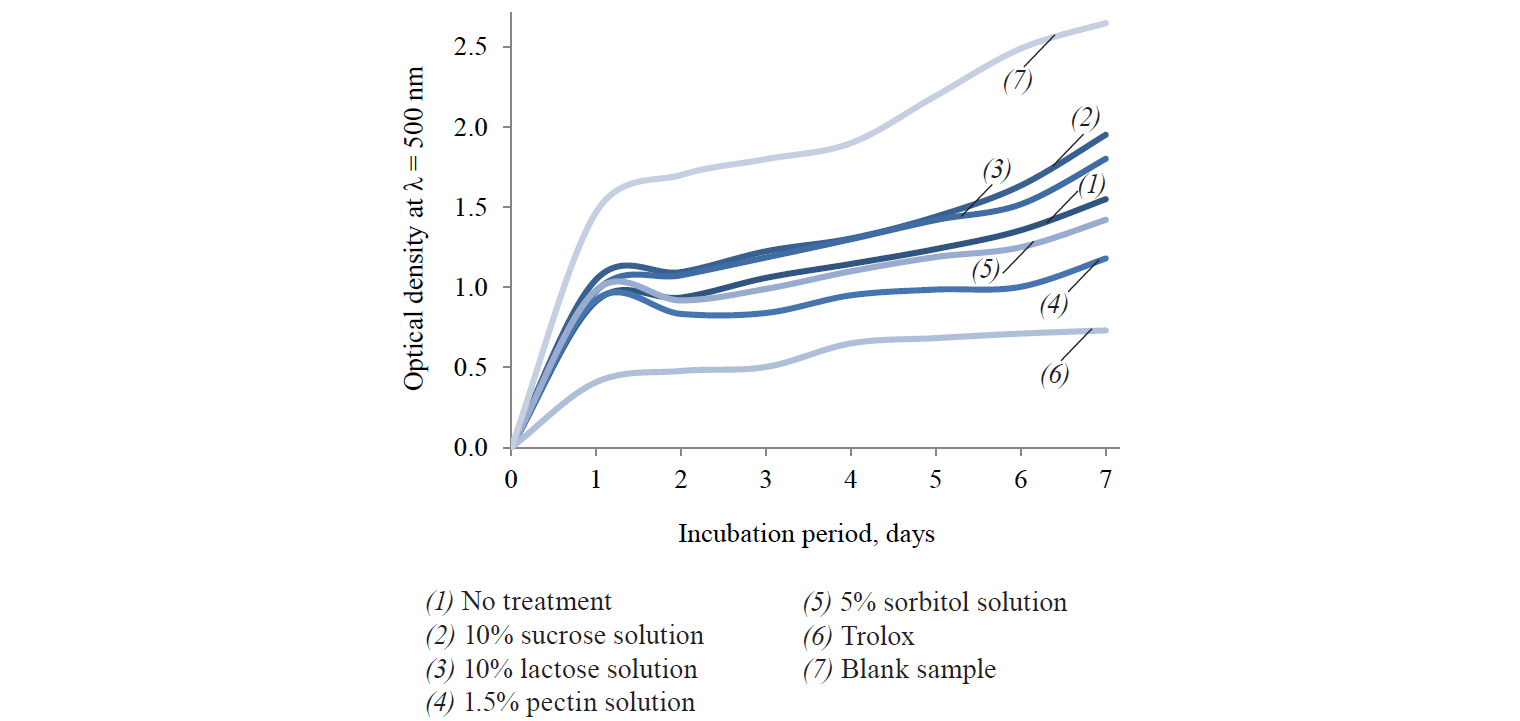
1.5% pectin solution proved to be the best cryoprotector for oyster mushrooms as it ensured the maximum preservation of antiradical and LPO inhibition (Fig. 3). The results were consistent with [75], according to which pectin has a greater water absorption capacity compared with sorbitol and monosaccharides. The results may be attributed to the antioxidant properties of pectin itself, since its diffused part could enhance the antioxidant properties of the obtained dry product. According to Kopjar et al., if added to bioactive substances containing phenolic compounds, pectin provides a synergistic effect on their antioxidant properties [76].
10% solutions of lactose and sucrose resulted in a significant decrease in the level of antiradical activity and LPO inhibition. On the one hand, these results may be explained by the extraction of antioxidant substances into the aqueous solutions of the cryoprotectors. On the other hand, the decrease might have been caused by the cryoprotective effect itself, since it reduces both ice crystal formation in the mushroom tissue matrix and damage to the cell structure. According to Yang et al., cryoprotective effect increases extraction of antioxidant substances, e.g. phenolic compounds, from cells [77].
Cooling is known to reduce the catalase properties of certain substances [78]. However, catalase activity decreased significantly in all the experimental samples. Its value was minimal in the sublimates pretreated with a 10% lactose solution and a 5% sorbitol solution.
The maximum anti-inflammatory activity was manifested in the sublimates pretreated with a 10% lactose solution. The results confirmed those published in [79], according to which lactose proved to be a more advantageous cryoprotector than sorbitol or sucrose when used in freeze-drying of liposome preparations. It can be explained by the fact that disaccharides produce a greater stabilising effect on cell membranes during freezing than other cryoprotectors, thereby preserving polysaccharides and glycoproteins of cell membranes.
As for the thrombolytic properties, sublimates pretreated with sorbitol showed the best results. Unlike mono-, di-, and oligosaccharides, sorbitol can penetrate into cells [80]. It protects intracellular proteases and their fibrinolytic properties from possible denaturation caused by low temperature.
ВЫВОДЫ
A set of biochemical experiments was performed to study the effect of various pre-treatment conditions on the biologically active properties that provide the antiatherogenic potential of freeze-dried oyster mushrooms. The antiatherogenic properties under study included the content of natural statin (lovastatin), as well as antioxidant, catalase, anti-inflammatory, and thrombolytic properties. The results showed that each pre-treatment method produced a different effect on the abovementioned properties of the freeze-dried product.
The experiments demonstrated that the best results for lovastatin were obtained when the raw material was ground to pieces with a side size of ≥ 0.5 cm, subjected to UV disinfection, blanched, treated with hot air, and cryoprotected with a 1.5% pectin solution.
As for the antioxidant properties, such as radical absorption and LPO inhibition, the best conditions included UV disinfection, ozonation, and cryoprotection with a 1.5% pectin solution. Homogenisation, blanching, and cryostabilisation with 10% solutions of sucrose and lactose were found critical for antioxidant properties.
The catalase activity of the product did not depend on the degree of grinding, blanching, and UV disinfection. It was maximal after ozonation. The list of critical pretreatment conditions included hot air treatment and exposure to all the cryoprotectors except pectin.
The anti-inflammatory properties were best preserved after UV disinfection and cryoprotection with a 10% lactose solution. Ozonation appeared to be the only critical pre-treatment factor.
The best results for thrombolytic properties were obtained when the oyster mushrooms were ozonated and cryoprotected using a 5% sorbitol solution. Critical factors included homogenisation and hot air treatment.
Thus, the experiments revealed advantages of individual pre-treatment conditions and their combinations. The applied conditions can turn freezedried oyster mushrooms into a functional food product or ingredient. The new functional product significantly improved the properties that affect such pathogenetic factors of atherogenesis as hyperlipidemia, oxidative stress, inflammatory reaction, and thrombosis.
КОНФЛИКТ ИНТЕРЕСОВ
The authors declare that there is no conflict of interests related to this article.
СПИСОК ЛИТЕРАТУРЫ
- Benjamin EJ, Blaha MJ, Chiuve SE, Cushman M, Das SR, Deo R, et al. Heart Disease and Stroke Statistics-2017 Update A Report From the American Heart Association. Circulation. 2017;135(10):E146–E603. DOI: https://doi.org/10.1161/CIR.0000000000000485.
- Mallika V, Goswami B, Rajappa M. Atherosclerosis Pathophysiology and the Role of Novel Risk Factors: A Clinicobiochemical Perspective. Angiology. 2007;58(5):513–522. DOI: https://doi.org/10.1177/0003319707303443.
- Mukhammed AA, Maksimov ML. Statiny: pobochnye ehffekty (nauchnyy obzor) [Statins: side effects (a scientific review)]. Terapevt [Therapist]. 2013;(7):78 84. (In Russ.).
- Mitina SS, Piskov SI, Koldunov IA. K voprosu o poiske alʹternativnykh gipolipidemicheskikh sredstv na osnove syrʹya prirodnogo proiskhozhdeniya [Alternative lipid-lowering drugs based on natural raw materials]. ‘Fiziologicheskie problemy adaptatsii’: sbornik nauchnykh statey mezhdunarodnoy konferentsii, posvyashchennoy 85-letiyu SKFU, 45-letiyu kafedry anatomii i fiziologii, edineniyu nauchnogo soobshchestva fiziologov Rossii i Respubliki Belarus [‘Physiological Problems of Adaptation’: Proceedings of the International conference dedicated to the 85th anniversary of the North Caucasus Federal University, the 45th anniversary of the Department of Anatomy and Physiology, and the union of the scientific community of physiologists of Russia and the Republic of Belarus]; 2015; Stavropol. Stavropol: North Caucasus Federal University; 2015. p. 111–113. (In Russ.).
- Singh SP, Sashidhara KV. Lipid lowering agents of natural origin: An account of some promising chemotypes. European Journal of Medicinal Chemistry. 2017;140:331–348. DOI: https://doi.org/10.1016/j.ejmech.2017.09.020.
- Carrasco-González JA, Serna-Saldívar SO, Gutiérrez-Uribe JA. Nutritional composition and nutraceutical properties of the Pleurotus fruiting bodies: Potential use as food ingredient. Journal of Food Composition and Analysis. 2017;58:69–81. DOI: https://doi.org/10.1016/j.jfca.2017.01.016.
- Amirullah NA, Abidin NZ, Abdullah N. The potential applications of mushrooms against some facets of atherosclerosis: A review. Food Research International. 2018;105:517–536. DOI: https://doi.org/10.1016/j.foodres.2017.11.023.
- Islam MR, Uddin MM. In vitro Doses and Incubations Dependent Thrombolytic Potential Study of Edible Mushrooms Pleurotus ostreatus, Ganoderma lucidum and Lentinula edodes Available in Bangladesh. British Journal of Pharmaceutical Research. 2015;7(1):44–51. DOI: https://doi.org/10.9734/BJPR/2015/18227.
- Zhang Y, Wang ZW, Jin G, Yang XD, Zhou HL. Regulating dyslipidemia effect of polysaccharides from Pleurotus ostreatus on fat-emulsion-induced hyperlipidemia rats. International Journal of Biological Macromolecules. 2017;101:107–116. DOI: https://doi.org/10.1016/j.ijbiomac.2017.03.084.
- Karomatov ID, Salomova MF. Medical value of the oyster mushroom. Biologiya i integrativnaya meditsina [Biology and Integrative Medicine]. 2017;(9):78–88. (In Russ.).
- Muszynska B, Grzywacz-Kisielewska A, Kala K, Gdula-Argasinska J. Anti-inflammatory properties of edible mushrooms: A review. Food Chemistry. 2018;243:373–381. DOI: https://doi.org/10.1016/j.foodchem.2017.09.149.
- Guillamon E, Garcia-Lafuente A, Lozano M, D’Arrigo M, Rostagno MA, Villares A, et al. Edible mushrooms: Role in the prevention of cardiovascular diseases. Fitoterapia. 2010;81(7):715–723. DOI: https://doi.org/10.1016/j.fitote.2010.06.005.
- Elsayed EA, El Enshasy H, Wadaan MAM, Aziz R. Mushrooms: A Potential Natural Source of Anti-Inflammatory Compounds for Medical Applications. Mediators of Inflammation. 2014;2014. DOI: https://doi.org/10.1155/2014/805841.
- Xu J-G, Duan J-L. Effects of Drying Methods on Physico-Chemical Properties and Antioxidant Activity of Shiitake Mushrooms (Lentinus Edodes). Agriculture and Food Sciences Research. 2015;2(2):51–55.
- Piskov SI, Timchenko LD, Rzhepakovsky IV, Avanesyan SS, Sizonenko MN, Areshidze DA, et al. The influence of the drying method for food properties and hypolidemic potential of oyster mushrooms (Pleurotus ostreatus). Problems of Nutrition. 2018;87(2):65–76. (In Russ.). DOI: https://doi.org/10.24411/0042-8833-2018-10020.
- Arshinova OYu, Oborotova NA, Sanarova EV. Vspomogatelʹnye veshchestva v tekhnologii liofilizatsii lekarstvennykh preparatov [Excipients in the technology of lyophilisation of drugs]. Drug development and registration. 2013;2(1):20–25. (In Russ.).
- Rabeta MS, Lin SP. Effects of Different Drying Methods on the Antioxidant Activities of Leaves and Berries of Cayratia trifolia. Sains Malaysiana. 2015;44(2):275–280. DOI: https://doi.org/10.17576/jsm-2015-4402-16.
- Karam MC, Petit J, Zimmer D, Djantou EB, Scher J. Effects of drying and grinding in production of fruit and vegetable powders: A review. Journal of Food Engineering. 2016;188:32–49. DOI: https://doi.org/10.1016/j.jfoodeng.2016.05.001.
- Rudy S, Dziki D, Krzykowski A, Gawlik-Dziki U, Polak R, Rozylo R, et al. Influence of pre-treatments and freezedrying temperature on the process kinetics and selected physico-chemical properties of cranberries (Vaccinium macrocarpon Ait.). LWT – Food Science and Technology. 2015;63(1):497–503. DOI: https://doi.org/10.1016/j.lwt.2015.03.067.
- Parniakov O, Bals O, Lebovka N, Vorobiev E. Pulsed electric field assisted vacuum freeze-drying of apple tissue. Innovative Food Science & Emerging Technologies. 2016;35:52–57. DOI: https://doi.org/10.1016/j.ifset.2016.04.002.
- Prosapio V, Norton I. Influence of osmotic dehydration pre-treatment on oven drying and freeze drying performance. LWT – Food Science and Technology. 2017;80:401–408. DOI: https://doi.org/10.1016/j.lwt.2017.03.012.
- Sheshma J, Raj JD. Effect of pre-drying treatments on quality characteristics of dehydrated tomato powder. International Journal of Research in Engineering & Advanced Technology. 2014;2(3).
- Al-Amin M, Sajjad Hossain M, Iqbal A. Effect of pre-treatments and drying methods on dehydration and rehydration characteristics of carrot. Universal Journal of Food and Nutrition Science. 2015;3(2):23–28. DOI: https://doi.org/10.13189/ujfns.2015.030201.
- Ando Y, Maeda Y, Mizutani K, Wakatsuki N, Hagiwara S, Nabetani H. Impact of blanching and freeze-thaw pretreatment on drying rate of carrot roots in relation to changes in cell membrane function and cell wall structure. LWT – Food Science and Technology. 2016;71:40–46. DOI: https://doi.org/10.1016/j.lwt.2016.03.019.
- Schneider I, Kressel G, Meyer A, Krings U, Berger RG, Hahn A. Lipid lowering effects of oyster mushroom (Pleurotus ostreatus) in humans. Journal of Functional Foods. 2011;3(1):17–24 DOI: https://doi.org/10.1016/j.jff.2010.11.004.
- Usall J, Ippolito A, Sisquella M, Neri F. Physical treatments to control postharvest diseases of fresh fruits and vegetables. Postharvest Biology and Technology. 2016;122:30–40. DOI: https://doi.org/10.1016/j.postharvbio.2016.05.002.
- Oner ME, Demirci A. Ozone for Food Decontamination: Theory and Applications. In: Lelieveld H, Holah J, Gabrić D. Handbook of Hygiene Control in the Food Industry (Second Edition). Woodhead Publishing; 2016. pp. 491–501. DOI: https://doi.org/10.1016/B978-0-08-100155-4.00033-9.
- Galoburda R, Kuka M, Cakste I, Klava D. The effect of blanching temperature on the quality of microwave-vacuum dried mushroom Cantharellus cibarius. Agronomy Research. 2015;13(4):929–938.
- Escriche I, Serra JA, Gomez M, Galotto MJ. Effect of ozone treatment and storage temperature on physicochemical properties of mushrooms (Agaris bisporus). Food Science and Technology International. 2001;7(3):251–258. DOI: https://doi.org/10.1106/6a9r-dkev-adv7-y30x.
- Avanesyan SS, Timchenko LD, Piskov SI, Rzhepakovskiy IV, Koldunov IA, Kolosov AV. Razrabotka metodiki kolichestvennogo opredeleniya lovastatina v plodovom tele veshenki obyknovennoy (Pleurotus ostreatus) [New method of quantitative evaluationof lovastatin in the fruit body of common oyster mushroom (Pleurotus ostreatus)]. Fiziko-khimicheskaya biologiya: sbornik trudov III mezhdunarodnoy nauchnoy internet-konferentsii [Physico-Chemical Biology: Proceedings of the III international scientific Internet conference]; 2015; Stavropol. Stavropol: Stavropol State Medical University; 2015. p. 32–35. (In Russ.).
- Avanesyan SS, Timchenko LD, Piskov SI, Kovalev DA. TLC determination of lovastatin. Sorption and chromatographic processes. 2015;15(5):693 – 698. (In Russ.).
- Kanibolotskaya LV, Fedoseeva AA, Odaryuk ID, Polokhina II, Triskiba SD, Shendrik AN. Antioxidant activity of mycothalluses of edible fungі series. Nutrition Problems. 2008;19(3–4):35–38. (In Russ.).
- Oreshko NA, Kiselev PA, Yuraga TM, Kokhnovich NN, Kamyshnikov VS. Razrabotka test-sistem i opredelenie antiradikalʹnoy aktivnosti biologicheskikh zhidkostey i farmatsevticheskikh substantsiy prirodnogo i sinteticheskogo proiskhozhdeniya [Development of test systems and determination of the antiradical activity of biological fluids and pharmaceutical substances of natural and synthetic origin]. Free Radicals in Chemistry and Life: Book of Abstracts of the International Conference; 2015; Minsk. Minsk: Belarusian State University; 2015. p. 122–124. (In Russ.).
- Kimatu BM, Zhao LY, Biao Y, Ma GX, Yang WJ, Pei F, et al. Antioxidant potential of edible mushroom (Agaricus bisporus) protein hydrolysates and their ultrafiltration fractions. Food Chemistry. 2017;230:58–67. DOI: https://doi.org/10.1016/j.foodchem.2017.03.030.
- Velichko AK, Solovev VB, Gengin MT. Methods of laboratory definition of the common peroxide of destroying activity of enzymes of plants. Izvestia Penzenskogo gosudarstvennogo pedagogicheskogo universiteta imeni V.G. Belinskogo. 2009;(18):44–48. (In Russ.).
- Varghese R, Bushra M, Nair NM, Jacob JT, Thomas SM, Leena PN. Studies on preliminary phytochemical, antioxidant and anti-inflammatory activities of Pleurotus florida by in vitro method. International Journal of Pharmacy and Technology. 2015;7(3):9945–9964.
- Sharmila G, Brindha D. In vitro antioxidant and antithrombotic activity of Cleome viscosa. International Journal of Current Research. 2017;9(05):49750–49754.
- Frishman WH, Rapier RC. Lovastatin: An HMG-CoA Reductase Inhibitor for Lowering Cholesterol. Medical Clinics of North America. 1989;73(2):437–448. DOI: https://doi.org/10.1016/S0025-7125(16)30681-2.
- Tsioufis C, Mantzouranis E, Kalos T, Konstantinidis D, Tousoulis D. Risk Factors of Atherosclerosis: Pathophysiological Mechanisms. In: Tousoulis D, editor. Coronary Artery Disease. Academic Press; 2018. pp. 43–66. DOI: https://doi.org/10.1016/B978-0-12-811908-2.00004-0.
- Tsybulkin NA, Tukhvatullina GV, Tsybulkina VN, Abdrakhmanova AI. Inflammatory mechanisms in pathogenesis of atherosclerosis. Practical medicine. 2016;96(4–2):165–169. (In Russ.).
- Hassan N, ElHana A. Microwave drying of apple. Misr Journal of Agricultural Engineering. 2008;25(3):980–1003.
- Piecha M. Stability and degradation studies of cholesterol-lowering statin drugs. Nova Gorica: University of Nova Gorica; 2009. 129 p.
- Gunde-Cimerman N, Cimerman A. Pleurotus Fruiting Bodies Contain the Inhibitor of 3-Hydroxy-3-Methylglutaryl-Coenzyme-A Reductase-Lovastatin. Experimental Mycology. 1995;19(1):1–6. DOI: https://doi.org/10.1006/emyc.1995.1001.
- Chen S-Y, Ho K-J, Hsieh Y-J, Wang L-T, Mau J-L. Contents of lovastatin, gamma-aminobutyric acid and ergothioneine in mushroom fruiting bodies and mycelia. LWT – Food Science and Technology. 2012;47(2):274–278. DOI: https://doi.org/10.1016/j.lwt.2012.01.019.
- Nattoh G, Gatebe E, Musieba F, Mathara J. Bioprospecting optimal phenology for bioactive molecules in native golden yellow Pleurotus citrinopileatus Singer. Asian Pacific Journal of Tropical Biomedicine. 2016;6(2):132–142. DOI: https://doi.org/10.1016/j.apjtb.2015.10.012.
- Susmitha S, Vidyamol KK, Ranganayaki P, Vijayaraghavan R. Purification of Catalase Enzyme from Pleurotus Ostreatus. International Journal of Applied Biology and Pharmaceutical Technology. 2014;5(1):28–34.
- Pandimeena M, Prabu M, Sumathy R, Kumuthakalavalli R. Evaluation of phytochemicals and in vitro antiinflammatory, anti-diabetic activity of the white Oyster mushroom, Pleurotus florida. International Research Journal of Pharmaceutical and Applied Sciences. 2015;5(1):16–21.
- Paciulli M, Ganino T, Pellegrini N, Rinaldi M, Zaupa M, Fabbri A, et al. Impact of the industrial freezing process on selected vegetables – Part I. Structure, texture and antioxidant capacity. Food Research International. 2015;74:329–337. DOI: https://doi.org/10.1016/j.foodres.2014.04.019.
- Inacio FD, Ferreira RO, de Araujo CAV, Brugnari T, Castoldi R, Peralta RM, et al. Proteases of Wood Rot Fungi with Emphasis on the Genus Pleurotus. Biomed Research International. 2015;2015. DOI: https://doi.org/10.1155/2015/290161.
- Javernik S, Kreft S, Strukelj B, Vrecer F. Oxidation of lovastatin in the solid state and its stabilization with natural antioxidants. Pharmazie. 2001;56(9):738–740.
- Jiang TJ, Jahangir MM, Jiang ZH, Lu XY, Ying TJ. Influence of UV-C treatment on antioxidant capacity, antioxidant enzyme activity and texture of postharvest shiitake (Lentinus edodes) mushrooms during storage. Postharvest Biology and Technology. 2010;56(3):209–215. DOI: https://doi.org/10.1016/j.postharvbio.2010.01.011.
- Yeoh WK, Ali A, Forney CF. Effects of ozone on major antioxidants and microbial populations of fresh-cut papaya. Postharvest Biology and Technology. 2014;89:56–58. DOI: https://doi.org/10.1016/j.postharvbio.2013.11.006.
- Wang Q, Chu LJ, Kou LP. UV-C Treatment maintains quality and delays senescence of oyster mushroom (Pleurotus ostreatus). Scientia Horticulturae. 2017;225:380–385. DOI: https://doi.org/10.1016/j.scienta.2017.07.019.
- Sudheer S, Yeoh WK, Manickam S, Ali A. Effect of ozone gas as an elicitor to enhance the bioactive compounds in Ganoderma lucidum. Postharvest Biology and Technology. 2016;117:81–88. DOI: https://doi.org/10.1016/j.postharvbio.2016.01.014.
- Kamel SM, Thabet HA, Algadi EA. Influence of Drying Process on the Functional Properties of Some Plants. Chemistry and Materials Research. 2013;3(7).
- Kapoor S, Aggarwal P. Drying Method Affects Bioactive Compounds and Antioxidant Activity of Carrot. International Journal of Vegetable Science. 2014;21(5):467–481. DOI: https://doi.org/10.1080/19315260.2014.895474.
- Amarowicz R. Antioxidant activity of Maillard reaction products. European Journal of Lipid Science and Technology. 2009;111(2):109–111. DOI: https://doi.org/10.1002/ejlt.200900011.
- Lam YS, Okello EJ. Determination of Lovastatin, beta-glucan, Total Polyphenols, and Antioxidant Activity in Raw and Processed Oyster Culinary-Medicinal Mushroom, Pleurotus ostreatus (Higher Basidiomycetes). International Journal of Medicinal Mushrooms. 2015;17(2):117–128. DOI: https://doi.org/10.1615/IntJMedMushrooms.v17.i2.30.
- Radzki W, Ziaja-Soltys M, Nowak J, Rzymowska J, Topolska J, Slawinska A, et al. Effect of processing on the content and biological activity of polysaccharides from Pleurotus ostreatus mushroom. LWT – Food Science and Technology. 2016;66:27–33. DOI: https://doi.org/10.1016/j.lwt.2015.10.016.
- Arumuganathan T, Manikantan MR, Indurani C, Rai RD, Kamal S. Texture and quality parameters of oyster mushroom as influenced by drying methods. International Agrophysics. 2010;24(4):339–342.
- Yaseen T, Ricelli A, Albanese P, Nicoletti I, Essakhi S, Carboni C, et al. Influence of postharvest ozone treatment on decay, catalase, lipoxygenase activities, and anthocyanin content of ‘Redglobe’ table grapes. IOA-EA3G International Conference Ozone and Related Oxidants for Water Treatment, Food Processing, Agriculture, Industry, Health and Environment; 2014; Dublin. Dublin: University College Dublin; 2014.
- Boonkorn P, Gemma H, Sugaya S, Setha S, Uthaibutra J, Whangchai K. Impact of high-dose, short periods of ozone exposure on green mold and antioxidant enzyme activity of tangerine fruit. Postharvest Biology and Technology. 2012;67:25–28. DOI: https://doi.org/10.1016/j.postharvbio.2011.12.012.
- Egbebi AO. and Fakoya S. Effects of various treatments on intrinsic properties of Agaricusbisporus. European Journal of Experimental Biology. 2014;4(6):15–21.
- Abdulaziz L, Yaziji S, Azizieh A. Effect of Preliminarily Treatments on Quality Parameters of Artichoke with Different Preservation Methods. International Journal of ChemTech Research. 2015;7(6):2565–2572.
- Du B, Lin CY, Bian ZX, Xu BJ. An insight into anti-inflammatory effects of fungal beta-glucans. Trends in Food Science & Technology. 2015;41(1):49–59. DOI: https://doi.org/10.1016/j.tifs.2014.09.002.
- Friedman M. Mushroom Polysaccharides: Chemistry and Antiobesity, Antidiabetes, Anticancer, and Antibiotic Properties in Cells, Rodents, and Humans. Foods. 2016;5(4). DOI: https://doi.org/10.3390/foods5040080.
- Bonarska-Kujawa D, Cyboran S, Zylka R, Oszmianski J, Kleszczynska H. Biological Activity of Blackcurrant Extracts (Ribes nigrum L.) in Relation to Erythrocyte Membranes. Biomed Research International. 2014;2014. DOI: https://doi.org/10.1155/2014/783059.
- Mzoughi Z, Chakroun I, Ben Hamida S, Rihouey C, Ben Mansour H, Le Cerf D, et al. Ozone treatment of polysaccharides from Arthrocnemum indicum: Physico-chemical characterization and antiproliferative activity. International Journal of Biological Macromolecules. 2017;105:1315–1323. DOI: https://doi.org/10.1016/j.ijbiomac.2017.07.151.
- Kazachenko SYu, Bezrukih EG, Hohlova AI, Stupko TV, Matyushev VV, Plehanova LV. The equipment for dry spilt materials ozonation. The Bulletin of KrasGAU. 2009;29(2):184–189. (In Russ.).
- Ali PPM, Sapna K, Mol KRR, Bhat SG, Chandrasekaran M, Elyas KK. Trypsin Inhibitor from Edible Mushroom Pleurotus floridanus Active against Proteases of Microbial Origin. Applied Biochemistry and Biotechnology. 2014;173(1):167–178. DOI: https://doi.org/10.1007/s12010-014-0826-1.
- Rai RD, Arumuganathan T. Post harvest technology of mushrooms. Solan: National Research Centre for Mushroom; 2008. 84 p.
- de Castro RJS, Sato HH. Protease from Aspergillus oryzae: Biochemical Characterization and Application as a Potential Biocatalyst for Production of Protein Hydrolysates with Antioxidant Activities. Journal of Food Processing. 2014;2014. DOI: https://doi.org/10.1155/2014/372352.
- Syazin IE, Kasʹyanov GI. Fenomen krioobrabotki produktov [Phenomenon of cryoprocessing products]. Saarbrucken: Palmarium Academic Publishing; 2012. 296 p. (In Russ.).
- Glushkov O. Study of cryoprotectors effect on oxidation processes at storage of frozen half-finished products. Journal of Food Science and Technology-Ukraine. 2016;10(4). DOI: https://doi.org/10.15673/fst.v10i4.248.
- Keniyz NV. Vidy krioprotektorov, ispolʹzuemykh pri zamorazhivanii khlebobulochnykh polufabrikatov [Types of cryoprotectors used in the freezing of semi-finished bakery products]. Young Scientist. 2014;(18):236 – 238. (In Russ.).
- Kopjar M, Lončarić A, Pichler A. Influence of disaccharides and pectin addition on antioxidant activity of phenolic. Journal of Nutrition & Food Sciences. 2016;6(7). DOI: https://doi.org/10.4172/2155-9600.C1.032.
- Yang J, Chen J-F, Zhao Y-Y, Mao L-C. Effects of Drying Processes on the Antioxidant Properties in Sweet Potatoes. Agricultural Sciences in China. 2010;9(10):1522–1529. DOI: https://doi.org/10.1016/S1671-2927(09)60246-7.
- Wang CY. Effect of temperature preconditioning on catalase, peroxidase, and superoxide-dismutase in chilled zucchini squash. Postharvest Biology and Technology. 1995;5(1–2):67–76. DOI: https://doi.org/10.1016/0925-5214(94)00020-s.
- Shanskaya AI, Puchkova SM, Yakovleva TE, Ivanova RP. Vliyanie razlichnykh krioprotektorov na stabilʹnostʹ liofilizirovannykh liposom [Effect of various cryoprotectors on the stability of lyophilized liposomes]. Transfusiology. 2008;9(3):27–33. (In Russ.).
- Konov KB. Issledovanie metodami EHPR vozdeystviya krioprotektorov sakharozy, tregalozy, glitserina i sorbita na strukturu i dinamiku modelʹnoy lipidnoy membrany [EPR studies of the effect of cryoprotectors of sucrose, trehalose, glycerol, and sorbitol on the structure and dynamics of lipid membrane model]. Cand. phys. and math. sci. dis. Kazan: Kazan E. K. Zavoisky PhysicalTechnical Institute; 2016. 22 p.